PHYTOCHEMICAL PROFILING OF BIOACTIVE METABOLITES IN METHANOLIC EXTRACT OF TWO WILD SOLANUM SPECIES AND EVALUATION OF THEIR ANTIOXIDANT ACTIVITY.
N. M. George1, E. Abdelhaliem1 and M. Abdel-Haleem1*
1“Department of Botany and Microbiology, Faculty of Science, Zagazig University, 44519, Zagazig, Egypt
*Corresponding author’s email: mohamedabdelhaleem20@yahoo.com; mahafez@zu.edu.eg
.
ABSTRACT
Medicinal plants abound in conventional medicines with antioxidant potentials that may be of value for the treatment of diseases, but with little or no scientific basis for their use. So, the present study aims to conduct a detailed investigation of phytochemicals present in two wild Solanum plants and also their potential use as an effective antioxidant agent. The objective of this present work was to identify the phytochemical compounds by using Gas chromatography–mass spectrometry technique and its biological activity of Solanum plant leaf extracts of methanol solvent systems were predicted using PASS online software. The methanolic leaf extract of Solanum nigrum and Solanum lycopersicum was tested to determine its chemical composition. The result of the preliminary phytochemical investigation in methanolic extract of Solanum revealed the presence of several bioactive secondary metabolites. In order to better understand the common uses of this plant, the antioxidant activity of the extracts was also measured using three separate assays.: “radical 2,2- diphenyl-1-picrylhydrazyl (DPPH) scavenging activity, Ferric-reducing antioxidant power (FRAP) and 2,2-azino-bis(3-etilbenzotiazolin)-6-sulfonic acid (ABTS) scavenging assay”. In different antioxidant assays, the methanolic extract showed different IC50 values slightly higher than the standard to confirm the importance of medicinal plant's bioactive metabolites.
Keywords: GC-MS analysis, PASS, S. nigrum, S. lycopersicum, phytochemical screening, antioxidant activity
https://doi.org/10.36899/JAPS.2022.6.0579
Published first online June 11, 2022
INTRODUCTION
Almost 80 % of the world's populations use medicinal plants or their metabolites as their primary health care or for treatment Mahomoodally (2013). They already known as a multi-purpose medicinal plant (MMP) and safe for consumption contributes to the availability of a spectrum of medicinal products originating from certain herbs and plants Popoola et al. (2020).
Solanum spp has been traditionally used as an emollient, antiseptic, analgesic, antidysentery, tonic, antispasmodic, laxative, antinarcotic, soporific, diuretic, antiulcer, anti-cancer and for disorders of neuro-vegetative system “Saijo et al. 1982; Akhtar and Muhammad (1989); Edmonds and Chweya (1997); Manoko et al. (2007); El-Shaboury et al. (2017)”.
Family Solanaceae exhibits A broad variety of secondary metabolites, making them very valuable for agricultural, economic and pharmaceutical purposes “Zadra et al. (2012); Mohasana et al. (2020)”. The per-centages of active constituent in the secondary substances determine the potential of its uses in the preparation of drugs. “Gas Chromatography Mass Spectroscopy (GC-MS)” is useful technique for separation and identification of complicated mixture of metabolites “Witte et al. (1987); Hussein et al. (2017).” Recently, gas chromatography mass spectroscopy (GC–MS) has been applied to determine the structures of different plant metabolites and other biological samples with great success “Anna et al. (2017)”. In the Egyptian flora, wild medicinal plants are exposed to serious risk due to heavy human impacts such as overgrazing, uncontrolled tourism and unregulated collection, mining and quarrying. Inclusion of wild plant conservation strategies into development plans of the Egyptian economy would minimize human impacts on these plants and ensure their sustainable use in both herbal medicine and pharmaceutical compounds preparations Badr et al. (2017). Secondary metabolites with promising biological activity, such as hydroxycinnamic acid amides (HCAAs), polyphenols, and steroid alkaloids, are known to be biosynthesized by Solanaceae, probably to protect themselves from phytopathogenic harm “Ajaiyeoba et al (1998); Macoy et al. (2015).” The presence of some phytochemical like flavonoids and alkaloids illustrate the medicinal action of the plant encountered in its therapeutic uses “Bamishaiye et al. (2011).” Several studies confirmed presence of several amounts of phytochemicals in Solanum species such as flavonoids, alkaloids, phenols and tannins “Manish (2012); Akilan et al. (2014); Indra et al. (2019), in family Solanaceae Popova et al. (2020).”
Antioxidant activity might be resulted from presence of polyphenolic compounds “Upadhyay et al. (2015).” The production and releasing of oxygen species (ROS) is a major cause of functional and structural modulation of cell membranes. Free radicals typically attack polyunsaturated fats in mitochondria and cellular membranes begin the uniqe source of ROS when the normal metabolic chain is impeded. In such cases, compound exhibiting antioxidant activity can be valuable in blocking the development of ROS and may also reduce oxidative cell damage, which is completely of interest if this activity is induced by a medicinal food “Lobo et al (2010).” ROS are associated in damage to most macromolecules, such as proteins, lipid and DNA, and also cause cell death “Uttara et al. (2009).” Plant secondary metabolites can be utilized to regulate the steadiness be-tween free radicals and antioxidants and can be a less dangerous alternative relative to synthetic antioxidants “Godwill (2018)”. Due to several adverse effects produced by synthetic antioxidants, the world now searching for safe therapeutic medicinal plants in the treatment of several diseases related to oxidative stress. As a result, a variety of contemporary studies have concentrated on seeking of promising novel antioxidants, especially flavonoids, and phenolics from natural resources, they have comparable characteristics but is far less poisonous and have clear and significant biological effects.
Plants generally have antioxidant properties, which have a good and various agreement of metabolites, for example, a few classes of flavonoids and non-flavonoids. These metabolites are varied in structure, OH groups, and their position is very essential to deviation in their antioxidant activity and also their biological actions “Wang et al. (2018)” Both of Herbs and vegetables especially wild species reach in antioxidant agents and several phytochemicals product is able to postpone oxidative pressure and the other related issues “Forni et al. (2019)” Therefore, the present study was designed to detect various phytochemicals present in methanolic extract of S. nigrum and S. lycopersicum and its use as a strong antioxidant agent.
The present study is an attempt to focus on identification of compounds by using GC-MS technique and biological activities of identified compounds from plant leaf extract to correlate the traditional medicine uses of Solanum with composition of extracts obtained from each plant and with their antioxidant activities. Finally, several compounds were isolated from two plant species and their structures were determined and also their antioxidant activities.
MATERIALS AND METHODS
Plant Material collection and Extraction Procedures: The whole wild of S. nigrum plants were collected in April 2020 fromFayoum governorate-Egypt, S. lycopersicum plant was collected in January 2020 from Ismailia Governorate- Egypt. The collected species washed thoroughly with distilled water. They were blotted with blotting paper, shade dried at room temperature, and ground into a fine powder. 100 gm. of powdered samples were extracted successively with 750 ml. of methanol. Extract was filtered through a 45 μm filter. The resulting solution was concentrated in vacuum to dryness to give methanol extract (5 gm.). The extract was stored at 4°C for use later.
GC-MS investigation: The phytochemical investigation of methanolic extract was performed on a GC-MS equipment “(GC/MS system: shimadza GC/MA-QP5050A, Searched library: Wiley. Software class: 5000. Column DBI : 30m : 0.53 mm ID 1.5 Mm film 9JCW scientific, Ionization model : EL, Carrier gas : helium, Ionization voltage : 70 ev, Temperature program 40 °C (1 min)- 150 °C (1min) at 3.5 °C /min- 250 °C (2min) at 5 °C /min- 270 °C (1min) at 7 °C /min, Injector temperature : 250 °C and Detector temperature : 300 °C.”
Identification of compounds was based on the molecular structure, molecular mass.: Mass spectrum data was provided using the database of online Wiley library. Molecular name, size and structure of the compounds of the test material were ascertained by correlating with the library. The biological activities were predicted using PASS online software after obtaining their SMILES by using Open Parser for Systematic IUPAC nomenclature (OPSIN).
Determination of Antioxidant Activity
2,2-diphenyl-1-picryl-hydrazyl-hydrate “DPPH assay”: Solutions of the provided samples were prepared in concentrations 1000 and 100µg/mL in methanol in order to identify a range of concentrations within which the inhibitory concentration 50 (IC50) lies. Extracts that surpassed 50% inhibition in in each of the initial screening phase were serially diluted to give 5 concentrations carried out according to the method of Boly et al. (2016) . “100µL of freshly DPPH reagent (0.1% in methanol) were added to 100µL of the sample/ compound (dissolved in MeOH) in 96 wells plate (n=3; Values are Mean of 3 replicates), the reaction was incubated at 37 °C. for 30 min in dark”. “The resulting decrease in DPPH color intensity was calculated at 540 nm at the end of the incubation period. The data was evaluated using Microsoft Excel® and the value of IC50 was determined.”
2,2-Ferric Reducing Antioxidant Power “FRAP assay”: Carried out according to the method of “Benzie et al. (1996)” but in microplates, briefly; freshly prepared “TPTZ rea-gent (300 mM Acetate Buffer (PH=3.6), 10 mM TPTZ in 40mM HCl, and 20 mMFeCl3, in a ratio of 10:1:1 v/v/v, respectively)”. 190 uL from the freshly prepared TPTZ were mixed with 10 μL of the sample in 96 wells plate (n=3; Values are Mean of 3 replicates), the reaction was incubated at room temp. for 30 min in dark. At the end of incubation time the resulting blue color was estimated at 593 nm at the end of the incubation cycle. Data was evaluated using Microsoft Excel® and ferric sample reduction capability is pre-sent as μM TE/mg.
2,2-azino-bis 3-ethylbenzothiazoline-6-sulfonic acid “ABTS assay”: According to Arnao et al. (2001) but carried out in microplates; 192 mg of ABTS were dis-solved in distilled water and transferred to 50 mL volumetric flask then the volume was completed with distilled water. 1mL of the previous solution was added to 17 µL of 140 mM potassium persulphate and the mixture was left in the dark for 24 hours. After that, 1mL of the reaction mixture was completed to 50 mL with methanol to obtain the final ABTS dilution used in the assay. 190 µL of the freshly prepared ABTS reagent were mixed with 10 µL of the sample/ compound in 96 wells plate (n=3; Values are Mean of 3 replicates), the reaction was incubated at room temp. for 120 min in dark. The decrease in ABTS color intensity was estimated at 734 nm at the end of the incubation period. The data was evaluated using Microsoft Excel® and the value of IC50 was determined.
RESULTS
Bioactive phytochemicals in the methanolic extract of S. nigrum and S. lycopersicum: Presence of diverse secondary metabolites has been reported with different structures and types present in S. nigrum revealed as different peaks and each one has different relative abundance at different retention times where the highest peak appeared after 18.81 min at figure 1.a, the variety of compound were identified and illustrated in Table 1. In S. lycopersicum revealed different peaks and each one has different relative abundance at different times where the highest peak appeared after 18.91 min at figure 1.b, the variety of compound were identified and illustrated in Table 2. The pharmacological activity of compounds varied this showing the importance of plant metabolites.
By GC-MS 27 metabolites identified in S. nigrum and 35 in S. lycopresicum. Among the 62 studied compounds 15 were found to be the most abundant compounds in the two Solanum species and these compounds were (1) 4-methyl-4-pentyl-3,4dihydrocoumarin (2) Eupomatidine (3) 5,7-dihydroxy- 4 - pentyl - 3,4 -di hydrocoumarin (4) Isoochiapin B (5) Palmitic acid (6) 3-(5-Bromoindol-3-yl)succinimide (7) 2,4 Dimethyl- 7 -phenyl pyrido[2,3-d]pyrimidin (8) 3- (4 Nitro phenyl quinazolin-4(3H)-one (9) Isoschumanniophytine (10) Isoorientin (11) Stearic acid (12) 10-(M ethoxycarbonyl)-N-acetylcolchinol (13) Noradrenaline tetra TM S (14) Epinephrine- Tetra TM S. and (15) Stearic acid.
Among 27 metabolites identified in S. nigrum about 12 metabolites characterize it, they were (1) Octyl phenol isomer (2) Phenol, 4-(1,1,3,3-tetramethylbutyl (3) 2-(Dodecyloxy) ethanol (4) (3,4-Diacetoxybenzylidene)-2-methyloxazol (5) 3-[(1-Hydroxymethyl) propyl] quinazolin (6) Dihydrovallesiachotamine (7) Fluoro metholone (8) phenylylindolizine (9) Indole-3-carboxylic acid, 5-hydroxy-6- 4-methoxyphenyl (10) 9-Octadecenoic acid (11) Dichotine (12) Lucenin 2.
Among 35 metabolites identified in S. lycopresicum about 20 metabolites characterize it, they were (1) Cinnamic acid, methyl ester (2) 1-Eicosanol (3) 4-(2-Propen-1-yloxy) benzeneamine (4) 3-cyano-4-methyl-6-ethyl-2(1H)-pyridone (5) Phenol, p- (1,1,3,3-tetramethylbutyl) (6) Ppropiolic acid, 3 - (1-hydroxy-2-isopropyl -5- methyl cyclohexyl) (7) Phenol, p-(1,1,3,3-tetramethylbutyl) (8) Tetracyclo quinone (9) Timolol (10) 2-Propanol, 1-[4-(1,1-dimethylethyl) phenoxy (11) 5,6-Benzoquinoline (12) 9-formylacridine (13) 2-Benzo[f] iso quinoline carbaldehyde (14) Quercetin 7,3,4 trimethoxy (15) 2,4 -Dimethyl -7- phenylpyrido [2,3-d]pyrimidin-5-one (16) D- erythron sphingosine (17) Stearin, 1,3-di (18) 3-(4-Nitrophenyl)quinazolin-4(3H)-one (19) 2,6-Dimethoxy -4- phenyl -7,8- quinolinedione (20) 1,2 Di-o-octa decanoyl ethandiol
Antioxidant Assay
Free radical scavenging activity in S. nigrum and in S. lycopersicum by DPPH assay: In S. nigrum the DPPH method revealed that the percent of inhibition increase with increasing concentration, where concentration 100 µg/mL yield 44.41± 3.40% inhibition while concentration 800 µg/mL yield 89.32± 1.51%. The relationship between the concentration and percent of inhibition were plotted to determine IC50 (50 % inhibitory concentration) where Ic50 found to be 38.52. (Table 3) Figure 2.a
In S. lycopersicum the DPPH method revealed that the percent of inhibition increase with increasing concentration, where concentration 100 µg/mL yield 44.65 ± 2.42 % inhibition while concentration 800 µg/mL yield 88.94 ± 1.51 % (Table 3). The relationship between the concentration and % inhibition was plotted to determine IC50 (50 % inhibitory concentration) where Ic50 found to be 37.29 (Table 3) Figure 2.b.
FRAP assay in S. nigrum and in S. lycopersicum: In S. nigrum Ferric reducing ability gave average absorbance at 593 nm = 0.682 ± 0.007), antioxidant activity using FRAP assay is = 132.57 ± 1.41 µMTE / mg, while in S. lycopersicum average absorbance at 593 nm = 0.676 ± 0.006), antioxidant activity using FRAP assay is = 131.59 ± 1.21 µMTE / mg. (Table 3).
HRP reducing ability of the S. nigrum and in S. lycopersicum: In S. nigrum average percentage inhibition of ABTS reagent at 734 nm = 65.09 ±0.035%, antioxidant activity using ABTS assay is = 142.63 ± 12.41 µMTE / mg. while in S. lycopersicum the average percentage inhibition of ABTS reagent at 734 nm = 0.727±0.035, antioxidant activity using ABTS assay is = 142.50 ± 12.08 µMTE / mg (Table 3)
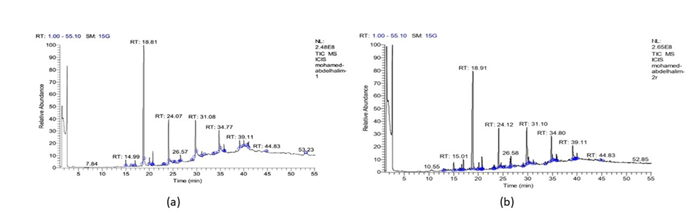
Figure 1: (a) Relative abundance of methanolic extract revealed by GC-MS analysis of leaves of S. nigrum. (b)Relative abundance of methanolic extract revealed by GC-MS analysis of leaves of S. lycopersicum.
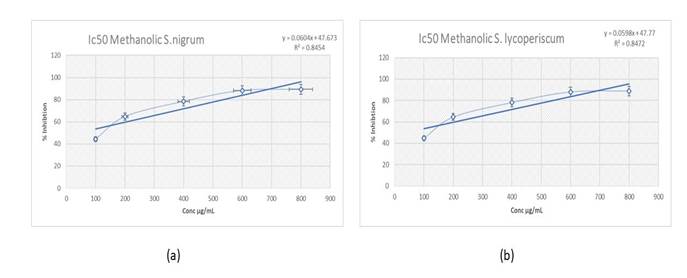
Figure (2):(a) Free radical scavenging activity at different concentrations of S. nigrum methanolic. Values are Mean of 3 replicates. (b) Free radical scavenging activity at different concentrations of S. lycopresicum methanolic. Values are Mean of 3 replicates.
Table 1: List of metabolites, their molecular formula, retention time, molecular weight and their biological activities and pharmacological activities identified in S. nigrum by GC-MS.
|
Biological activities
|
Molecular Weight
|
Molecular Formula
|
Compound Name
|
Rt
|
No
|
|
CYP2C12 substrate, Ubiquinol-cytochrome-c reductase inhibitor and Antieczematic
|
232
|
C15H20O2
|
4-methyl-4-pentyl-3,4dihydrocoumarin
|
16.94
|
1
|
|
CYP2C12 substrate,Taurine dehydrogenase inhibitor and Antiseborrheic
|
206
|
C14H22O
|
OCTYL PHENOL ISOM ER
|
18.81
|
2
|
|
Glyceryl-ether monooxygenase inhibitor, Dextranase inhibitor and Phobic disorders treatment
|
206
|
C14H22O
|
“Phenol, 4 - (1,1,3,3-tetramethylbutyl)”
|
20.02
|
3
|
|
Antiseborrheic, Antineoplastic (bladder and colon cancer) and Kinase inhibitor
|
262
|
C16H10N2O2
|
Eupomatidine
|
20.68
|
4
|
|
Eye irritation inactive, Dextranase inhibitor and Antiseptic
|
230
|
C14H30O2
|
2-(Dodecyloxy)ethanol
|
20.76
|
5
|
|
Kinase inhibitor and Aspulvinone dimethylallyltransferase inhibitor.
|
303
|
C15H13NO6
|
-(3,4-Diacetoxybenzylidene)-2-methyloxazol
|
23.02
|
6
|
|
CYP2C12 substrate, Ubiquinol-cytochrome-c reductase inhibitor and Antimutagenic
|
250
|
C14H18O4
|
5,7-dihydroxy-4-pentyl-3,4-dihydrocoumarin
|
24.07
|
7
|
|
Glycosylphosphatidylinositol phospholipase D inhibitor and Carboxypeptidase Taq inhibitor
|
250
|
C12H14N2O2S
|
3-[(1-Hydroxymethyl)propyl]quinazolin
|
24.07
|
8
|
|
Not identified
|
346
|
C19H22O6
|
ISOCHIAPIN B
|
25.09
|
9
|
|
Anesthetic general, Enzymes inhibitor and Antieczematic
|
256
|
C16H32O2
|
Palmitic acid
|
26.32
|
10
|
|
Antileukemic, Antineoplastic and Antiviral (Rhinovirus)
|
352
|
C21H24N2O3
|
Dihydrovallesiachotamine
|
26.57
|
11
|
|
Androgen antagonist, Antiallergic and Antiinflammatory, ophthalmic
|
376
|
C22H29FO4
|
Fluorometholone
|
29.07
|
12
|
|
Anticonvulsant
|
292
|
C12H9BrN2O2
|
3-(5-Bromoindol-3-yl)succinimide
|
29.77
|
13
|
|
Taurine dehydrogenase inhibitor and Glycosylphosphatidylinositol phospholipase D inhibitor
|
251
|
C15H13N3O
|
“2,4 Dimethyl -7- phenylpyrido [2,3-d] pyrimidin”
|
31.08
|
14
|
|
Lysase inhibitor and Glucan endo -1,6- beta-glucosidase inhibitor
|
267
|
C14H9N3O3
|
3-(4-Nitrophenyl)quinazolin-4(3H)-one
|
34.77
|
15
|
|
Glycosylphosphatidylinositol phospholipase D inhibitor and Mucomembranous protector
|
295
|
C22H17N
|
phenylylindolizine
|
35.79
|
16
|
|
Anaphylatoxin receptor antagonist, HIF1A expression inhibitor and Antineoplastic
|
295
|
C16H9NO5
|
Isoschumanniophytine
|
35.79
|
17
|
|
Cardioprotectant, TP53 expression enhancer and Cytostatic
|
578
|
C27H30O14
|
Isoorientin
|
35.95
|
18
|
|
Acylcarnitine hydrolase inhibitor and CYP2J substrate
|
578
|
C38H74O3
|
Stearic acid
|
39.11
|
19
|
|
Antiviral (Influenza) and CYP2H substrate
|
339
|
C20H21NO4
|
Indole-3-carboxylic acid, 5-hydroxy-6- 4-methoxyphenyl
|
39.98
|
20
|
|
CYP2J , CYP2J2 substrate and Antieczematic
|
576
|
C38H72O3
|
9-Octadecenoic acid
|
41.07
|
21
|
|
Antineoplastic
|
457
|
C20H43NO3Si4
|
Noradrenaline tetraTM S
|
44.83
|
22
|
|
CYP2E1 inducer, Leukopoiesis inhibitor and CYP2C9 inducer
|
415
|
C22H25NO7
|
10-(M ethoxycarbonyl)-N-acetylcolchinol
|
44.83
|
23
|
|
Cardiovascular analeptic, Respiratory analeptic and Spasmolytic
|
471
|
C21H45NO3Si4
|
EPINEPHRINE-TETRATM S
|
44.83
|
24
|
|
Neurodegenerative diseases treatment, Alzheimer's disease treatment and CYP2H substrate
|
502
|
C25H30N2O9
|
Dichotine
|
44.83
|
25
|
|
Cardioprotectant, Hepatoprotectant, Antioxidant and Antiviral (Herpes).
|
610
|
C27H30O16
|
Lucenin 2
|
53.15
|
26
|
|
Acylcarnitine hydrolase inhibitor and CYP2J substrate
|
594
|
C39H78O3
|
Stearic acid
|
53.15
|
27
|
[1]
Table 2: List of metabolites, their molecular formula, retention time , molecular weight and their biological activities and pharmacological activities identified in S. lycopresicum by GC-MS.
|
Biological activities
|
Molecular Weight
|
Molecular Formula
|
Compound Name
|
Rt
|
No
|
|
Membrane integrity agonist, Carminative, Insulysin inhibitor and Antihypoxic
|
162
|
C10H10O2
|
Cinnamic acid, methyl ester
|
12.99
|
1
|
|
Sugar-phosphatase inhibitor and Carboxypeptidase Taq inhibitor
|
298
|
C20H42O
|
1-Eicosanol
|
16.25
|
2
|
|
Respiratory analeptic, Eye and Skin irritation in active
|
149
|
C9H11NO
|
4-(2-Propen-1-yloxy)benzeneamine
|
16.62
|
3
|
|
CYP2C12, CYP2J substrate and Oxidoreductase inhibitor
|
232
|
C15H20O2
|
4-M ethyl-4-pentyl-3,4-dihydrocoumarin
|
16.97
|
4
|
|
CYP2C12 substrate and Testosterone 17beta-dehydrogenase (NADP+) inhibitor
|
162
|
C9H10N2O
|
3-cyano-4-methyl-6-ethyl-2(1H)-pyridone
|
16.97
|
5
|
|
Glyceryl-ether monooxygenase inhibitor and Nicotinamidase inhibitor
|
206
|
C14H22O
|
Phenol, p-(1,1,3,3-tetramethylbutyl)-
|
18.91
|
6
|
|
CYP2H substrate, Acylcarnitine hydrolase inhibitor and Carminative
|
224
|
C13H20O3
|
“Ppropiolic acid, 3- (1-hydroxy-2-isopropyl -5- methylcyclohexyl)”
|
19.92
|
7
|
|
Glyceryl-ether monooxygenase inhibitor and Dextranase inhibitor
|
206
|
C14H22O
|
Phenol, p-(1,1,3,3-tetramethylbutyl)-
|
20.10
|
8
|
|
Antiseborrheic and Antineoplastic
|
262
|
C16H10N2O2
|
Eupomatidine (9-M ethoxynaphtho [2,7]naphthyridin
|
20.70
|
9
|
|
Complement factor D inhibitor and HIF1A expression inhibitor
|
261
|
C17H11NO2
|
Tetracycloquinone
|
23.06
|
10
|
|
Adrenaline antagonist, Ophthalmic drug and Cardiotonic
|
316
|
C13H24N4O3S
|
Timolol
|
23.06
|
11
|
|
CYP2D2, CYP2C12 Substrates and Anesthetic
|
208
|
C13H20O2
|
2-Propanol, 1-[4-(1,1-dimethylethyl)phenoxy]
|
23.28
|
12
|
|
CYP2C12 substrate and Antimutagenic
|
250
|
C14H18O4
|
5,7-dihydroxy-4-pentyl-3,4-dihydrocoumarin
|
24.12
|
13
|
|
Nitrate reductase (cytochrome) inhibitor and NADPH peroxidase inhibitor
|
179
|
C13H9N
|
5,6-Benzoquinoline
|
24.12
|
14
|
|
Aldehyde oxidase inhibitor and Taurine dehydrogenase inhibitor
|
207
|
C14H9NO
|
9-formylacridine
|
24.60
|
15
|
|
Antiischemic and Antineoplastic
|
207
|
C14H9NO
|
2-Benzo[f]isoquinolinecarbaldehyde
|
24.60
|
16
|
|
Not identified
|
350
|
C19H26O6
|
ISOCHIAPIN B
|
25.11
|
17
|
|
Anesthetic general, Enzymes inhibitor and Antieczematic
|
256
|
C16H32O2
|
Palmitic acid
|
26.48
|
18
|
|
Chlordecone reductase inhibitor and Antimutagenic
|
344
|
C18H16O7
|
Quercetin 7,3,4 trimethoxy
|
26.56
|
19
|
|
Anticonvulsant
|
292
|
C12H9BrN2O2
|
3 (5-Bromoindol-3-yl) succinimide
|
29.81
|
20
|
|
Thioredoxin inhibitor
|
251
|
C15H13N3O
|
“2,4 Dimethyl-7-phenylpyrido [2,3-d] pyrimidin-5-one”
|
30.17
|
21
|
|
Thioredoxin inhibitor
|
251
|
C15H13N3O
|
“2,4 Dimethyl-7-phenylpyrido[2,3-d]pyrimidin-5-one”
|
31.10
|
22
|
|
Aspulvinone dimethylallyltransferase inhibitor
|
284
|
C16H16N2OS
|
7-Methylthio-2-methyl-5-(4-methoxyphenyl)pyrazolo[1,5-a]pyridine
|
31.10
|
23
|
|
All-trans-retinyl-palmitate hydrolase inhibitor and Eye irritation, inactive
|
624
|
C39H76O5
|
Stearin, 1,3-di
|
33.58
|
24
|
|
Glucan endo-1,6-beta-glucosidase inhibitor
|
267
|
C14H9N3O3
|
3-(4-Nitrophenyl)quinazolin-4(3H)-one
|
34.80
|
25
|
|
Sphinganine kinase inhibitor and Angiogenesis stimulant
|
299
|
C18H37NO2
|
D-erythro-C18-sphingosine
|
34.80
|
26
|
|
Nicotinic alpha4beta4 receptor agonist and JAK2 expression inhibitor
|
295
|
C17H13NO4
|
2,6-Dimethoxy-4-phenyl-7,8-quinolinedione
|
34.98
|
27
|
|
Anaphylatoxin receptor antagonist
|
295
|
C16H9NO5
|
Isoschumanniophytine
|
35.81
|
28
|
|
Cardioprotectant, TP53 expression enhancer and Cytostatic
|
578
|
C27H30O14
|
Flavone, 5,7-dihydroxy-6c-glucoside
|
35.96
|
29
|
|
L-iduronidase inhibitor and Osmotic diuretic
|
311
|
C17H24O7
|
“3,5:6,7-Di-O -cyclopentylidene -D-glycero -D-gulo -heptono-ç-lacton”
|
39.11
|
30
|
|
All-trans-retinyl-palmitate hydrolase inhibitor and Eye irritation, inactive
|
578
|
C38H74O3
|
Stearic acid, 2
|
39.65
|
31
|
|
GST A substrate and Saccharopepsin inhibitor
|
594
|
C38H74O4
|
1,2 Di-o-octa decanoyl ethandiol
|
39.70
|
32
|
|
Antineoplastic
|
457
|
C20H43NO3Si4
|
Noradrenaline tetraTM S
|
44.83
|
33
|
|
CYP2E1, CYP2C9 inducer and inducer, Leukopoiesis inhibitor
|
415
|
C22H25NO7
|
10-(M ethoxycarbonyl)-N-acetylcolchinol
|
44.83
|
34
|
|
Cardiovascular analeptic, Respiratory analeptic and Spasmolytic
|
471
|
C21H45NO3Si4
|
EPINEPHRINE-TETRATM S
|
44.83
|
35
|
Table (3): Antioxidant activity for S. nigrum and S. lycopersicum using different antioxidant markers
|
Samples
|
Antioxidant activities
|
|
DPPH (IC 50)
|
FRAP (µMTE / mg)
|
ABTS (µMTE / mg)
|
|
S. nigrum
|
38.52 ±0.25
|
132.57 ±1.41
|
142.63 ± 12.41
|
|
S. lycopersicum
|
37.29 ±0.33
|
131.5 ±1.21
|
142.50 ±12.08
|
DISCUSSION
The GC-MS analysis separated all of the metabolites in the examined samples and provided a representative spectral output. Each metabolite ideally produced a specific spectral peak. Retention time also help to discriminate between them. The size of the peaks is equivalent to the quantity of the related compounds in the analyzed specimens. Many medicinal plants have a rich supply of secondary metabolites, such as phenol, alkaloids, flavonoids, glycosides, terpenoids and tannins, determined by GC-MS “Lewis and Ausubel (2006).” There are several and different compounds of alkaloids and phenols identified in S. nigrum revealed as different peaks and each one has different relative abundance value at different times where the highest peak appeared after 18.81 min the variety of compound were identified and Relative abundance of methanolic extract revealed by GC-MS analysis of leaves of S. nigrum population are almost some of compounds identified have been reported such as phenols and acids in S. nigrum “Indra et al. (2019).” There are different compounds of alkaloids and phenols present in S. lycopersicum revealed as different peaks and each one has different relative abundance at different times where the highest peak appeared after 18.91 min, the variety of compound were identified and illustrated in Table 2, Relative abundance of methanolic extract revealed by GC-MS analysis of leaves of S. lycoperisicum.
Similar to our results, “Ammar et al. (2017)” have determined the chemical constituents in the bark of S. verbascifolium Linn. They have identified a total of 21 phytochemical product in three different extracts from the bark using GC-MS analysis. “El-Shaboury et al. (2017)” Identified 116 and 172 compounds extracted at retention time of 5 min, 88 and 138 compounds at retention time of 10 min,186 and 196 compounds extracted at retention time of 25 min and two compounds extracted at the retention time of 30 min. “Indra et al. (2019)” determined the essential chemical constituents in the bark of S. nigrum. They have identified a total of 43 phytocompounds using GC-MS analysis. In present study they were 27 and 35 compounds were identified for S. nigrum and S. lycopersicum respectively. The ratio of active constituent determines the potential of its uses for drug preparation and this is elucidated in all extracted metabolites and this is great evidence that medicinal plant metabolites are very excellent and potential source for drug preparation and treatment of diseases “Deepak and Gopal (2014).”
Oxidative stress is the main reason in a many of human disorders, including inflammation, carcinogenesis, neurodegenerative diseases, and aging. Although there are many determinants contribute to the emergence of these diseases, presumed experimental evidence links the production of ROS to biological damage, which can eventually provide a mechanistic foundation for treatment “Nistico et al. (2013); Schieber and Chandel (2014); Del Río (2015); Moloney et al. (2018); Campisi et al. (2019)”
Recently, there seems to be a substantial influence in the health impacts of several natural products, as well as in the in vivo protective function of natural antioxidants derived from medicinal plants or included in dietary food against oxidative damage caused by ROS “Muscoli et al. (2014); Loffredo et al. (2017); Rizzo et al. (2017); Carresi et al. (2018); Sharma et al. (2018).” In the present study, the antioxidant activity of extracts of plants were assessed using three different methods “(DPPH scavenging activity, FRAP assay and ABTS)” of methanolic extracts at different concentrations (200-1000 µg/mL). Trolox reagent was used as a standard and its activity was evaluated under the same conditions as the extracts revealed that the IC50 of standard was slightly lower than the sample indicate its efficacy in scavenging free radicals, have Powerful antioxidant activity, With fascinating insights for their future valorization as pharmaceuticals. The DPPH technique is based on the measurement of antioxidant scavenging potential towards the stable DPPH radical. This method offers a quick and easy to evaluate the antiradical activities of antioxidants, as the radical compound is stable and would not need to be generated as in other radical scavenging techniques “Zhang et al. (2010)”. The FRAP method is based on the ability of an antioxidant to transfer an electron for reduction to any compound, such as carbonyl groups, metals and radicals “Brand et al. (1995)”. ABTS was generated in situ in the presence of antioxidants in the lag-time assay “Benzie et al. (1999)”.
The scavenging activity of free radical is measured by the ability to quench the stable DPPH radical. The assay gave data on the reactivity of examined compounds with a stable free radical. S. nigrum and S. lycopersicum extracts were able to quench the DPPH-radical and had nearly equivalent ability. The two extracts content have different level of phenolic and alkaloids compounds, but phenolic and flavones are not significantly different. Only Timolol and Quercetin are more abundant in S. lycopersicum. Similar results obtained by “Campisi et al. (2019) ” which assess the antioxidant effect of two leave extracts of S. nigrum L. and the results showed that the extracts were able to quench the radical and restore the oxidative status in in vitro.
It can be inferred that the therapeutic effects of medicinal plants are usually attributed to their phytochemicals especially phenols and flavonoid; in our extract both of phenol and flavonoid are abundant and are the major cause for antioxidant activity. Phenols play important roles in plants, such as protection against insect pathogens and herbivores. Both of phenols and flavonoids are involved in cementing the material linking phenolic polymers to cell wall polysaccharides “Graham et al. (1994); Ilyasov et al. (2018).” Flavonoids are the most com-mon and important group of naturally occurring phenolic, because of their wide range of functions. Flavonoids generally act through a chelation or scanning. Flavonoids act as antioxidants by splitting radical chains in more stable products in the membranes of liver microsomes. They also play an significant role in protection against oxidative stress “Oriakhi et al. (2014); Martial-didier et al. (2017); Nea et al. (2021).”
Generally, antioxidants (vitamins C-, E, carotenoids, and polyphenols) are important for good bone health. They neutralize reactive particles called free radicals that are associated with all inflammatory and painful phenomena “Arulselvan et al. (2016); Nea et al. (2021).”
Recommendation: The therapeutic properties of medicinal plants can be attributed to their phytochemicals, which are the primary cause of both antioxidant activities through quenching different radicals such as DPPH, FRAP and ABTS.
REFERENCES
- Ajaiyeoba, E.O., A.U. Rahman and I.M. Choudhary (1998). Preliminary antifungal and cytotoxicity studies of extracts of Ritchiea cap-paroides var. longipedicellata. J Ethnopharmacol. 1998. 62(3), 243–246.
- Akhtar, M.S. and M. Muhammad (1989). Evaluation of the gastric antiulcerogenic effects of Solanum species of economic importance. Annals of the Missouri Botanical Garden. 75 (3).
- Akilan, C.A., C. Priya, J. Samuel and M.A. Mahalingam (2014). Comparative analysis of phytochemicals, antibiogram of selected plants in Solanaceae family and its characterization studies. International Journal of Pharmacy and Pharmaceutical Sciences. 6. 946-950.
- Ammar, A., L. Naoufal, B. Azam, G. Dennis and A. David (2017). Phytochemicals: Extraction, Isolation, and Identification of Bioactive Compounds from Plant Extracts. Plants. 6, 42.
- Anna, P., K. Piotr and S. Maciej (2019). Analytical Methods for Detection of Plant Metabolomes Changes in Response to Biotic and Abiotic Stresses. Int. J. Mol. Sci. 20, 379.
- Arnao, M. B., A. Cano and M. Acosta (2001). The hydrophilic and lipophilic contribution of total antioxidant activity. Food chemistry. 73 239-244.
- Arulselvan, P., M.T. Fard, W.S. Tan, S. Gothai, S. Fakurazi, M.E. Norhaizan and S.S. Kumar (2016). Role of Antioxidants and Natural Products in Inflammation. Oxid. Med. Cell. Longev. 5276130.
- Badr, A., H. H. El-Shazly, A.H. I. Sayed, M. Hamouda, E. El-Khateeb and M. Sakr (2017). Genetic diversity of Achillea fragrantissima in Egypt inferred from phenotypic variations and ISSR markers with reference to traits of plant size and seed yield. Plant Genetic Resources. 15 (3): 239-247.
- Bamishaiye, E. I., F.F. Olayemi, E.F. Awagu and O.M. Bamshaiye (2011). Proximate and phytochemical composition of Moringa oleifera leaves at three stages of maturation. Advance Journal of Food Science and Technology. 2011, 3(4): 233-237.
- Benzie, I.F. and J.J. Strain (1996). The ferric reducing ability of plasma (FRAP) as a measure of antioxidant power: The FRAP assay. Analytical Biochemistry. 239; 70-76.
- Benzie, I.F. and J.J. Strain (1999). Ferric reducing/antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Methods Enzyme. 299, 15.
- Boly, R., T. Lamkami, M. Lompo, J. Dubois and I. Guissou (2016) DPPH Free Radical Scavenging Activity of Two Extracts from Age-lanthus dodoneifolius (Loranthaceae) Leaves. International Journal of Toxicological and Pharmacological Research. 8(1); 29-34.
- Brand, W. ME. Cuvelier and C. Berset (1995). Use of a free radical method to evaluate antioxidant activity. Food Sci Technol.28:25–30.
- Campisi, A., R. Acquaviva, G.Raciti, A. Duro, M. Rizzo and N.A. Santagati (2019). Antioxidant Activities of Solanum nigrum L. Leaf Extracts Determined in In Vitro Cellular Models Foods. 8, 63.
- Carresi, C., V. Musolino, M. Gliozzi, J. Maiuolo, R. Mollace, S. Nucera, A. Maretta, D.Sergi, S. Muscoli and S. Gratteri (2018). Antioxidant effect of bergamot polyphenolic fraction counteracts doxorubicin-induced cardiomyopathy: Role of autophagy and c-kitpos CD45neg CD31neg cardiac stem cell activation. J. Mol. Cell. Cardiol. 119, 10–18.
- Deepak, P., and G. Gopal (2014). GC-MS analysis of various solvent extracts of bark in Solanum verbascifolium Linn. American Journal of Advanced Drug Delivery. 2. 384-392.
- Del Río, L.A. (2015). ROS and RNS in plant physiology An overview. J. Exp. Bot. 2015, 66, 2827–2837.
- Edmonds, J.M. and J.A. Chweya (1997). Black Nightshades Solanum nigrum L. and Related Species, Institute of Plant Genetics and Crop PlantResearch/ International Plant Genetic Resources Institute, Rome. 5-113.
- El-Shaboury, G. A., S. Haroun, K. Shaker and A. Badr (2017). Systematics implications of GC-MS analysis of secondary metabolites in the ethanol extract of Solanum species from South West Saudi Arabia. Egypt. J. Bot. Vol. 57, No.3,.429- 444.
- Forni C., F. Facchiano, M. Bartoli, S. Pieretti, A. Facchiano, D. D’Arcangelo, S. Norelli, G. Valle, R. Nisini, S. Beninati and C. Tabolacci (2019). Beneficial role of phytochemicals on oxidative stress and age-related diseases. Biomed Res.Int. 8748253.
- Godwill, A.E. (2018). Free radicals and the role of plant phytochemicals as antioxidants against oxidative stress-related diseases, phytochemicals source of antioxidants and role in disease prevention, Toshiki Asao and Md Asaduzzaman, Intech Open. 978-1-83881-711-4.
- Graham, Wallace and S.C. Fry (1994). Phenolic components of the primary cell wall. Inter. Rev. Cytol.51, 229–267.
- Hussein, J. H., I. Hameed and H. Al-Masudi (2017). Using Gas Chromatography-Mass Spectrometry (GC-MS) Technique for Analysis of Bioactive Compounds of Methanolic Leaves extract of Lepidium sativum. Research Journal of Pharmacy and Technology. 10.
- Ilyasov, I.R. V.L. Beloborodov and I.A. Selivanova, (2018). Three ABTS radical cation-based approaches for the evaluation of antioxidant activity: Fast- and slow reacting antioxidant behavior. Chem. Pap. 72, 1917–1925.
- Indra, R. P. Asifa, S.Pragati and D. S. Manish (2019). GC-MS analyses of ethanolic leaf extract of medicinal plant solanum nigrum. World Journal of Pharmaceutical Research. 8, Issue 7, 2299-2307.
- Lewis, K. and F. Ausubel (2006). Prospects for plant-derived antibacterials. Nat Biotechnol., 24, 1504–1507.
- Lobo, V., A. Patil, A. Phatak and N. Chndra (2010). Free radicals, antioxidants and functional foods: Impact on human health. Phar-macogn. Rev. 4, 118–126.
- Loffredo L., L. Perri, C. Nocella and F. Violi (2017). Antioxidant and antiplatelet activity by polyphenol-rich nutrients:Focus on extra virgin olive oil and cocoa. Br. J. Clin. Pharmacol. 2017, 83, 96–102.
- Macoy, D. M., W.Y. Kim, S.Y. Lee and M. G. Kim (2015). Biosynthesis, physiology, and functions of hydroxycinnamic acid amides in plants. Plant Biotechnol. Rep. 2015, 9 269–278.
- Mahomoodally, M.F. (2013). Traditional medicines in Africa: An appraisal of ten potent African medicinal plants. Evid-Based Complement Alternat Med. 617459, 14.
- Manish, T. (2012). Phytochemical contents and antimicrobial activity of Solanum sisymbriifoloum extracts. Journal of Applied Phar-maceutical Science. 4(3):75-80
- Manoko, M.L.K., R.G. Van den Berg, R.M.C. Feron, G.M. Van der Weerden and C. Mariai (2007). AFLP markers support separation of Solanum nodiflorum from Solanum americanum (Solanaceae). Plant Systematics and Evolution, 2007, 267, 1-11.
- Martial-didier, A.K., K.K. Hubert, K. Eugène, J. Parfait, T. Kablan (2017). Phytochemical Properties and Proximate Composition of Papaya (Carica papaya L. var solo 8) Peels. Turk. J. Agric. Food Sci. Technol. 5, 676–680.
- Mohasana, A., A. Sanzida, A. Asif, R. Razina, A. S. Jamil, T. Evelin, D. S. Satyajitand and J. U. Shaikh (2020). Ethnobotany and Antimicrobial Peptides from Plants of the Solanaceae Family: An Update and Future Prospects. Front. Pharmacol. 2020,11:565.
- Moloney, J.N. and T.G. Cotter (2018). ROS signalling in the biology of cancer. Semin. Cell Dev. Biol. 80, 50–64.
- Muscoli, C., F. Lauro, C. Dagostino, S. Ilari, L.A. Giancotti, M. Gliozzi, N. Costa, C. Carresi, V. Musolino and F. Casale (2014). Olea Europea-derived phenolic products attenuate antinociceptive morphine tolerance:An innovative strategic approach to treat cancer pain. J. Biol. Regul. Homeost. Agents. 28, 105–116.
- Nea, F. M.B. Bitchi, M. Genva, A. Ledoux, A.T. Tchinda, C. Damblon, M. Frederich, Z.F. Tonzibo and M.L. Fauconnier (2021). Phy-tochemical Investigation and Biological Activities of Lantana rhodesiensis. Molecules., 26, 846.
- Nistico, S., D. Ventrice, C. Dagostino, F. Lauro, S. Ilari, M. Gliozzi, M.C. Strongoli, , I. Vecchio, M. Rizzo, V. Mollace and C. Muscoli (2013). Effect of MN (III) tetrakis (4-benzoic acid) porphyrin by photodynamically generated free radicals on SODs keratinocytes. J. Biol. Regul. Homeost. Agents. 27, 781–790.
- Oriakhi, K., E.I. Oikeh, N. Ezeugwu, O. Anoliefo and O. Aguebor (2014). Comparative Antioxidant Activities of Extracts of Vernonia amygdalina and Ocimum gratissimum Leaves. J. Agric. Sci. 2014, 6, 13–20.
- Popoola, J.O., O.S. Aworunse, O.L. Oyesola, O.O. Akinnola and O.O. Obembe (2020). systematic review of pharmacological activities and safety of Moringa oleifera. J Herbmed Pharmacol. 9, 174–190.
- Popova, V., A. Stoyanova and N. Mazova (2020). Phytochemical composition and biological activity of Physalis spp.: A mini-review. Food Science and Applied Biotechnology. 2020, 3. 56.
- Rizzo, M., D. Ventrice, F. Giannetto, S. Cirinnà, N.A. Santagati, A. Procopio, V. Mollace and C. Muscoli (2017). Antioxidant activity of oleuropein and semisynthetic acetyl-derivatives determined by measuring malondialdehyde in rat brain. J. Pharm. Pharmacol. 69, 1502–1512.
- Saijo, R., K. Murakami, T. Nohara, A. Tomimatsa, A. Saito and K. Matsuoka (1982). Studies on the constituents of Solanum plants II: Constituents of the immature fruits of Solanum nigrum. YakugakuZasshi. 102, 300-305.
- Schieber, M. and N.S. Chandel (2014). ROS function in redox signaling and oxidative stress. Curr. Biol., 24, 453–462.
- Sharma, A., M. Kaur, J.K. Katnoria and A.K. Nagpal (2018). Polyphenols in Food: Cancer Prevention and Apoptosis Induction. Curr. Med. Chem. 25, 4740–4757.
- Upadhyay, P., S. Ara and P. Prakash (2015). Antibacterial and Antioxidant Activity of Solanum nigrum Stem and Leaves. Chem. Sci. 4, 1013–1017.
- Uttara, B., A.V. Singh P. Zamboni and R.T. Mahajan (2009). Oxidative stress and neurodegenerative diseases: a review of upstream and downstream antioxidant therapeutic options. Curr. Neuropharmacol. 7(1):65–74.
- Wang, TY., Q. Li and K.S. Bi (2018). Bioactive flavonoids in medicinal plants: structure, activity and biological fate. Asian J. Pharm. Sci. 13(1):12–23.
- Witte, L., K. Muller and H. A. Arfmann (1987). Investigation of the alkaloids pattern of Datura innoxia plants by capillary gmass spec-troscopy. Planta Medica. 192-197.
- Zadra, M., M. Piana, F. T. Brum, A. A. Boligon, B. R. Freitas, M. M. Machado, T. S. Stefanello, A. A. F. Soares and L. M. Athayde (2012). Antioxidant activity and phytochemical composition of the leaves of Solanum guaraniticum A. St.-Hil. Molecules. 17, 12560-12574.
- Zhang, Y., X. Li and Z. Wang (2010). Antioxidant activities of leaf extract of Salvia miltiorrhiza Bunge and relatedphenolic constituents. Food Chem. Toxicol. 48, 2656–2662. [1] Rt= Retention time
|





