CRYOPRESERVATION OF MENTHA PIPERITA L. GERMPLASM AND CONFIRMATION OF GENETIC STABILITY AFTER CRYO-STORAGE
S. Galatali1, M. Ceylan1, E. Ogur2, N. Adanacioglu2 and E. Kaya*1
1Molecular Biology and Genetics Department, Faculty of Science, Mugla Sitki Kocman University, 48000, Kotekli, Mugla, Turkey
2Biodiversity and Genetic Resources Department, Aegean Agricultural Research Institute, Menemen-Izmir, Turkey
*Corresponding author’s e-mail address: ergunkaya@mu.edu.tr (Ergun Kaya)
ABSTRACT
Peppermint is an important aromatic-medicinal plant species and it has valuable essential oil contents such as menthol, linalool and limonene. The main purpose of the current study was to optimize a protocol for cryopreservation of M. piperita local cultivars (G-74, Candarli and Gomec). The secondary aim was to investigate the genetic stability after cryopreservation using ISSR marker system. Three different single step freezing techniques were compared for long-term preservation of these local cultivars and the optimum regeneration percentages were acquired by using the droplet vitrification technique. The PVS2 treatments of this technique showed succesful long-term preservation of M. piperita cultivars with regeneration percentages of 72% to Candarli cultivar (60 min PVS2), 52% to Gomec cultivar (75 min PVS2) and 62.5% to G-74 cultivar (30 min PVS2) respectively. The ISSR PCR results showed that the genetic stability from cryopreserved M. piperita cultivars were high. While genetic stability percentage was ~99% for the Candarli cultivar, for the Gomec and G-74 cultivars the genetic fidelity was100%. The shoots that come from the cryopreserved shoot tips showed normal and health rooting, and all of them were also easily adapted to greenhouse conditions.
Key words: DMSO, encapsulation, ISSR-PCR, PVS2, vitrification
https://doi.org/10.36899/JAPS.2023.2.0625
Published first online December 18, 2022
INTRODUCTION
M. piperita L. belong to the Lamiaceae family which is a medicinal and aromatic plant species, traditionally used for treating nausea, headache, and vomiting. Its essential oil has very popular industrial usage for production of pharmaceuticals, cosmetics, personel hygiene products, food and beverage aroma (Rita and Animesh, 2011; Mehta et al., 2012). M. piperita is indicated by the fact that it’s flower male organs never develop in entirely sterile hybrid because of their short remaining and abnormally developing stamens however the pollens anomalously develop (Sunandakumari et al., 2004).
Short, mid and long-term storage of species such as endangered, endemic, and rare species which can be medicinal, aromatic, agricultural and ornemental plants, are progressed with plant biotechnology improvements using conservation and micropropagation techniques (Ruta et al., 2020). In vitro cell, embryo, tissue and organ culture protocols use allow for short-term conservation and supply fast, reliable and sufficient production and multiplication of plant materials under sterile and standard conditions. On the other hand, mid-term conservation is based on slow-growing conditions by the reducing of carbon sources and/or temperature changes that contribute to subculture periods varying from one or two months to several years depending on species (Torres-Silva et al., 2021). To achieve long-term storage of plant species have been used and are based on immersing plant structures in liquid nitrogen (-196°C). Cryopreservation is the more popular strategy and can provide cheap, effective and safe preservation (Ozudogru et al., 2010; Kaya et al., 2016; Souza et al., 2017; Pence et al., 2020; Kaya et al., 2020; Ozkaya et al., 2022).
Increasing concentration of cell cytoplasm via water removal using physical dehydration or chemical vitrification performs a crucial role during the cryostorage to prevent fatal ice nucleation injury and maintaining viability, germination or regeneration of cryopreserved plant in liquid nitrogen. Cryopreservation procedures are basically divided into two main categories: classical slow cooling or two step freezing and one step freezing (fast cooling) methods. Classical slow cooling technique is based on vitrification procedures using different kind of cryopretective solution but the main highlight of this technique is that the material used before rapid plungging into the liquid nitrogen is slowly cooled to -40 °C (determined as water super cooling temperature) by reducing 0.5-1 °C/min in the temperature (Reed and Uchendu, 2008; Kulus, 2019). One step freezing methods, including chemical vitrification or physical dehydration, are based on following vitrification and/or dehydartion treatments. Physical dehydration of the samples is achieved by using activated silica gel or laminar flow cabinet. On the other hand, the Chemical vitrification of the samples is carried out using cryoprotectant solution, then all treated samples direct and fast immersion in liquid nitrogen. There are also several application based on fast cooling such as single vitrification, dehydration/vitrification with encapsulation, droplet vitrification and cryo-plate techiques that were developed recently (Yamamoto et al., 2011; Montalbán et al, 2020).
Various kind of chemical components such as polyphenols having strong antioxidant activitiy against superoxide anion, peroxyl and hydroxyl radicals which are found in plants especially those of economical importance like aromatic/medicinal plants which are the source material of valuable secondary metabolites (Halliwell, 2008). Genetic stability after cryogenic treatments is essential to ensure identical genetic and phenotypic propagation to the mother plant. Genetic instability in aromatic and medicinal plants can cause reduction, differentiation and/or disappearance of secondary metabolites (Saha et al., 2016; Kaya and Souza, 2017) compromising their functionality (Saha et al., 2016; Kaya and Souza, 2017). Genetic stability can be determined using chromosome analyses, isoenzyme techniques and the DNA marker systems based on polymerase chain reactions such as inter simple sequence repeats (ISSR).
Since PCR based molecular marker systems doesn’t influence environmental conditions and produce reproducible, fast and trustworthy results, they have many advantageous when compare with the other techniques. A variety of PCR based molecular markers have been useful tool to determine genetic stability of in vitro grown, clonal propagated, short, mid and long-term conservated plant material. Among them, ISSR marker system is one of the most effective, cheap, simple and rapid method (Kaya, 2015; Faisal et al., 2021). ISSR marker system is normally used for determination of genetic variation and stability of some economically important woody plants (Ilczuk and Jacygrad, 2016), crops (Werner et al., 2015), medicinal plant (Thakur et al., 2016) species and performing phylogenetic analyses for some problematic species, cultivars and varieties (Kaya and Souza, 2017, Hatzilazarou et al., 2021).
It is hard to perform short, mid and/or long term storage of these species (having sterile seeds) using traditional techniques due to their decreased fertility. This work aimed to optimize an effective procedure via cryogenic procedures for long term conservation of three different M. piperita local cultivars and confirmed their genetic stabilitiy after liquid nitrogen treatment via ISSR-PCR technique.
MATERIALS AND METHODS
Plant material: The in vitro plants of three different local M. piperita L. cultivars “Candarli, Gomec and G-74” were obtained from the Republic of Turkey Ministry of Agriculture and Forestry, Aegean Agricultural Research Institute, Biodiversity and Genetic Resources Department, Menemen, Izmir, Turkey. The current work were carried out at Aegean Agricultural Research Institute, Biodiversity and Genetic Resources Department, in the plant biotechnology laboratory between April and December 2019.
Cold-hardening and sucrose preculture steps: For cold-hardening treatment, all in vitro shoots of M. piperita cultivars were directly transferred to +4 °C temperature and left in dark conditions for one week (Ozudogru et al., 2011 a,b). For sucrose preculture, ~ 0.3-0.7 mm shoot tips (Figure 1a-c) were excised from one week cold hardened M. piperita shoots and they were transferred to semi-solid MS preculture media (Murashige and Skoog, 1962) including 0.4 M sucrose (Mehta et al., 2012).
Cryopreservation via one-step freezing methods: In this work, three different fast cooling procedures based on chemical vitrification (direct-, encapsulation-, and droplet-vitrification) were compared for cryopreservation of three different M. piperita cultivars. For all methods, PVS2 [liquid MS medium supplemented with 0.4 M sucrose, 15% (w/v) ethylene glycol, 30% (w/v) glycerol, and 15% (w/v) dimethyl sulfoxide (DMSO)] (Sakai et al., 1990) were used as cryoprotectant solution. In addition, for direct PVS2-vitrification and encapsulation-vitrification techniques, all explants were soaked in loading solution (LS, MS liquid medium containing 2.0 M glycerol and 0.4 M sucrose) before the treatment with PVS2 (Matsumoto et al., 1994). To reduce the toxic influence of DMSO, samples were also treated with cold PVS2 (0°C) on ice and the treatment time was setted as 15, 30, 45, 60, 75 and 90 minutes for each method seperately.
For direct PVS2-vitrification method, cold-hardened and sucrose precultured samples were directly transferred to cryovial tubes containing 1 ml LS for 20 min and then, after removing LS solution, 1 ml cold PVS2 solution was added for different time periods (as described above) on ice. After removing PVS2 solution, 1 ml fresh cold cryoprotectant solution was added and then the samples were directly plunged into liquid nitrogen.
For encapsulation-vitrification method, after cold-hardening and sucrose preculture steps, the shoot tips were coated with calcium alginate [shoot tips washed with calcium-free MS liquid medium were transfered to 3% sodium alginate solution supplemented with calcium-free MS liquid medium and the each shoot tip coated with sodium alginate solution were dropped into 100 mM calcium cloride solution for polimerization. After this time they were rinsed with sterile distilled water to prevent viscosity and were kept at room temperature] (Ozudogru and Kaya, 2012). For cryopreservation of encapsulated shoot tips (Figure 2a), the beads were treated with LS and PVS2 solution as described in the direct-vitrification method and directly transferred to liquid nitrogen.
For droplet-vitrification method, each shoot tip was transferred to 3-5µl PVS2 drop on 3x12mm aluminium foil stripe (Figure 2b). After treatment of PVS2 drops, the aluminium foil strips with five shoot tip were placed in 1.5 ml cryovial tubes with liquid nitrogen. Than the cryovials were directly plunged into liquid nitrogen.
Twenty shoot tips of each samples were used for each treatment times. Ten of them for cryopreservation (LN+ group) and the others for PVS2 treatment control (PVS2 control group) and thirty shoot tips (for each step ten shoot tips) were used for control of shoot tip culture, cold-hardening and sucrose-preculture. To each protocol (direct PVS2-vitrification, encapsulation vitrification and droplet-vitrification) three experimental repetitions were performed.
Thawing and regrowth of cryopreserved shoot tips: Thawing processes of direct and encapsulation-vitrification techniques were the same. In this process, cryovials 24 storaged at least 24 hours in liquid nitrogen (Kaya et al., 2013) were directly plunged into the water-bath at 40 °C, after thawing, all explants were transferred to 1 M sucrose solution supplemented with standard liquid MS for 15 minutes, and then the samples were transferred to regeneration medium (MS medium including 4.44 µM benzyl adenine) at standard growth-room conditions [25 ± 2 °C, 16/8 hour photoperiod supplied via cool daylight fluorescent lamps (50 μmol−1m−2s−1)]. Thawing process for droplet vitrification was achieved by direct transferring of frozen shoot tips to 1 M sucrose solution supplemented with standard liquid MS for 15 minutes at room temperature. After thawing process, the samples were transferred to regeneration medium at standard growth-room condition described before.
Evaluation of Cryopreservation Data: Shoot tip regeneration percentages of all explants (control and LN+) were assessed after transferring to regeneration medium. Each shoot tip of both groups producing at least two standard leaves (morphologically normal) were evaluated as successful regeneration. Regeneration percentages of control and LN+ shoot tips were compared with multiple X2 test by SPSS statistics program (IBM SPSS Statistics 21.0) and statistical analyses were performed by One Way ANOVA, followed by LSD test at P ≤ 0.05. Percentage values also statistically analyzed by a non-parametric test, the post hoc multiple comparisons test (Marascuilo and McSweeney 1977).
Acclimatization of seedlings obtained from cryopreserved shoot tips: The rooted shoots obtained from cryostoraged shoot tips were adapted to the greenhouse conditions, the seedlings were transferred to 0.2 L plastic pots containing peat and perlite under 70% or more relative humidity. The humidity level were kept via transparent plastic lids, including nitrogen-rich peat, with a hole over the rooted shoots which was gradually decreased by daily drilling a hole on top of the transparent plastic pot. The transparent plastic pot were removed after ten days and the shoots were easily adapted to the greenhouse conditions.
Molecular analysis for determination of the genetic stability: Ten ISSR primers were used for determination of genetic stability and total genomic DNA’s of fresh leaf samples came from the mother plants (in vitro grown shoots), cold hardened and sucrose precultured plants, PVS2 treated (from each treatment time, control groups) and cryopreserved (after regeneration) plants were isolated using cetyltrimethylammonium bromide (CTAB) based manuel protocol (Doyle and Doyle, 1987; Ozudogru et al., 2011). PCRs were realized in a 25 µl reaction mix (1 unit Taq DNA polymerase, 0.4 mM dNTP, 1X PCR Buffer, and 2.5 mM MgCl2) using 40 ng/µl DNA template and ten ISSR primers [ISSR1 (AG)8T (GenBank accession number, UBC807); ISSR2 (AG)8G (UBC809); ISSR3 (GA)8T (UBC810); ISSR4 (GA)8C (UBC811); ISSR5 (CA)8A (UBC817); ISSR6 (TC)8C (UBC823); ISSR7 (AC)8C (UBC826); ISSR8 (AG)8CTT (UBC846); ISSR9 (CA)8AGT (UBC855); ISSR10 (GT)8CTA (UBC856)] (Martins-Lopes et al., 2009; Smykal et al., 2011). Thermocycler setting were 3 min at 95 ºC for primary denaturation, then it was continued by 35 cycles with 15 sec at 95 °C, 30 sec at 55 °C, 3 min at 72 °C and for final extension completed by at 72 ºC for 10 min. Gel electrophoresis was performed with 1.5% agarose gel to analyse the PCR products and the band profiles dyed with ethidium bromide, visualized under UV light, and monitored with image analysis system. DNA band profiles of ISSR PCRs were noted by their absence (0) or existence (1), and the ones at low intensity were noted just they were reproducible in both the PCR runs. Cluster analyses were achieved to make a dendrogram of the Unweighted Pair Group Method with Arithmetic mean (UPGMA) from the similarity data matrices using Jaccard’s coefficient (Marascuilo and McSweeney 1977).
RESULTS
Direct PVS2-vitrification: Regeneration percentages of control group shoot tips of three different M. piperita cultivars treated with PVS2 reduced gradually. The best regeneration percentages were obtained from PVS2 treated samples for 15 min (cv. “Candarli”, 10%) and 30 min (cv. “Gomec”, 37.5%; cv. G-74, 32.5%) and there was no regeneration, after liquid nitrogen, of cv. “Candarli” and cv. “G-74” shoot tips treated for 60-75-90 min with vitrification solution. In relation to cv. “Gomec”, no regeneration was obtained with samples treated with 60 and 90 min vitrification solution, but weak regeneration was registered after 75 min PVS2 treatment (Table 1-3).
Encapsulation vitrification: While very high regeneration was obtained after 6 weeks of incubation in all shoot tips of control groups (control, cold hardened, sucrose precultured, and PVS2 tereated), no regeneration was observed in "Candarli" and "Gomec" local cultivars after liquid nitrogen treatment. However, a low percentage of regeneration of 14% and 6%, respectively, was obtained in the liquid nitrogen group after 60 and 75 minutes of PVS2 treatment in the local cultivar "G-74" (Table 1-3).
Droplet vitrification: The regeneration percentages of control, cold hardened and sucrose precultured samples of all the three cultivars were 100%, however, the regeneration percentage of the samples dehydrated with PVS2 decreased gradually. After cryopreservation, the maximum regeneration percentages were obtained from PVS2 treated shoot tips of M. piperita cv. “G-74” (62.5%), cv. “Candarli” (72%), and cv. “Gomec” (52%) for 30, 60, and 75 min respectively (Table 1-3).
Comparison of three one-step freezing methods based on PVS2 vitrification: The best results of regeneration and viability were obtained via droplet vitrification technique for all of the three clones. This technique enabled the highest regeneration percentages after cryostorage compared with direct PVS2-vitrification and encapsulation vitrification techniques (Table 1-3). After droplet vitrification applications, the shoot tips of each local cultivars belonging to all liquid nitrogen groups formed shoots as seen in all control groups (Figure 3a-c). However, in both PVS2 vitrification and encapsulation vitrification applications, either no or very low percentage of regeneration was observed in the shoot tips of all local cultivars after liquid nitrogen.
Adaptation to greenhouse conditions: Seedlings obtained from cryopreserved shoot tips of cultivars M. piperita “G-74”, “Candarli” and “Gomec” showed good rooting and adequate acclimatization (Figure 4a-d).
Analysis of genetic stability via ISSR primers: While PCR reactions were performed on all of M. piperita cultivars using ten ISSR primers, reproducible band profiles were obtained only from six of them (ISSR1-6) for cv. Candarli, five (ISSR1, 2, 4, 5, and 6) for cv. Gomec and cv G-74. A total of 72 reproducible band profiles ranging from 340 bp to 2500 bp were observed for cv. Candarlı. However just one of them were polymorphic and the genetic stability percentage was calculated as ~ 99% (Figure 5a-c). A total of 59 and 62 repducible bands ranging from 290 bp to 2900 bp for cv. Gomec and 200 bp to 1750 bp for cv. G-74 was scored respectively however, none of these reproducible bands were polymorphic and their genetic stability percentage was 100%.
Table 1.Regeneration percentages of 6 weeks shoot tips of three Mentha x piperita L. cultivar “Candarli” cryopreserved via droplet vitrification, vitrification, encapsulation-vitrification techniques (SE, standart error).
|
Treatments
|
Regeneration percentages of Mentha × piperita L. cv. “Candarli” (% ± SE*)
|
|
Droplet vitrification
|
Vitrification
|
Encapsulation - vitrification
|
|
Control
|
100 ± 0a**
|
100 ± 0a
|
100 ± 0a
|
|
Cold Hardening
|
100 ± 0a
|
100 ± 0a
|
100 ± 0a
|
|
Sucrose Preculture
|
100 ± 0a
|
100 ± 0a
|
100 ± 0a
|
|
PVS2 (Control Groups) Exposure Times
|
15 min.
|
98 ± 4.10a
|
76 ± 16.33de
|
100 ± 0a
|
|
30 min.
|
92 ± 4.08b
|
74 ± 13.63ef
|
100 ± 0a
|
|
45 min.
|
80 ± 6.43cd
|
64 ± 12.06h
|
94 ± 4.91b
|
|
60 min.
|
76 ± 4.97de
|
62 ± 4.02h
|
92 ± 4.01b
|
|
75 min.
|
82 ± 7.58c
|
56 ± 17.51i
|
80 ± 10.98cd
|
|
90 min.
|
76 ± 6.4de
|
52 ± 9.84j
|
78 ± 19.44d
|
|
PVS2 (Liquid Nitrogen Groups, LN+) Exposure Times
|
15 min.
|
0
|
10 ± 12.7m
|
0 ± 0
|
|
30 min.
|
0
|
0
|
0
|
|
45 min.
|
28 ± 7.55l
|
0
|
0
|
|
60 min.
|
72 ± 4.03f
|
0
|
0
|
|
75 min.
|
68 ± 4.01g
|
0
|
0
|
|
90 min.
|
48 ± 7.53k
|
0
|
0
|
*Percentage values statistically analyzed by a non-parametric test, the post hoc multiple comparisons test (Marascuilo and McSweeney 1977)
**Statistical analysis performed by ANOVA, followed by LSD test at P ≤ 0.05
Table 2.Regeneration percentages of 6 weeks shoot tips of three Mentha x piperita L. cultivar “Gomec” cryopreserved via droplet vitrification, vitrification, encapsulation-vitrification techniques (SE, standart error).
|
Treatments
|
Regeneration percentages of Mentha × piperita L. cv “Gomec” (% ± SE*)
|
|
Droplet vitrification
|
Vitrification
|
Encapsulation - vitrification
|
|
Control
|
100 ± 0a
|
96,7 ± 4,76b
|
100 ± 0a
|
|
Cold Hardening
|
100 ± 0a
|
100 ± 0a
|
100 ± 0a
|
|
Sucrose Preculture
|
100 ± 0a
|
100 ± 0a
|
100 ± 0a
|
|
PVS2 (Control Groups) Exposure Times
|
15 min.
|
100 ± 0a
|
78 ± 16.08f
|
98 ± 4.01b
|
|
30 min.
|
100 ± 0a
|
68 ± 11.72g
|
100 ± 0a
|
|
45 min.
|
88 ± 4.07d
|
68 ± 13.33g
|
96 ± 4.91b
|
|
60 min.
|
92 ± 7.59c
|
60 ± 16.81h
|
90 ± 6.34cd
|
|
75 min.
|
92 ± 7.58c
|
50 ± 11.00j
|
90 ± 6.04cd
|
|
90 min.
|
78 ± 4.05f
|
56 ± 12.01i
|
82 ± 9.82e
|
|
PVS2 (Liquid Nitrogen Groups, LN+) Exposure Times
|
15 min.
|
0
|
0
|
0
|
|
30 min.
|
7.5 ± 4.38o
|
37.5 ± 4.31l
|
0
|
|
45 min.
|
0
|
10 ± 6.05n
|
0
|
|
60 min.
|
42 ± 7.41k
|
0
|
0
|
|
75 min.
|
52 ± 7.54j
|
4 ± 4.92p
|
0
|
|
90 min.
|
34 ± 10.27m
|
0
|
0
|
*Percentage values statistically analyzed by a non-parametric test, the post hoc multiple comparisons test (Marascuilo and McSweeney 1977)
**Statistical analysis performed by ANOVA, followed by LSD test at P ≤ 0.05
Table 3.Regeneration percentages of 6 weeks shoot tips of three Mentha x piperita L. cultivar “G-74” cryopreserved via droplet vitrification, vitrification, encapsulation-vitrification techniques (SE, standart error).
|
Treatments
|
Regeneration percentages of Mentha × piperita L. cv “G-74” (% ± SE*)
|
|
Droplet vitrification
|
Vitrification
|
Encapsulation - vitrification
|
|
Control
|
100 ± 0a
|
100 ± 0a
|
100 ± 0a
|
|
Cold Hardening
|
100 ± 0a
|
100 ± 0a
|
100 ± 0a
|
|
Sucrose Preculture
|
100 ± 0a
|
100 ± 0a
|
100 ± 0a
|
|
PVS2 (Control Groups) Exposure Times
|
15 min.
|
88 ± 10.05f
|
72 ± 0.01hi
|
100 ± 0a
|
|
30 min.
|
90 ± 6.45ef
|
74 ± 0.03gh
|
98 ± 0.01ab
|
|
45 min.
|
74 ± 4.98g
|
70 ± 4.24ij
|
98 ± 4.07ab
|
|
60 min.
|
64 ± 12.18lm
|
66 ± 7.59kl
|
92 ± 7.51de
|
|
75 min.
|
68 ± 4.05jk
|
64 ± 7.58lm
|
94 ± 7.08cd
|
|
90 min.
|
68 ± 9.90jk
|
62 ± 4.05m
|
96 ± 4.13bc
|
|
PVS2 (Liquid Nitrogen Groups, LN+) Exposure Times
|
15 min.
|
32 ± 7.56p
|
0
|
0
|
|
30 min.
|
62.5 ± 4.38m
|
32.5 ± 4.03p
|
0
|
|
45 min.
|
56 ± 8.07n
|
14 ± 10.24r
|
0
|
|
60 min.
|
34 ± 4.94p
|
0
|
14 ± 7.54r
|
|
75 min.
|
46 ± 4.71o
|
0
|
6 ± 7.11s
|
|
90 min.
|
0
|
0
|
0
|
*Percentage values statistically analyzed by a non-parametric test, the post hoc multiple comparisons test (Marascuilo and McSweeney 1977)
**Statistical analysis performed by ANOVA, followed by LSD test at P ≤ 0.05

Figure 1.In vitro grown shoots of M. piperita L. cv. “Candarli” (a); excised shoot tips of cv. “Gomec” (b); cv. “G-74” (c).

Figure 2.Encapsulated shoot tips of M. piperita L. cv. “Candarli” in calcium-alginate beads (a); cv. “Gomec” shoot tips transferred to 3µl PVS2 drop on 3x12mm aluminium foil stripe (b).
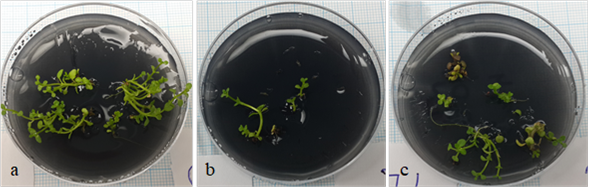
Figure 3. Shoots derived from cryopreserved shoot tips of M. piperita cv. “Candarli” (a), “Gomec” (b), “G-74” (c).

Figure 4.Shoots derived from cryopreserved shoot tips of M. piperita cv. “Candarli” (a), “Gomec” (b), “G-74” (c) developed well-formed roots, and cv. “Candarli” acclimated to greenhouse conditions (d).
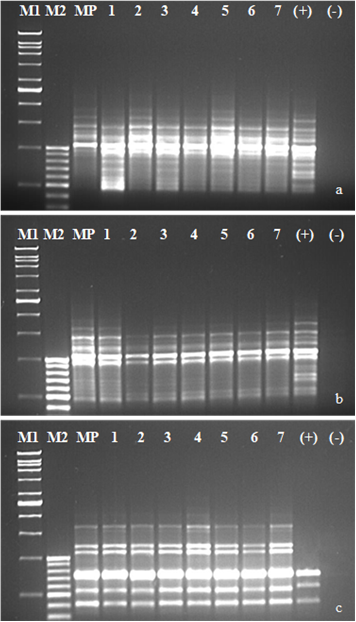
Figure 5.Band profiles of PCR products belonging to M. piperita cv. “Candarli” used ISSR4 (a), ISSR5 (b) and, ISSR6 (c) primers were visualized under UV light on 1.5% agarose gel stained etidium bromide. M1, 1kb ladder (GeneRulerTM); M2, 100bp ladder (GeneRulerTM); MP, Mother Plant (untreated shoots from cv. “Candarli” in vitro cultures); 1, cold hardened shoots; 2, sucrose pre-cultured shoots; 3-6, 15-30-45-60 min PVS2-treated shoots respectively (PVS2 controls); 7, shoots derived from cryopreserved shoot tips via 60 min PVS2; (+), positive control (PCR reaction was performed different Mentha spp. DNA); (-), negative control (PCR reaction was performed DNA-free). All samples derived from in vitro grown shoots of each line came from the same clone.
DISCUSSION
The Mentha genus consists of more than 25 species and numerous varieties and cultivars that are used as medicinal plant (Brickel et al., 2009), but just a few genotypes of this germplasm have been successfully conserved for long-term storaged via cryopreservation (Senula et al., 2007; Uchendu and Reed, 2008). In our study, three different fast cooling techniques based on chemical vitrification of cells via PVS2, encapsulation-, direct-, and droplet-vitrification, were compared for cryopreservation of M. piperita local cultivars, “Candarli”, “Gomec” and, “G-74”. Uchendu and Reed (2008) cryopreserved four Mentha spp. using controlled rate cooling, encapsulation dehydration, PVS2 vitrification techniques and they obtained the best regeneration from these four Mentha spp. using controlled rate cooling. This technique was significantly better than encapsulation dehydration and vitrification techniques. In another study, Hirai and Sakai (1999) cryopreserved encapsulated shoot-tip explants of M. spicata via vitrification technique and they obtained nearly 90% of shoot formation rate. Senula et al. (2007) cryopreserved shoot tip explants of Mentha x piperita, M. x villosa, and M. spicata accessions via droplet vitrification technique in a previous study and the regeneration percentage obtained from cryopreserved shoot tips ranged between 50% and 89%. The results obtained from these studies are in line with the results obtained in our study, except for the numerical differences seen in the percentage values. These numerical differences may have been caused by usage of different cold hardening and preculture conditions and/or different PVS2 exposure times. For example, Senula et al. (2007) precultured M. piperita shoot tip explants on liquid MS basal media containing 3% sucrose for 20-24 hours, then, the explants were placed to LS (described before) for 2 h, followed by chemical dehydration in PVS2. On the other hand, Hirai and Sakai (1999) cold hardened M. piperita meristems at 4 ºC for 3 weeks, the explants were then encapsulated in calcium-alginate capsules and they were treated with loading solution A (described above) and after all the explants were dehydrated by using PVS2 for 3 hours at 0 ºC before being immerged into liquid nitrogen.
Since cryopreservation methods need to be optimized for each plant species exclusively, only limited plant species could be long-term storaged for their germplasm conservation (Kaviani, 2011). Although the number of plant species cryopreserved via vitirification based techniques has continuously risen for each species, these techniques needs optimization and standardization for effective rejeneration after cryostorage (Reed, 2001; Panis et al., 2002). For this reason, in our study, the three local cultivars of M. piperita were tested using three different fast cooling techniques based on PVS2 vitrification, encapsulation virtification and droplet vitrification. Regeneration percentages of the cryo-storaged explants by using droplet vitrification technique were significantly better than encapsulation- and single-vitrification techniques for these local cultivars. Many plant species such as Thymus spp. (Ozudogru and Kaya, 2012), Ananas comosus (L.) Mer. (Souza et al., 2015), Saccharum spp. (Kaya and Souza, 2017), Panax ginseng Meyer (Le et al., 2019), Ribes nigrum L. (Rantala et al., 2021) were successfully cryopreserved via droplet vitrification technique. This technique has also many advantages such as reaching very high cooling/warming rates due to the use of small volume of vitrification solution.
Cold hardening step supports low temperature and desiccation tolerance due to the expression of cold stress genes (Takagi, 2000). Niino et al. (1992) indicated that before vitrification treatment of shoot tips, cold hardening of explant for 3 weeks at 5 °C increased regeration of Malus domestica Borkh. cv. Fuji after cryopreservation. In one another study, Chen et al. (2011) used a temperature of 4 °C for 1 week a temperature of cold hardened in vitro plantlets of three Lilium spp. cultivars followed by vitrification treatment and, they were able to successfully cryopreserve of these cultivars by droplet vitrification technique. Same results were also obtained in the current study (Table 1-3). Sucrose preculture of samples prior to treatment with cryoprotectant solution such as PVS2, has been reported to be useful for effective regeneration after cryostorage in some species, e.g. Eucalyptus spp. and Saccharum spp. (Kaya and Souza, 2017; Kaya et al., 2013). Sugar accumulation in cells can cause slow removal of water content which could induce fatal ice-nucleation and improve cell membrane stability during dehydration and cryostorage (Salama et al., 2018; Lata et al., 2019). In our study, the most effective sucrose preculture condition was achieved using MS semi-solid medium containing 0.4 M sucrose, the explants were placed in this culture medium for 16-18 hours for effective sucrose acumulation. After cold hardening and sucrose preculture steps, explants of M. piperita were treated with PVS2 as cryoprotective/dehydration solution at different exposure times, long time exposure with PVS2 was toxic fo both of the control and the LN+ (Table 1-3). The ideal PVS2 treatment time can vary for different plant species, subspecies and, cultivars (Pennycooke and Towill, 2000; Kaya et al., 2013). In this study, droplet vitrification was the most effective cryopreservation technique for local M. piperita cultivars Candarli, Gomec and G-74 and the best PVS2 treatment time periods were 60, 75 and 30 min respectively (Table 1-3).
The successful long-term storage of plant germplasm is based on high regeneration rate and genetic stability. Theoretically, cryo-storaged biological materials can be storaged without any metabolic and molecular variations for long time (Helliot et al., 2002), however, regenerated cultures derived from cryopreserved plants face a series of stressess such as those in the dehydration process and the exposure to ultra-low temperature which could induce genetic variations or modifications. Therefore, confirmation of genetic stability of cryostoraged plant material is important. Helliot et al. (2002) investigated genetic stability in cryopreserved Prunus using molecular markers and they found no genetic variations among the cryopreserved apices in comparison to the control. Kaya and Souza (2017) used ISSR primer to determine genetic stability for cryopreserved Saccharum spp. varieties, and they found no crucial variation for all the cryopreserved varieties. Our study is the first time where ISSR primers were used to detect genetic stability of cryopreserved M. piperita cultivars and their genetic stability percentages were were determined to be over 99% for all cultivars. The band profiles of PCR products showed that these small differences for only cv. “Candarli” could be caused by dehydration of plant material with PVS2 containing DMSO reported in several studies as toxic, for it induces the benzene metabolism and increases the toxical effects of other aromatic hydrocarbons (Kocsis et al., 1968; Kaya and Souza, 2017).
Conclusion: This study compared three different fast cooling techniques based on chemical vitrification for cryopreservation of three local cultivar of M. piperita, direct PVS2- vitrification, encapsulation vitrification and droplet vitrification, it’s primary aim was determine the most effective technique among the three techniques. The droplet vitrification technique was found to be the most effective than the other two techniques. The other goal of the study was to detect genetic uniformity of cryostoraged cultivars for the most effective technique (droplet vitrification). Genetic uniformity of shoots derived from cryo-storaged expalnts of three local M. piperita cultivars was very high. Alhough the chosen procedure for effective cryostorage need to be optimized for each species, subspecies and also cultivars exclusively, the droplet vitrification technique based on usage of small volume cryoprotectant solution, hence, it can be effective for wide range of plant taxa cryopreservation at the same time supporting genetic stability.
Competing Interests: The authors declare that they have no competing interests.
Funding: Authors declare that they didn’t receive any type of funds from any organization.
REFERENCES
- Brickel, C.D., C. Alexander, J.C. David, W.L.A. Hetterscheid, A.C. Lesslie, V. Malecot, J. Xiaobai and J.J. Cubez (2009). International code of nomenclature for cultivated plants. Scr. Hortic. 10: 1–184.
- Chen, X.L., J.H. Li, X. Xin, Z.E. Zhang, P.P. Xin and X.X. Lu (2011). Cryopreservation of in vitro-grown apical meristems of Lilium by droplet-vitrification. S. Afr. J. Bot. 77: 397-403.
- Doyle, J.J. and J.L.A. Doyle (1987). Rapid DNA isolation procedure for small quantities of fresh leaf tissue. Focus. 12: 13-15.
- Faisal, M., E.M. Abdel-Salam, A.A. Alatar and A.A. Qahtan (2021). Induction of somatic embryogenesis in Brassica juncea and analysis of regenerants using ISSR-PCR and flow cytometer. Saudi J. Biol. Sci. 28(1): 1147-1153.
- Halliwell, B. (2008). Are polyphenols antioxidants or pro-oxidants? What do we learn from cell culture and in vivo studies? Arch. Biochem. Biophys. 476: 107–112.
- Hatzilazarou, S., S. Kostas, T. Nendou and A. Economou (2021). Conservation, regeneration and genetic stability of regenerants from alginate-encapsulated shoot explants of Gardenia jasminoides Polymers. 13(10): 1666.
- Helliot, B., D. Madur, E. Dirlewanger and M.T. De Boucaud (2002). Evaluation of genetic stability in cryopreserved Prunus. In Vitro Cell. Dev. Biol. 38(5): 493-500.
- Hirai, D. and A. Sakai (1999). Cryopreservation of in vitro-grown axillary shoot-tip meristems of mint (Mentha spicata) by encapsulation vitrification. Plant Cell Rep. 19: 150–155.
- Ilczuk, A. and E. Jacygrad (2016). In vitro propagation and assessment of genetic stability of acclimated plantlets of Cornus alba using RAPD and ISSR markers. In Vitro Cell. Dev. Biol. 52(4): 379–390.
- Kaviani, B. (2011). Conservation of plant genetic resources by cryopreservation. Aust. J. Crop Sci. 5(6): 778-800.
- Kaya, E. (2015). ISSR analysis for determination of genetic diversity and relationship in some turkish olive (Olea europaea L) cultivars. Not. Bot. Horti Agrobot. Cluj-Napoca. 43(1): 96-99.
- Kaya, E., A. Alves, L. Rodrigues, M. Jenderek, M. Hernandez-Ellis, A. Ozudogru and D. Ellis (2013). Cryopreservation of eucalyptus genetic resources. Cryo-Lett. 34(6): 608-618.
- Kaya, E. and F.V.D. Souza (2017). Comparison of two PVS2-based procedures for cryopreservation of commercial sugarcane (Saccharum) germplasm and confirmation of genetic stability after cryopreservation using ISSR markers. In Vitro Cell. Dev. Biol. 53: 410-417.
- Kaya, E., F.V.D. Souza, J.A. dos Santos-Serejo and S. Galatali (2020). Influence of dehydration on cryopreservation of Musa spp. germplasm. Acta Bot. Croat. 79(2): 99-104.
- Kaya, E., F.V.D. Souza, E. Yilmaz-Gokdogan, M. Ceylan and M. Jenderek (2016). Cryopreservation of citrus seed via dehydration followed by immersion in liquid nitrogen. Turk. J. Biol. 41: 242-248.
- Kocsis, J.J., S. Harkaway, M.C. Santoyo and R. Snyder (1968). Dimethyl sulfoxide: interactions with aromatic hydrocarbons. Science. 160: 427-428.
- Kulus, D. (2019). Managing plant genetic resources using low and ultra‑low temperature storage: a case study of tomato. Biodivers. Conserv. 28: 1003–1027.
- Lata, H., E. Uchendu, S. Chandra, C.G. Majumdar, I.A. Khan and M.A. ElSohly (2019). Cryopreservation of axillary buds of Cannabis sativa by v-cryoplate droplet-vitrification: The critical role of sucrose preculture. Cryo-Lett. 40(5): 291-298.
- Marascuilo, L.A. and M. McSweeney (1977). Post-hoc multiple comparisons in sample preparations for test of homogeneity. In: McSweeney, M. and L.A. Marascuilo (eds.), Non-Parametric and Distribution-Free Methods for the Social Sciences, 141–147. Brooks/Cole Publications, Pacific Grove, CA, USA.
- Martins-Lopes, P., S. Gomes, J. Lima-Brito, J. Lopes and H. Guedes-Pinto (2009). Assessment of clonal genetic variability in Olea europaea ‘Cobrançosa’ by molecular markers. Sci. Hortic. 123: 82-89.
- Matsumoto, T., A. Sakai and K. Yamada (1994). Cryopreservation of in vitro grown apical meristems of wasabi (Wasabia japonica) by vitrification and subsequent high plant regeneration. Plant Cell Rep. 13: 442–446.
- Mehta, J., R. Naruka, M. Sain, A. Dwivedi, D. Sharma and J. Mirza (2012). An efficient protocol for clonal micropropagation of Mentha piperita (Pipperment). Asian J. Plant Sci. 2(4): 518-523.
- Montalbán, I.A., A. Castander-Olarieta, C. Pereira, J. Canhoto and P. Moncaleán (2020). Use of Biotechnology in Forestry Breeding Programs for Natural Resources and Biodiversity Conservation: Creating Super Trees for the Future. In: Chong P., D. Newman and D. Steinmacher (eds) Agricultural, Forestry and Bioindustry Biotechnology and Biodiscovery. Springer, Cham.
- Murashige, T. and F. Skoog (1962). A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15: 473–497.
- Niino, T., A. Sakai, H. Yakuwa and K. Nojiri (1992). Cryopreservation of in vitro-grown shoot tips of apple and pear by vitrification. Plant Cell, Tissue Organ Cult. 28: 261–266.
- Ozkaya, D.E., F.V.D. Souza and E. Kaya (2022). Evaluation of critical points for effective cryopreservation of four different Citrus germplasm. Horticulturae. 8(11): 995.
- Ozudogru, E.A., M. Capuana, E. Kaya, B. Panis and M. Lambardi (2010). Cryopreservation of Fraxinus excelsior embryogenic callus by one-step freezing and slow cooling techniques. Cryo-Lett. 31(1): 63-75.
- Ozudogru, E.A. and E. Kaya (2012). Cryopreservation of Thymus cariensis and vulgaris shoot tips: comparison of there vitrification-based methods. Cryo-Lett. 33(5): 363-375.
- Ozudogru, E.A., E. Kaya and E. Kirdok (2011a). Development of protocols for short-, medium- and long-term conservation of thyme. Acta Hortic. 918: 43-50.
- Ozudogru, E.A., E. Kaya, E. Kirdok and S. Issever-Ozturk (2011b). In vitro propagation from young and mature explants of thyme (Thymus vulgaris and longicaulis) resulting in genetically stable shoots. In Vitro Cell. Dev. Biol. 47: 309-320.
- Ozudogru, E.A., E. Kirdok, E. Kaya, M. Capuana, A. De Carlo and F. Engelmann (2011). Medium-term conservation of redwood [Sequoia sempervirens (D. Don.) Endl.] in vitro shoot cultures and encapsulated buds. Sci. Hortic. 127: 431–435.
- Panis, B., H. Strosse, S. Van den Henda and R. Swennen (2002). Sucrose preculture tosimplify cryopreservation of banana meristem cultures. Cryo-Lett. 23: 375-384.
- Pence, V.C., D. Ballesteros, C. Walters, B.M. Reed, M. Philpott, K.W. Dixon, H.W. Pritchard, T.M. Culley and A.C. Vanhovea (2020). Cryobiotechnologies: Tools for expanding long-term ex situ conservation to all plant species. Biol. Conserv. 250: 1087362.
- Pennycooke, J.C. and L.E. Towill (2000). Cryopreservation of shoot tips from in vitro plants of sweet potato [Ipomoea batatas (L.) Lam.] by vitrification. Plant Cell Rep. 19: 733–737.
- Rantala, S., J. Kaseva, A. Nukari, J. Laamanen, S. Tuohimetsä, S. Karhu, M. Veteläinen and H. Häggman (2021). Droplet vitrification technique for cryopreservation of a large diversity of blackcurrant (Ribes nigrum) cultivars. Plant Cell, Tissue Organ Cult. 144: 79-90.
- Reed, B.M. (2001). Implementing cryogenic storage of clonally propagated plants. Cryo-Lett. 22: 97-104.
- Reed, B.M. and E. Uchendu (2008). Controlled Rate Cooling. In: Reed, B.M. (eds.), Plant Cryopreservation: A Practical Guide, 77-92. Springer-Verlag, Berlin, Germany.
- Rita, P. and D.K. Animesh (2011). An updated overview on peppermint (Mentha piperita ). Int. J. Res. Pharm. Sci. 2(8): 1-10.
- Ruta, C., M. Lambardi and E.A. Ozudogru (2020). Biobanking of vegetable genetic resources by in vitro conservation and cryopreservation. Biodivers. Conserv. 29: 3495-3532.
- Saha, S., S. Adhikari, T. Dey and P. Ghosh (2016). RAPD and ISSR based evaluation of genetic stability of micropropagated plantlets of Morus alba variety S-1. Meta Gene. 7: 7-15.
- Sakai, A., S. Kobayashi and I. Oiyama (1990). Cryopreservation of nucellar cells of navel orange (Citrus sinensis var. Brasiliensis Tanaka) by vitrification. Plant Cell Rep. 9: 30-33.
- Senula, A., E.R.J. Keller, T. Sanduijav and T. Yohannes (2007). Cryopreservation of cold-acclimated mint (Mentha) shoot tips using a simple vitrification protocol. Cryo-Lett. 28(1): 1-12.
- Smykal, P., N. Bacova-Kerteszova, R. Kalendar, J. Corander, A.H. Schulman and M. Pavelek (2011). Genetic diversity of cultivated flax (Linum usitatissimum) germplasm assessed by retrotransposon-based markers. Theor. Appl. Genet. 122: 1385-1397.
- Souza, D., E. Kaya, J. Vieira, E. Hilo de Souza, O. Amorim, D. Skogerboe, T. Matsumoto, C. Alves, S. Ledo and M. Jenderek (2015). Droplet-vitrification and morphohistological studies of cryopreserved shoot tips of cultivated and wild pineapple genotypes. Plant Cell, Tissue Organ Cult. 124 (2): 351-360.
- Souza, F.V.D., E. Kaya, L. de J. Vieira, A. da S. Souza, M.de J.da S. Carvalhoe, E.B. Santos, A.A.C. Alvesa and D. Ellis (2017). Cryopreservation of hamilin sweet orange [(Citrus sinensis (L.) Osbeck)] embryogenic calli using a modified aluminum cryo-plate technique. Sci. Hortic. 224: 302–305.
- Sunandakumari, C., K.P. Martin, M. Chithra, S. Sini and P.V. Madhusoodanan (2004). Rapid axillary bud proliferation and ex vitro rooting of herbal spice, Mentha piperita Indian J. Biotechnol. 3: 108-112.
- Takagi, H. (2000). Recent developments in cryopreservation of shoot apices of tropical species. In: Engelmann, F., H. Takagi (eds.), Cryopreservation of Tropical Plant Germplasm, Current Research Progress and Application, 178-193. Rome, Italy.
- Thakur, J., M.D. Dwivedi, P. Sourabh, P.L. Uniyal and A.K. Pandey (2016). Genetic homogeneity revealed using SCoT, ISSR and RAPD markers in micropropagated Pittosporum eriocarpum Royle-an endemic and endangered medicinal plant. PLoS One. 11(7): e0159050.
- Torres-Silva, G., A.S. Schnadelbach, H.B. Bezerra, A. Lima-Brito and S.V. Resende (2021). In vitro conservation and genetic diversity of threatened species of Melocactus (Cactaceae). Biodivers. Conserv. 30: 1067–1080.
- Uchendu, E.E. and B. Reed (2008). A comparative study of three cryopreservation protocols for effective storage of in vitro-grown mint (Mentha). Cryo-Lett. 29(3): 181-188.
- Werner, E.T., T.C.B. Soares, A.B.P.L Gontijo, J.D. Souza Neto and J.A.T. do Amaral (2015). Genetic stability of micropropagated plants of Crambe abyssinica Hochst using ISSR markers. Genet. Mol. Res. 14(4): 16450-16460.
- Yamamoto, S., T. Rafique, W.S. Priyantha, K. Fukui, T. Matsumoto and Niino (2011). Development of a cryopreservation procedure using aluminium cryo-plates. Cryo-Lett. 32: 256-265.
|





