TOXICITY, OXIDATIVE STRESS AND GENO-TOXICITY: LETHAL AND SUB-LETHAL EFFECTS OF THREE DIFFERENT INSECTICIDES MIXTURES ON CIRRHINA MRIGALA
H. Naz1*, S. Abdullah2, T. Ahmed3*, K. Abbas2, M. U. Ijaz2, S. Kumar1 and M. A. Hassan4
1Department of Zoology, Cholistan University of Veterinary and Animal Sciences, Bahawalpur, Pakistan
2Department of Zoology, Wildlife and Fisheries, University of Agriculture, Faisalabad, Pakistan
3Department of Life Sciences, Khwaja Fareed University of Engineering and Information Technology, Rahim Yar Khan, Pakistan
4Department of Parasitology, Cholistan University of Veterinary and Animal Sciences, Bahawalpur, Pakistan
Corresponding Author’s Email: dr.humanaz98@gmail.com, humanaz@cuvas.edu.pk, tanvirahmeduaf@gmail.com
ABSTRACT
In this study, 96 h LC50 and lethal values of three different mixtures of insecticides viz. endosulfan (E), chlorpyrfos (C) and bifenthrine (B) for Cirrhina mrigala were calculated by using Probit Analysis. The activities of enzymes viz. superoxide dismutase (SOD), peroxidase (POx), catalase (CAT) and glutathione S-transferase (GST) in organs and genotoxic parameters (DNA damage and micronuclei) in blood of fish exposed to acute and sub-lethal dose (1/3rd LC50) were also assessed. Analysis of Variance under completely randomized design (CRD) was applied to check the differences among organs for enzymes activities. The genotoxic data was compared by applying the non-parametric Mann-whitney U-test. Toxicity results showed that fish had higher tolerance limit against B+C mixture followed by B+E and C+E mixture. During both acute and chronic exposure activities of SOD, POx and GST significantly (P<0.05) increased in organs of stressed fish. The CAT activity showed some different trend it was increased in gills, liver and kidney of fish exposed to insecticides mixtures while it was decreased in brain, muscle and heart of fish. In both acute and chronic trails all the insecticides mixture induced significant damaged to DNA, MN and NAs in RBCs of C. mrigala. Maximum induction was observed due to C+E mixture of insecticides. During acute trail, NAs followed the order: DEN>BIN>MN>BN>DN>NN while in chronic order was as MN>DEN>BIN>BN>NN>DN. Duration dependent response showed that DNA damage, MN and NAs were increased with exposure period while in chronic exposure damage increased during first 30 days afterthat these were decreased.
Keywords: Fish, Toxicants, Enzymes, DNA damage, Organs
List of Abbreviations: Micronuclei=MN; Binucleated nuclei=BIN; Dumble nuclei=DN; Blebbed nuclei=BN; Notched nuclei=NN; Deshaped nuclei=DEN; Damaged nuclei=DN; Genetic Damage index=GDI
https://doi.org/10.36899/JAPS.2022.1.0421
Published online June 14, 2021
INTRODUCTION
Pesticides are widely applied to boost up the agriculture yield and in fish farms to kill the population of pests including insects and aquatic weeds, respectively. These synthetic chemicals ultimately find their way into natural aquatic system by spraying on target pest or through transfer of pesticides from soil. Some economically important non-target organism such as prawn and fish are most vulnerable to these pesticides (Saravanan et al., 2010). From last few decades, some insecticides like endosulfan an organochlorine (Piazza et al., 2015), chlorpyrifos an organophosphate (Anita et al., 2016) and bifenthrin an pyrethroid (Johnson et al., 2010) are most extensively applied in agriculture to control pests. Extensive use of these insecticides may cause the lethal and sub-lethal effects to fish depending upon the species, gender and life stage of organism (Majumder and Kaviraj, 2018). Lethal exposure cause mass mortalities in fish while sublethal effects include morphological, oxidative, biochemical, neurobehavioural, tissue damage, haematological and developmental abnormalities (Dar et al., 2015; Sunanda et al., 2016).
Pesticides induce toxicity by producing the reactive oxygen species (ROS) like super-oxide radicals, hydroxyl and hydrogen peroxide (Kumar et al., 2011); these can cause oxidation of lipid, protein, carbohydrates and nucleic acids (Kaur and Jindal, 2017). The key antioxidant enzymes for scavenging the ROS are superoxide dismutase, peroxidase, catalase and glutathione S-transferase. Superoxide dismutase transfers the super-oxide anion in to hydrogen peroxide (Modesto and Martinez, 2010). The hydrogen peroxide further converted into oxygen and water by catalase in peroxisomes (Mani et al., 2014) and peroxidase in mitochondria (Vijayakumar et al., 2016). Glutathione S-transferase belongs to phase II enzymes responsible for detoxification of xenobiotics.
Mutagenic and geno-toxic effects of pesticides can be assessed in apparently healthy aquatic animal like fish by genetic biomarkers. Among these genetic biomarkers, micronucleus (MN) test is most reliable assay due its simplicity and sensitivity for identifying DNA damage (Anbumani and Mohankumar, 2011). According to Da-Silva and Fontanetti (2006), nuclear abnormalities such as blebbed, lobed, notched nuclei and bi-nucleated cells can also serve as a good indicator of genotoxicity. Beside MN, comet assay is also being commonly applied to see the genotoxic consequences of toxicants in aquatic individuals (Yin et al., 2008). Both techniques have been effectively applied on fish blood as the fish possess nucleated red blood (Ventura-Campos de et al., 2008).
Extensive use of different insecticides at same time in agriculture means that insecticides are present in cocktail and organisms exposed to the mixture of insecticides rather than single. Therefore, this work was done to check the effect of different insecticides in mixture form on oxidative stress parameters and genotoxicity in Cirrhina mrigala exposed to acute and sub-lethal dose.
MATERIALS AND METHODS
Cirrhina mrigala were acquired from the fish seed Hatchery, Faisalabad and moved to the wet laboratory, University of Agriculture, Faisalabad and placed in smooth concrete tank to acclimatize the laboratory environment for a couple of weeks. The one gram of pure technical grade insecticides viz. endosulfan (E), chlorpyrifos (C) and bifenthrin (B), separately, mixed in analytical grade methanol (100 ml) to prepare stock-1 solution. However, mixtures (stock-II solutions) were ready by further dilutions of stock-I in deionized water by the ratio of 1:1.
Trial-I: Toxicity Assay: The toxicity tests (LC50 and lethal conc. for 96 h) of insecticides mixtures viz., B+C, B+E and C+E for C. mrigala (90-day old) were carried out. The experiment was started in February 2016. To carry out the toxicity test fish (n=10) were kept in 70-L glass aquaria (n=11) which were facilitated with automated air pump to provide air. The total hardness (223 mgL-1), pH (7.0) and temperature (28ᵒC) of water were stabilized throughout the toxicity trial. The mixtures concentration starts from zero with an increase of 0.01 μgL-1. During 96 h trial, mortality of fish was noticed after each 12 h to remove dead fish. To see the acute effects, fish were alone, kept in 96 h LC50 conc. of each insecticide mixture for 4-days. Fish sample (n=5) were collected after interval of 1-day. For control, fish were kept without any treatment.
Trial-II: Chronic Assay: Fish (n=20) were kept in sub-lethal conc. (1/3rd LC50) of each mixture for 60-days. In sub-lethal trial, sample of fish (n=5) was collected after 15-day interval. The unstressed fish (without pesticide) was labeled as a negative control (NC). In positive control (PC), fish was injected with cyclophosphamide (20 μgg-1 body weight). Each trial was carried out in triplicate and twenty fishes (n=20) were used for each test mixture.
Antioxidant Enzymes: After both acute and sub-lethal trial, superoxide dismutase (SOD), peroxidase (POx), catalase (CAT), and glutathione S-transferase (GST) were evaluated from muscle, gills, brain, liver, heart and kidney of fish. The phosphate buffer (0.2 M) of pH 7.0 was mixed in organs by the ratio of 1:4 (w/v) and homogenized for 10 minutes. The homogenates were filtered and centrifuged at 12,000 rpm and at 4ºC for 10 minutes. Following the centrifugation transparent supernatants were collected and stored at 80ºC for enzyme analyses. Giannopolitis and Ries (1977) method was followed to check the SOD activity. The CAT and POx activities were checked according to protocol described by Chance and Mehaly (1977). For GST activity Mannervik (1985) method was followed.
Comet Assay: A sterilized syringe was used to collect the blood from fish caudal vein and processed according to Singh et al. (1988) after each sampling. The damaged DNA was measured according to the Grover et al. (2003). Five types of damaged DNA known as “comet” according to the length of tail were evaluated. DNA damage was quantified by applying following formulae:
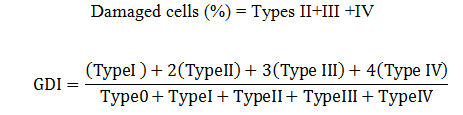
Micronucleus test: Barsiene et al. (2004) method was applied to prepare the slides for micronuclei (MN). Scoring of micronuclei and other nuclear anomalies were done according to Fenech et al. (2003). Following formula was applied to calculated MN frequency:
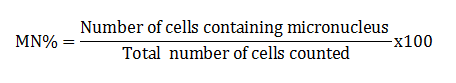
Data Analyses: Probit analysis was used to compute LC50, lethal conc. (96 h), and regression equation of insecticides mixtures. The software package Statistix (8.1 version) was used for statistical analyses. Statistical linear model Analysis of Variance under completely randomized design (CRD) up to three levels was applied to check the differences among organs for enzymes activities, treatments and duration of exposure followed by Tukey HSD for comparison of means. The P-value less than 0.05 was considered as significant (Steel et al., 1996). Genotoxic data was compared by applying the non-parametric Mann-whitney U-test.
RESULTS AND DISCUSSION
Toxicity Assay: Fish was more sensitive toward C+E mixture with mean LC50 and lethal conc. (96 h) of 1.269±0.03 and 4.86±0.15 μgL-1, respectively. Toxicity of mixtures towards fish followed the trend B+C<B+E<C+E (Figure 1). The widespread use of different forms of pesticides to improve agricultural production contributes to the existence of pesticides mixture in aquatic bodies (Belenguer et al., 2014; Masia et al., 2015).Aquatic pesticide contamination has caused unjustified fish mortality (Gupta et al., 2012). The main factors responsible for sudden death of fish are exposure duration and dosage of toxicant (pesticides) together with age, size, biology, habitat, diet and life cycle of species (Al-Rudainy and Kadhim, 2012; Piazza et al., 2015). Insecticidal toxicity including organochlorines, pyrethroids, organophosphates and carbamides has already been reported for many fish species (Naz et al., 2019a and 2019b; Desai and Bhilave, 2018; Naz et al., 2017; Vijayakumar et al., 2016; Ambreen and Javed, 2015).
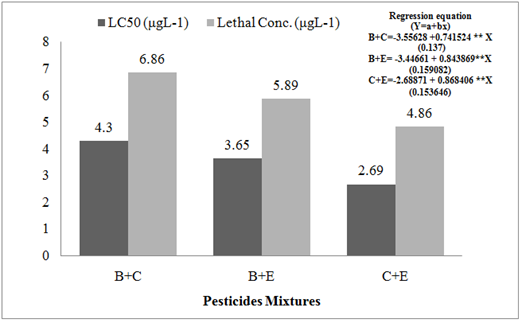
Fig. 1 The 96-h toxicity of insecticides mixtures (μgL-1±SD) for C. mrigala
Antioxidant enzymes: In acute and chronic experiment, activities of SOD, POx and GST significantly increased in organs of exposed fish. The SOD activity increased as muscle<heart<gills<kidney<brain<liver. The POx activity in exposed fish followed the trend: muscle<heart<kidney<gills<brain<liver. In acute exposure GST activity followed the trend: heart<muscle<gills<kidney<liver<brain while in chronic exposure it was increased as heart<muscle<kidney<gills<brain<liver. Similarly, Naz et al. (2019a) noticed the increased level of SOD, POx and GST in muscle, liver, heart, gills, brain and kidney of rohu exposed to END+CPF mixture. Naz et al. (2017) also observed the duration specific increase in liver SOD level of Indian carps under acute exposure of bifenthrin+endosulfan+chlorpyrifos mixtures. SOD activity tends to increase in different tissues of exposed Cirrhinus mrigala in order of liver>muscle>gill>brain at both the lethal and sub-lethal concentrations of methanol (Desai and Bhilave, 2018). Endosulfan+deltamethrin mixture increased level of GST in kidney, gills, muscle and liver of Channa striata (Abdullah et al., 2018). Endosulfan+chlorpyrifos mixture caused the raised in SOD, POx and GST activities in all organs of C. catla (Naz et al., 2021).
CAT activity showed some different results, it was increased in gills, liver and kidney of fish exposed to insecticides mixtures while it was decreased in brain, muscle and heart of fish. Chlorpyrifos or its metabolites when binds with CAT may cause reduction in its activity. It also affects the production and breakdown of the enzymes. Naz et al. (2019a) noticed significant raise in liver, gills and kidney catalase activity of END+CPF mixture exposed L. rohita while it was decreased in muscle, brain and heart. Cypermethrin at sub-lethal dose caused increase in SOD and CAT in bronchial, hepatic and renal tissues of fish (Vijayakumar et al., 2016). Treatment specific response showed that C+B mixture caused maximum increase in activities followed by E+B and B+C mixture. The enzymes activities were increased as the exposure duration increased while in chronic exposure activities increased during first 30 days afterthat these were decreased (Figure 2-3). Usman et al. (2020) also noticed the time specific decline in neural, cardiac, and muscle tissues of C. idella when exposed two different insecticides mixtures.
The two possible pathways of organophosphate pesticides (chlorpyrifos) to produce reactive oxygen species (ROS) are included i) the enzyme cytochrome P450s catalyzes the oxidation-reduction cycle in which bond between–P=S is converted into –P=O or it is already exist in organophosphate pesticides, has ability to acquire an electron and transfer O2 into superoxide anion which can easily produce other ROS like OH- (Kovacic, 2003) ii) these pesticides can inhibit the antioxidant enzymes which can diminish the ROS resulted in excessive amassment of ROS (Karaoz et al., 2002).One of the most toxic organochlorine pesticides is endosulfan which degrade into more stubborn metabolites as compared to original compound such as endosulfan ether, endosulfan α-hydroxy ether, endosulfan sulfate, endosulfan alcohol and endosulfan lactone (Awasthi et al., 2000). This insecticide can amass and induced toxic effects in the body including respiratory sickness, tissue impairment, changes in biochemical, physiological and molecular processes and ultimately cause the organismal mortality (Dar et al., 2015; Piazza et al., 2015). Bifenthrin is a pyrethroid insecticide which cause neurotoxicity in fish by binding transiently itself to sodium channel present in the nerve cells (Burr and Ray, 2004). This will happen after action potential and resulted in non-stop firing of axon however, bifenthrin did not disturbed the resting membrane potential. The disparity in pesticides toxicity may be due to variation in vulnerability and resistance with respect to pesticides absorption, bio-transformation and removal (Oruc and Usta, 2007).The toxicity of pesticides also associated with their formulation as well as some water quality variables including temperature, pH and dissolved oxygen (Tripathi and Yadav, 2015).
Genotoxicity: In both acute and chronic trials all the insecticides mixture induced significant formation of damaged nuclei (%),GDI, MN and NAs in RBCs of C. mrigala. Maximum induction was observed due to C+E mixture of insecticides. During acute trail, NAs followed the order DEN>BIN>MN>BN>DN>NN while in chronic order was as MN>DEN>BIN>BN>NN>DN. Duration dependent response showed that DNA damage MN and NAs were increased with exposure period while in chronic exposure damage increased during first 30 days after that these were decreased (Figure 4-5). Significant induction in micronuclei, blebbed, notched, lobed, binuceated and pear shape nuclei in RBCs of rohu exposed to triazophos was observed by Ghaffar et al. (2015). Labeo rohita showed increase in DNA damage in RBCs when exposed to both acute and chronic dose of cypermethrin (Gadhia et al., 2016). Ambreen et al. (2018) confirmed the damage to nuclei and GDI in RBCs of Ctenopharyngodon idella under sub-lethal dose of CPF+END. Naz et al. (2019b) also documented the END+CPF induced damage to nuclei and GDI in RBCs of C. catla in duration-specific manner. E+C mixture exposure caused increase in the formation of MN and NAs in RBCs of C. catla (Naz et al., 2021).
During metabolism, pesticides produce the ROS (Kovacic, 2003)includes hydroxyl radical, superoxide anion and hydrogen peroxide (Guney et al., 2007; Banudevi et al., 2006), which interact with nucleic acid by generating breaks in DNA strand and also damage the purine and pyrimidine bases (Mohanty and Mohanty, 2018; Lu et al., 2013). The two electrophilic groups, i.e. the alkyl- and phsophoryl-groups were formed by organophosphate pesticides metabolism. They can interact with DNA via mechanism of phosphorylation (Ali et al., 2009; Blasiak et al., 1999). According to Fenech and Ferguson (2001) living organisms have ability to synthesize and control specific enzyme that can repair these DNA damages.
Conclusion: This work suggests that insecticides mixtures have potential to induce oxidative stress and genotoxicity in C. mrigala. This study also concludes that antioxidant enzymes and molecular biomarkers are useful tools for assessing the toxicity of agrochemical to aquatic animal and might be helpful in environmental monitoring programs for estimation of aquatic pollution.
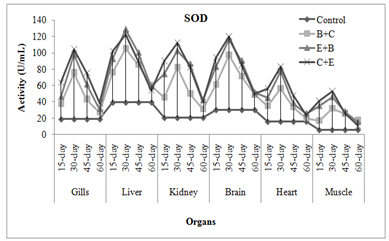 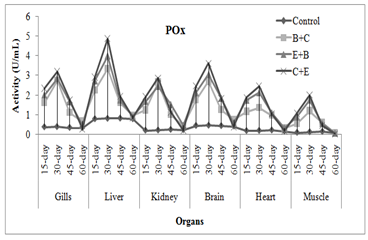 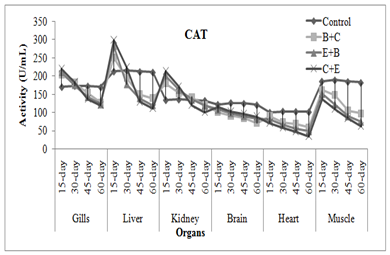 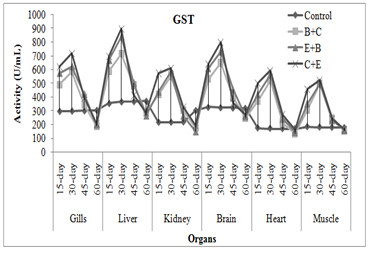
Fig. 3 Treatment and duration specific evaluation of enzymes activities in C. mrigala under chronic exposure
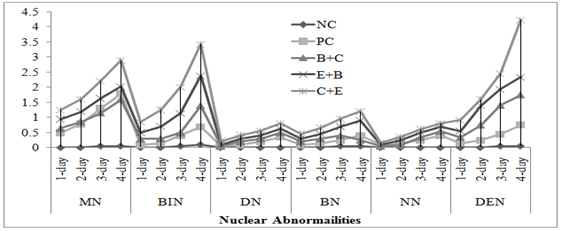 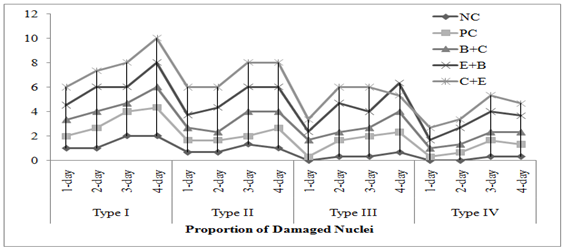
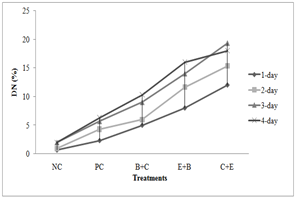 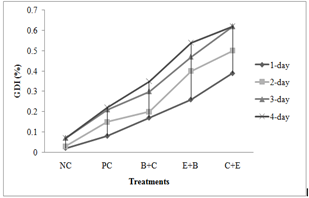
Fig. 4 Damaged to DNA in fish under acute insecticides mixtures exposure
REFERENCES
- Abdullah, S., A. Mateen, K. Abbas, H. Naz, W. Hassan and S. Anum (2018). Changes in glutathione S-transferase activity in fish Channa striata exposed to different aquatic pollutants (heavy metals and pesticides mixture). Pakistan J. Zool. Suppl. Ser. 13: 42-47.
- Anbumani, S. and M.N. Mohankumar (2011). Nuclear and cytoplasmic abnormalities in the fish Catla catla (Hamilton) exposed to chemicals and ionizing radiation. Res. J. Environ. Sci. 5: 867–877.
- Ali, D., N.S. Nagpure, S. Kumar, R. Kumar, B. Kushwaha and W. S. Lakra (2009). Assessment of genotoxic and mutagenic effects of chlorpyrifos in freshwater fish Channa punctatus (Bloch) using micronucleus assay and alkaline single-cell gel electrophoresis. Food Chem. Toxicol. 47: 650-656.
- Al-Rudainy, A.J. and M.H. Kadhim (2012). Hematological and neurotoxic effects of endosulfan pesticide on common carp Cyprinus carpio. The Iraqi J. Vet. Med. 36: 58-67.
- Ambreen, F. and M. Javed (2015). Assessment of acute toxicity of pesticides mixtures for Cyprinus carpio and Ctenopharyngodon idella. J. Zool. 47(1): 133-139.
- Ambreen, F., M. Javed, S. Abbas, S. Kousar, R. Ilyas and M. Batool (2018). DNA Damage in peripheral erythrocytes of Ctenopharyngodon idella during chronic exposure to pesticide mixture. Pakistan J. Zool. Suppl. Ser., No.13, pp.19-25.
- Anita, B., A.S. Yadav and N. Cheema (2016). Genotoxic effects of chlorpyrifos in freshwater fish Cirrhinus mrigala using micronucleus assay, Adv. Biol. pp.1-6. org/10.1155/2016/9276963
- Awasthi, N., R. Ahuja and A. Kumar (2000). Factors influencing the degradation of soil-applied endosulfan isomers. Soil Biol. Biochem. 32: 1697-1705.
- Banudevi, S., G. Krishnamoorthy, P. Venkatataman, C. Vignesh, M. M. Aruldhas and J. Arunakaran (2006). Role of a-tocopherol on antioxidant status in liver, lung and kidney of PCP exposed male albino rats. Food Chem. Toxicol. 44: 2040-2046.
- Barsiene, J., J. Lazutka, J. Syvokiene, V. Dedonyte, A. Rybakovas, A. Bjornstad and O.K. Andersen (2004). Analysis of micronuclei in blue mussels and fish from the Baltic and North seas. Environ. Toxicol. 19(4): 365-371. DOI: 1002/tox.20031.
- Belenguer, V., C.F. Martinez, A. Masia and Y. Pico (2014). Patterns of presence and concentration of pesticides in fish and waters of the Jucar River (Eastern Spain). J. Hazard. Mat. 265: 271-279.
- Blasiak, J., P. Jaloszynski, A. Trzeciak and K. Szyfter (1999). In vitro studies on the genotoxicity of the organophosphorus insecticide malathion and its two analogues. Mutat. Res. 445: 275-283.
- Burr, S.A. and D.E. Ray (2004). Structure-activity and interaction effects of 14 different pyrethroids on voltage-gated chloride ion channels. Toxicol. Sci. 77: 341-6.
- Chance, M. and A.C. Mehaly (1977). Assay of catalase and peroxidase. Methods Enzymol. 2: 764-817.org/10.1016/S0076-6879(55)02300-8.
- Dar, S.A., A.R. Yousuf, M.H. Balkhi, F.A. Ganai and F.A. Bhat (2015). Assessment of endosulfan induced genotoxicity and mutagenicity manifested by oxidative stress pathways in freshwater cyprinid fish crucian carp (Carassius carassius). Chemosphere, 120: 273-283. DOI: 10.1016/j.chemosphere.2014.07.031.
- Da-Silva S.T. and C.S. Fontanetti (2006). Micronucleus test and observation of nuclear alterations in erythrocytes of Nile tilapia exposed to waters affected by refinery Mutat. Res. 605(1-2): 87-93. DOI: 10.1016/j.mrgentox.2006.02.010.
- Desai, T.H. and M. Bhilave (2018). Toxicological studies of methanol on superoxide dismutase (SOD) activity of freshwater fish Cirrhinus mrigala. J. Fish. Aquat. Stud. 6(5): 20-22.
- Fenech, M. and L.R. Ferguson (2001). Vitamins/minerals and genomic stability in humans. Editorial. Mutat. Res. 475: 1-6.
- Fenech, M., W.P. Chang, M. Kirsch-Volders, N. Holland, S. Bonassi and E. Zeiger (2003). Human project: Detailed description of the scoring criteria for the cytokinesis block micronucleus assay using isolated human lymphocyte culture. Mutat. Res. 534(1-2): 65-75. DOI: 1016/s1383-5718(02)00249-8.
- Gadhia, M., R. Prajapati, and P. Gadhia (2016). Cypermethrin induced DNA damage in Labeo rohita assessed by comet assay. J. Environ. Sci.6: 1113- 1116.
- Ghaffar, A., R. Hussain, A. Khan and R.Z. Abbas (2015). Hemato-biochemical and genetic damage caused by triazophos in fresh water fish, Labeo rohita. J. Agric. Biol. 17(3): 637-642. DOI: 10.17957/IJAB/17.3.14.1016.
- Giannopolitis, C.N. and S.K. Ries (1977). Superoxide dismutase occurrence in higher plants. Plant Physiol. 59(2): 309-314. DOI: 1104/pp.59.2.309
- Grover, P., K. Danadevi, M. Mahboob, R. Rozati, B.S. Banu and M.F. Rahman (2003). Evaluation of genetic damage in workers employed in pesticide production utilizing the Comet assay. Mutagenesis, 18 (2): 201-205. https://doi.org/10.1093/mutage/18.2.201
- Guney, M., O. Baha, D. Hilmi, T. Gulnur, G.G. Seren, A. Irfan and M. Tamer (2007). Fallopian damage induced by organophosphate insecticide methyl-parathion, and protective effect of vitamins E and C on ultra-structural changes in rats. Ind. Health. 23: 429-438.
- Gupta, S.K., A.K. Pal, N.P. Sahu, N. Saharan, S.C. Mandal, M.S.C. Akhtar and A.K. Prusty (2012). Dietary microbial levan ameliorates stress and augments immunity in Cyprinus carpio fry (Linnaeus, 1758) exposed to sub-lethal toxicity of fipronil. Aquacult. Res. pp.1-14.
- Johnson, M., B. Luukinen, J. Gervais, K. Buhl and D. Stone (2010). Bifenthrin Technical Fact Sheet. National Pesticide Information Center, Oregon State University Extension Services. http://npic.orst.edu/factsheets/ biftech.pdf.
- Karaoz, E., F. Gultekin, M. Akdogan, M. Oncu and A. Gokcimen (2002). Protective role of melatonin and combination of vitamin C and vitamin E on lung toxicity induced by chlorpyrifos-ethyl in rats. Exp. Toxicol. Pathol. 54: 97-108.
- Kaur, M., and R. Jindal (2017). Oxidative stress response in liver, kidney and gills of Ctenopharyngodon Idellus (Cuvier & Valenciennes) exposed to chlorpyrifos. M.O.J. Biol. Med. 1(4):103-112. DOI: 15406/mojbm.2017.01.00021.
- Kovacic, P. (2003). Mechanism of organophosphates (nerve gases and pesticides) and antidotes: Electron transfer and oxidative stress. Curr. Med. Chem. 10: 2705-2709.
- Kumar, N., P.A.J. Prabhu, A.K. Pal, S. Remya, M. Aklakur, R.S. Rana, S. Gupta, R.P. Raman and S.B. Jadhao (2011). Anti-oxidative and immuno-hematological status of Tilapia (Oreochromis mossambicus) during acute toxicity test of endosulfan. Pestic. Biochem. Physiol. 99(1): 45-52. DOI: 10.1016/j.pestbp.2010.10.003.
- Lu ,Y., A. Zhang, C. Li, P. Zhang, X. Su, Y. Li, C. Mu and T. Li (2013). The link between selenium binding protein from Sinonovacula constricta and environmental pollutions exposure. Fish Shellfish Immunol. 35: 271-277.
- Majumder, R. and A. Kaviraj (2018). Acute and sublethal effects of organophosphate insecticide chlorpyrifos on freshwater fish Oreochromis niloticus. Drug Chem. Toxicol. 42(5): 487-495 DOI: 10.1080/01480545.2018.1425425.
- Mani, R., B. Meena, K. Valivittan and A. Suresh (2014). Glutathione-S-Transferase and catalase activity in different tissues of marine catfish Arius arius on exposure to cadmium. J. Pharm. Pharm. Sci. 6(1): 326-332.
- Mannervik, B. (1985). The isozymes of glutatione transferase. Advances in enzymology and related areas of molecular biology, 57: 357-417.
- Masia, A., J. Campo, A. Ortega, D. Barcelo and Y. Pico (2015). Pesticide monitoring in the basin of Llobregat River (Catalonia, Spain) and comparison with historical data. Sci. Total Environ. 503-504: 58-68.
- Modesto, K.A. and C.B.R. Martinez (2010). Effects of roundup transorbon fish: Hematology, antioxidant defenses and acetylcholinesterase activity. Chemosphere, 81(6): 781-787. DOI: 1016/j.chemosphere.2010.07.005
- Mohanty, G. and J. Mohanty (2018). Potentiality of short – term cultured cells of carps in DNA damage and repair studies. Int. J. Biotechnol. Biomed. Sci. 4(2): 67-73.
- Naz, H., S. Abdullah, K. Abbas and M.A. Zia (2017). Pesticides mixture toxicity; Effects on superoxide dismutase activity in Indian major carps. J. Agri. Sci. 54(3): 607-611. DOI: 10.21162/PAKJAS/17.5939.
- Naz, H., S. Abdullah, K. Abbas, M.R. Tariq, L. Shafique and G. Nazeer (2019b). Comet Assay: Quantification of damaged DNA in Catla catla exposed to endosulfan+chlorpyrifos. Punjab Uni. J. Zool. 34(1): 85-88. http://dx.doi.org/10.17582/journal.pujz/2019.34.1.85.88.
- Naz, H., S. Abdullah, K. Abbas, W. Hassan, M. Batool, S. Perveen, S. Maalik and S. Mushtaq (2019a). Toxic effect of insecticides mixtures on antioxidant enzymes in different organs of fish, Labeo rohita. J. Zool. 51(4): 1355-1361. DOI: http://dx.doi.org/10.17582/journal.pjz/2019.51.4.1355.1361.
- Naz, H., S. Abdullah, T. Ahmed, K. Abbas and M.U. Ijaz (2021). Regression analysis for predicting the duration dependent response of oxidative stress dynamics and nuclear anomalies in Catla catla exposed to chlorpyrifos and endosulfan. The J. Anim. Plant Sci. 31(4): 1167-1173.
- Oruc, E.O. and D. Usta (2007). Evaluation of oxidative stress responses and neurotoxicity potential of diazinon in different tissues of Cyprinus carpio. Environ. Toxicol. Pharmacol. 23: 48-55.
- Piazza, Y., M. Pandolfi, R. Da-Cuna, G. Genovese and F. Nostro (2015). Endosulfan affects GnRH cells in sexually differentiated juveniles of the perciform Cichlasoma dimerus. Ecotoxicol. Environ. Saf. 116: 150-159.
- Saravanan, M., K.P. Kumar, and M. Ramesh (2010). Haematological and biochemical responses of freshwater teleost fish Cyprinus carpio (Actinopterygii: Cypriniformes) during acute and sublethal exposure to lindane. Pestic. Biochem. Physiol. 100: 206–211. DOI: 10.1016/j.pestbp.2011.04.002
- Singh, N.P., M.T. McCoy, R.R. Tice and E.L. Schneider (1988). A simple technique for quantization of low levels of DNA damage in individual cells. Exp. Cell Res. 175(1): 184-191. DOI: 1016/0014-4827(88)90265-0.
- Steel, R.G.D., J.H. Torrie and D.A. Dickey (1996). Principles and procedures of statistics: A Biometrical Approach (3rd Ed.). McGraw Hill Book Co, Singapore, pp. 627.
- Sunanda, M., J.C.S. Rao, P. Neelima, K.G. Rao and G. Simhachalam (2016). Effects of chlorpyrifos (an organophosphate pesticide) in fish. Int. J. Pharm. Sci. Rev. Res. 39(1): 299-305.
- Tripathi, V.K. and R.K. Yadav (2015). Effect of pesticide (organophosphorus) on aquatic fish Labeo Rohita. J. Chem. Sci. 13: 625-640.
- Usman, T., S. Abdullah, H. Naz, K. Abbas, L. Shafique, and Q. Siddique (2020). Acute toxic effect of technical grade insecticides on behavior, catalase activity and total protein contents of fish, Ctenopharyngodon idella. Pakistan J. Zool. 52(5): 2023-2026. doi: https://dx.doi.org/10.17582/journal.pjz/20181103091108.
- Ventura Campos-de, B., D.F. Angelis-de and M.A. Marin-Morales (2008). Mutagenic and genotoxic effects of the atrazine herbicide in Oreochromis niloticus (Perciformes, Cichlidae) detected by the micronuclei test and the comet assay. Pestic. Biochem. Physiol. 90(1): 42-51. DOI: 10.1016/j.pestbp.2007.07.009.
- Vijayakumar, A., N. Thirnavukkarasu, K. Jayachandran and M. Susiladevi (2016). Attenuating properties of atropine against the cypermethrin toxicity in the oxidative stress in the fresh water fish Labeo rohita (Hamilton). J. Modn. Res. Revs. 4(1): 1088-1093.
- Yin, X.H., S.N. Li, L. Zhang, G.N. Zhu and H.S. Zhuang (2008). Evaluation of DNA damage in Chinese toad (Bufo bufo gargarizans) after in vivo exposure to sublethal concentrations of four herbicides using the comet assay. Ecotoxicology, 17(4): 280-286. doi: 10.1007/s10646-008-0195-z.
|





