INFLUENCE OF NANO ZINC OXIDE ON THE IN VITRO CALLUS GROWTH, EX VITRO TUBER YIELD AND NUTRIONAL QUALITY OF POTATO (Solanum tuberosum L.) CULTIVARS UNDER SALT STRESS
E. S. Alghamdi1, M. Farooq2 and E.M.R. Metwali3*
1Food and Nutrition Department, College of Human Science and Designs, King Abdulaziz University, 21589 Jeddah, Saudi Arabia.
2Department of Plant Sciences, College of Agricultural and Marine Sciences, Sultan Qaboos University, 123 Al-Khoud, Oman
3Genetic Branch, Botany Department, Faculty of Agriculture, Suez Canal University, 41522 Ismailia, Egypt.
*Corresponding author’s email: ehab_ghareb@agr.suez.edu.eg ; asalghamdy2@kau.edu.sa
ABSTRACT
Potato (Solanum tuberosum L.) is one of the leading vegetable crops around the world. However, its growth, yield, and quality are reduced by several abiotic stresses such as salt stress. This study, consisted of two independent experiments was conducted to examine the effects of salt stress in potato and influence of nanoparticle-sized zinc (ZnO-NPs) in improving salt tolerance. The first experiment was conducted under in vitro conditions to evaluate the regenerated callus growth in response to the presence or absence of ZnO-NPs under different levels of NaCl (0, 17, 34, 51 and 68 mM). The second experiment was conducted using in vitro derived plantlets treated ex vitro with ZnO-NPs to evaluate its effect on tuber yield and nutritional quality under salt stress (0.0, 1.0, 2.0, 3.0 and 4.0 g L-1). In both experiments, ZnO-NPs were applied at 0.0 or 50 mg L-1 to three potatoes cvs. Spunta, Nicola, and Hermes raised under five levels of NaCl-induced salt stress: 0, 17, 34, 51 and 68 mM. Salt stress significantly reduced the in vitro callus growth progressively with increasing NaCl levels, while ZnO-NPs had a positive impact on growth under salt stress. Potato tubers harvested ex vitro differed significantly for calcium and iron under different levels of salt stress in response to ZnO-NPs application. The number of tubers responded negatively to increasing levels of NaCl with or without ZnO-NPs application. All three potato cultivars, grown under 34 mM NaCl, regardless of ZnO-NPs application, had a significantly higher number of tubers than when grown at other NaCl concentrations. The highest number of tubers was recorded for all three tested potato cultivars receiving ZnO-NPs under non-saline conditions, whereas the lowest number was recorded in plants under high levels of salinity, with or without ZnO-NPs. The cv. Spunta was the least affected by salt stress followed by the cv. Hermis and cv. Nicola for most traits. Application of ZnO-NPs proved effective in ameliorating the salinity-induced harmful effects on yield and quality of potato.
Keywords: Potato, nanoparticles, NaCl, meristem culture, calcium, iron, protein
Abbreviation: AdSO4 = Adenine sulfate; CFW = Callus fresh weight; BA = Benzyl adenine; GA3 = Gibberellic acid; MS = Mushige and Skoog's medium; NAA = Napthalene acetic acid; NaCl = Sodium chloride; NPs = Nanoparticles; ZnO = Zinc oxide; PSi: Pound per square inch; Calcium (Ca); Iron (Fe).
https://doi.org/10.36899/JAPS.2022.2.0441
Published first online August 13, 2021
INTRODUCTION
Potato (Solanum tuberosum L.) is one of the leading vegetable crops grown and consumed worldwide (Liao et al., 2016). However, its sustainable production and quality are constrained by several challenges including temperature extremes drought, and salinity (Hijmans 2003; Levy et al., 2013). Among these, salt stress is one of the major abiotic stresses which limits the growth and productivity of all the crops including potato (Jaarsma et al., 2013). In general, potato is considered as moderately sensitive to salt stress (3-4 g L-1) (Batelli et al., 2012). However, there exists genotypic variation, among potato genotypes, to salt stress. For instance, potato cv. Draja is moderately sensitive to salt stress whereas cv. Sponta is salt tolerant and cv. Diamont is salt sensitive (Jaarsma et al., 2013).
Salt stress disturbs the plant water relations and causes reduction in plant photosynthesis performance (Akhtar et al., 2015). At the cellular level, salt stress increases the cell vacuolization, causes swelling of the thylakoid membrane, and reduces the stacking of grana (Fidalgo et al., 2004). These changes affect the yield and quality of economic plant parts (Farooq et al., 2017). Although potato genotypes may vary in their response to salinity (Aghaei et al., 2009), a linear decrease was observed in most of growth and chemical traits such as shoot length, fresh and dry weight, and K+ content, respectively in all genotypes with the increase in severity of salt stress (Ahmed et al., 2020).
In addition to carbohydrates, potato tubers also contain dietary proteins and fair amounts of minerals (Martínez-Ballesta et al., 2010). The potato tuber dietary protein helps to reduce blood cholesterol by increasing the circulation of cholesterol levels (Seo et al., 2014; Gambuti et al., 2016). However, drought stress may result in significant (P < 0.0001) increase in tuber soluble protein (Wegener et al., 2015). Potato tuber has a higher concentration of iron 41.09 mg kg−1 dry weight and traces of calcium 4.3 mg kg−1 dry weight (Gąsiorowska et al., 2018). However, Farooq et al. (2017) noticed that salt stress may reduce the uptake of micronutrients due to stronger competition by salt cations at the root surface.
Zinc (Zn) is one of the essential microelements and plays a key role in carbohydrate metabolism, protein biosynthesis, and gene expression related to abiotic stresses (Rehman et al., 2018a; Rossi et al., 2014). Plants raised from seed with high intrinsic Zn can grow well and yield better under less than optimum conditions (Faran et al., 2019). An adequate supply of Zn enables plants to tolerate abiotic stresses (Rehman et al., 2019). External Zn application may help improve nutrient uptake and modulate the key enzymes involved in plant metabolism (Aktas et al., 2006). The use of nanoparticles (NPs) is a novel approach in plant micronutrition (Liu and Lal 2015). Application of NP (1-100 nm) fertilizers helps to improve the nutrient absorption, translocation, and use efficiency compared to traditional fertilizers of non-nanoparticles sizes (Usman et al., 2020). Therefore, the application of Zn-containing nanoparticles can help in improving Zn availability. Application of Zn as ZnO-NPs has been very effective in improving the germination, metabolism, and growth of some vegetable crops such as cabbage, cauliflower, habanera peppers, basil and tomato (García-López et al., 2019).
The application of tissue culture has gained much momentum in plant improvement programs and commercial scale plant propagation under optimal and less than optimum conditions (Gu et al., 2004). Under salt stress, tissue culture technique may help to isolate salt-tolerant cells from successfully regenerated (Lutts et al., 2004; Bündig et al., 2017; Gowayed et al., 2017). The technique would allow mass screening of genotypes in a very limited space and time.
Zinc application helps improving salt tolerance in crop plants including potato (Aktas et al., 2006; Mahmoud et al., 2020). However, Zn application as NPs was found more effective than other forms of Zn (Hussein and Abou-Baker 2008). To the best of our knowledge, the influence of ZnO-NPs on the in vitro growth, and subsequent ex vitro tuber yield and quality of potato under salt stress has not been investigated. This study was, therefore, conducted to investigate the influence of ZnO-NPs on in vitro growth and in vivo tuber yield and quality of three potato cultivars under NaCl-induced salt stress.
MATERIALS AND METHODS
Plant materials and experimental conditions: Virus-indexed potato tubers of cultivars Spunta, Nicola, and Hermes, collected from Agricultural Research Center, Egyptian Ministry of Agriculture, Cairo, Egypt. The experiments were conducted during the period of July 2018 to May 20219 at Tissue Culture Laboratory, SCU, Egypt and Nutrition Lab, KAU, Saudi Arabia. Seeds were allowed to sprout in clay pots filled with wetted Vermiculite in a greenhouse. Sprouts (3 cm long) were taken, and surface sterilized with 10% Clorox for 5 min followed by washing three times with sterilized water. Meristem tip explants (0.5 mm) were isolated under a binocular and were cultured in test tubes containing 10 mL agar-solidified (7.0 g L-1) MS medium (Murashige and Skoog 1962) containing basal salts, vitamins and supplemented with sucrose (3%). One shoot tip explant was cultured per tub, using 20 culture test tubes per each potato cultivar to obtain enough plantlets for further experiment. Medium pH was adjusted to 5.7 before the addition of agar, and the medium was autoclaved for 20 min at 121°C and 15 psi pressure. Cultures were incubated at 24 °C under cool-white fluorescent lamps (45 µmol m-2 s-1 at culture level) under a 16-h photoperiod for the development of plantlets.
Preparation of nano-ZnO particle suspension: Nanoparticles of ZnO with size of 30 nm, were purchased from Sigma–Aldrich Company, California, USA. For the preparation of nano-ZnO particle suspension, with concentrations of 50 mg L-1, 1.5 g ZnO-NPs was dissolved in a liter of distilled water following to Kim et al. (2017). Good dissolution and homogeneity of the solution were ensured using a sonicator. The nanoparticle suspensions were centrifuged at 3000 × g under cooling (-40C) for one hour and then filtered using 0.7 μm glass filter. The filtrate was used for addition to the culture media.
Embryogenic callus and experimental details: Leaf disk explants (5 × 5 mm) from the fourth leaves (Fig. 1) of the previously formed potato plantlets were in vitro cultured on MS medium supplemented with sucrose (3%), benzyl adenine (BA; 3.0 mg L-1) + naphthaleneacetic acid (NAA; 2.0 mg L-1) for callus induction (Kumlay and Ercisli 2015). Media were adjusted for pH at 5.7, solidified with 7.0 g L-1 agar, and sterilized by autoclaving for 20 min at 121°C and 15 psi pressure. Cultures were incubated at 25°C in the dark for one week, followed by 4 weeks at 16 h photoperiod using cool white fluorescent light (45 µmol m-2 s-1). The medium was refreshed by subculture the calli every 12 days to maintain the presence and effectiveness of ZnO-NPs. After 5 weeks the soft, friable and light-yellow primary calli (about 1 cm in diameter) induced from the previous experiment were subcultured into fresh MS medium supplemented with gibberellic acid (GA3; 1.0 mg L-1) + adenine sulfate (AdSO4; 20 mg L-1) + BA (2.0 mg L-1) for the induction of embryonic callus under the influence of ZnO-NPs and NaCl stress following Khatri et al. (2019) with minor modification. For this aim, medium treatments were: T1=0.0 mM NaCl; T2=17.00 mM NaCl; T3=34.00 mM NaCl; T4=51.00 mM NaCl; T5=68.00 mM NaCl; T6= 50.00 mg L-1 ZnO-NPs; T7= 17.00 mM NaCl+50.00 mg L-1 ZnO-NPs; T8= 34.00 mM NaCl +50.00 mg L-1 ZnO-NPs; T9= 51.00 mM NaCl+50.00 mg L-1 ZnO-NPs; T10= 68.00 mM NaCl +50.00 mg L-1 ZnO-NPs. The choice of these gradients in concentrations of NaCl was based on previous studies (Aghaei et al. 2008) and pre-experiments. Media preparation and culture incubation conditions were the same as described above. Jars were arranged in a growth room in a factorial completely randomized design (CRD). There were 8 jars per treatment, each jar representing a replication, with a total 80 jars. Seven weeks later, regenerated callus mass/treatment was assessed randomly to record the callus fresh and dry weights.

Fig. 1: (a) Leaf disc explants taken from plantlets of potato cultivars for callus induction under in vitro NaCl stress and (b) Potato leaf node (5×5 mm) used as explants under Binuclear microscope
Ex vitro experiment: To examine the tuberization potential of the tested potato cultivars under salt stress, 5-week-old in vitro regenerated plantlets from the previous experiment, of cvs. Spunta, Nicola and Hermes were removed from the culture jars, washed with tap water to remove the excess of agar and soaked in fungicide solution (2.0 g L-1 Benlate) for 5 min, and transferred into a glasshouse. Plantlets were cultured in clay pots (25 cm) filled with peat moss and vermiculite (3:1 v/v), each pot included 3 plantlets. Salt and ZnO-NPs treatments were applied through irrigation with nutrient solution containing 2 g L-1 of (19-19-19 NPK) supplemented with NaCl at (0.0, 1.0, 2.0, 3.0 and 4.0 g L-1) and ZnO-NPs at 0.0 and 50 mg L-1 twice a week starting from 01 January 2019 to 28 February 2019. Potato tubers were harvested on 30 March 2019. The plants were kept at 22/16°C (16 day/8 night) and undergone an average humidity of 65% for the whole cultivation phase. Data on tuber number and weight were recorded of at final harvest.
Chemical analysis of potato tubers: Samples of tuber were dried at 70°C in an oven then ground. Dry powdered samples (0.5 g) were digested by a wet digestion method following Jackson (1973). Total tuber nitrogen was determined by the Kjeldahl method (Chapman and Pratt, 1961). Crude protein was estimated by multiplying total nitrogen with a factor 6.25 (AOAC, 1995). Total calcium (Ca) and iron (Fe) concentration in tuber extracts were determined by an Inductively Coupled Plasma (ICP-OES, Spectro Analytical Instruments, Kleve, Germany) according to Sen and Pendam (2016).
Statistical analysis: Experimental data were analyzed by Fisher’s analysis of variance using statistical software Costat (2011). Duncan’s new multiple range test was used for mean separation (Duncan, 1955).
RESULTS
The analysis of variance showed that potato cultivars differed significantly (p≤0.001) for callus fresh (CFW) and dry (CDW) weights, tuber number, tuber weight, tuber protein, tuber iron and tuber calcium. Salt stress and application of ZnO-NPs also affected all the studied parameters significantly, except for tuber protein by ZnO-NPs application (Table 1). Two-way (cultivars × salinity and salinity × ZnO-NPs) and three-way (cultivars × salinity × ZnO-NPs) interactions were significant for all the parameters except for CDW and number of tubers in case of three-way interaction. Two-way interaction of cultivars × ZnO-NPs was only significant for CFW, tuber protein, and tuber iron (Table 1).
Under in vitro culture conditions, embryogenic calli were successfully induced and proliferated on MS medium supplemented using varying plant growth regulators (Fig. 2). The maximum CFW was recorded in cv. Spunta (0.315 g) followed by cv. Hermes (0.152 g) and cv. Nicola (0.124 g) over their respective control. Comparing the results of different levels of salinity and ZnO-NPs on CFW showed that addition of NaCl salt to the growth medium led to a negative impact on the growth of callus, which caused a reduction in CFW and this effect is directly related with the concentration of NaCl salt in the medium (Fig. 2). The cultivar Spunta was the least affected by the salt stress followed by the cv. Hermis and cv. Nicola (Fig. 3). The application of ZnO-NPs significantly improved the growth of callus under salt stress and normal conditions. Highest CFW (0.491 ± 0.064a; 0.270 ± 0.058a; 0.378 ± 0.091 a) were recorded in cvs. Spunta, Nicola and Hermis with the application of ZnO-NPs under 17.0, 68.0, and 34.0 mM NaCl levels of salt stress, respectively (Fig. 3). Application of ZnO-NPs, under salt stress levels to 17 and 34 mM NaCl in the growth media, produced the highest CDW in cvs. Spunta (0.0351 g) and Hermes (0.0194 g), while the CDW was highest in cv. Nicola (0.0194 g) under control treatment without ZnO-NPs application (Fig. 3).
Table 1: Analysis of variance for the influence of salt concentration and ZnO-NPs on callus growth (g), tuber protein (%), tuber minerals (%) and yield components (g) of potato cultivars
|
SOV
|
df
|
Mean Sum of Squares
|
|
CFW
|
CDW
|
Protein
|
Calcium
|
Iron
|
Tubers number
|
Tuber weight
|
|
Main effects
|
|
|
|
|
|
|
|
|
|
Cultivars
|
2
|
0.1289***
|
5.670***
|
0.3574***
|
0.0124***
|
21.49***
|
69.03***
|
113.40***
|
|
Salinity
|
4
|
0.0358***
|
2.465*
|
0.0480***
|
0.0123***
|
223.54***
|
66.15***
|
48.304***
|
|
Zinc-NPs
|
1
|
0.1688***
|
4.199*
|
0.0033ns
|
5.329***
|
13.814***
|
12.1**
|
102.122***
|
|
Interactions
|
|
|
|
|
|
|
|
|
|
Cultivars × Salinity
|
8
|
0.0215***
|
2.336*
|
0.0284***
|
0.002***
|
9.040***
|
3.908*
|
19.92***
|
|
Cultivars × Zinc-NPs
|
2
|
0.0234**
|
2.500ns
|
0.0128***
|
7.09ns
|
0.898***
|
1.633ns
|
2.097ns
|
|
Salinity × Zinc-NPs
Cultivars × Salinity × Zinc-NPs
|
4
8
|
0.0161**
0.0201***
|
2.613*
1.692 ns
|
0.0179***
0.0064***
|
2.87**
1.593**
|
1.033***
0.587***
|
5.405*
1.230 ns
|
5.082***
11.21***
|
|
Error
|
68
|
0.0040
|
9.040
|
1.723
|
4.5722
|
0.0182
|
1.522
|
0.783
|
|
CV%
|
|
30.90
|
64.48
|
1.0098
|
1.971
|
1.898
|
17.056
|
5.250
|
*, **, ***Significant at p≤0.05, p≤0.01 and p≤0.001, respectively.
ns = Non-significant; SOV = Source of variance; df = Degrees of freedom; CV = Coefficient of variance; CFW = Callus fresh weight; CDW = Callus dry weight

Fig. 2: Six-week-old embryogenic callus of potato cultivars Spunta (Sp.), Nicola (Ni.) and Hermis (H.) under different degrees of salt stress minus (T1-T5) or plus (T6-T10) addition of ZnO-NP to MS medium.
T1=0.0 mM NaCl; T2=17.00 mM NaCl; T3=34.00 mM NaCl; T4=51.00 mM NaCl; T5=68.00 mM NaCl; T6= 50 mg L-1 ZnO-NPs; T7= 17.00 mM NaCl+50 mg L-1 ZnO-NPs; T8= 34.00 mM NaCl +50 mg L-1 ZnO-NPs; T9= 51.00 mM NaCl+50 mg L-1 ZnO-NPs; T10= 68.00 mM NaCl +50 mg L-1 ZnO-NPs.
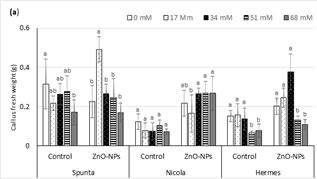 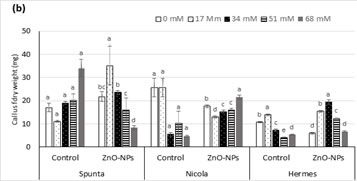
Fig. 3. Influence of ZnO-NPs on (a) callus fresh and (b) dry weight of potato cultivars under salt stress.
Data are represented as mean ± standard deviation from three replicates.
Means sharing the same letters, within each set of bars for a parameter, do not differ significant at p ≤ 0.05.
Salt stress significantly reduced the tuber protein in potato cultivars. However, potato cultivars differ greatly for tuber protein content at different salt stress levels. The cultivars also respond differently to ZnO-NPs application. In cv. Spunta, lowest tuber protein was recorded at 34 mM NaCl stress for plants without ZnO-NPs application, whereas plants receiving ZnO-NPs application accumulated more tuber protein when grown at 0.0 or 34 mM NaCl. Nevertheless, the highest tuber protein contents were observed at 51 mM NaCl salinity stress with or without ZnO-NPs application (Table 2). In cv. Nicola, the highest tuber protein was recorded at 51 and 68 mM NaCl-induced salinity stress with and without ZnO-NPs application respectively, while plants grown under control or at 17 mM NaCl stress condition exhibited lowest tuber protein contents irrespective of ZnO-NPs application. In cv. Hermes, potato tuber harvested from plants grown at 34 mM NaCl with ZnO-NPs application accumulated the highest protein, while plants receiving ZnO-NPs accumulated more protein in tubers when grown at 17 and 34 mM NaCl. However, the lowest tuber protein was observed in plants grown under normal growth conditions irrespective of ZnO-NPs application (Table 2).
Salinity stress significantly decreased tuber calcium. However, the cultivars greatly differed for calcium (Ca) concentration and response to ZnO-NPs application. In the case of cv. Spunta, plants grown under control condition, and at 34 mM NaCl without ZnO-NPs application showed the highest tuber calcium accumulation, while plants the receiving ZnO-NPs accumulated more tuber calcium under control conditions. Furthermore, the highest salinity level (68 mM NaCl) caused the farthest decline in tuber calcium concentration irrespective of ZnO-NPs application (Table 2). In cv. Nicola plants grown at normal conditions and not receiving ZnO-NPs had more calcium, while the lowest tuber calcium was recorded in plants grown at mild salinity stress (17 mM NaCl). However, potato plants receiving ZnO-NPs application accumulated more calcium in tubers at 51 mM NaCl, while the lowest tuber calcium concentration was recorded at 34 mM NaCl salinity level. In cv. Hemes, the highest and lowest Ca concentration was recorded in plants grown under normal and extreme saline conditions (68 mM NaCl) irrespective of ZnO-NPs application (Table 2).
Salt stress drastically reduced the tuber iron concentration in all three cultivars. Cultivars also differed significantly for tuber iron concentration. However, ZnO-NPs application reduced the extent of decrease in tuber iron concentration in all cultivars. In cv. Spunta, the lowest tuber iron concentration was recorded for a plant grown at 68 mM NaCl salinity stress, whereas the highest tuber iron concentration was recorded in control plants irrespective of ZnO-NPs application. In cv. Nicola, the highest iron concentration was recorded in control plants receiving no ZnO-NPs; while those treated with ZnO-NPs accumulated more iron when grown at 17 mM NaCl. Moreover, plants of cv. Hermes treated with ZnO-NPs showed more iron accumulation under noon-saline conditions, while plant grown with ZnO-NPs supply had the highest tuber iron concentration at 17 mM NaCl. Nevertheless, lowest tuber iron concentration was recorded for plants grown at 68 mM NaCl salinity stress, irrespective of ZnO-NPs application (Table 2).
The highest number of tubers were recorded for all three tested potato cultivars receiving ZnO-NPs under non-saline conditions, whereas the lowest number of tubers was recorded in plants under high levels of salinity with or without ZnO-NPs (51 mM NaCl; 68 mM NaCl; 51 mM NaCl + ZnO-NPs; 68 mM NaCl +ZnO-NPs). Overall, the number of tubers responded negatively to increasing levels of NaCl treatment with or without ZnO-NPs application. All three potato cultivars grown under 34 mM NaCl, regardless of ZnO-NPs application, had a significantly higher number of tubers compared to other treatments (Table 4). A significant increase in tuber weight, of tested potato cultivars, was recorded under 51.00 mM NaCl, regardless of ZnO-NPs treatment. However, the average tuber weight was highest under 34 mM NaCl in cv. Hermes (Table 3; Fig. 4).

Fig. 4. Influence of ZnO-NPs on tuber yield of potato cv. Hermis under salt stress T1=0.0 mM NaCl; T2=17.00 mM NaCl; T3=34.00 mM NaCl; T4=51.00 mM NaCl; T5=68.00 mM NaCl; T6= 50 mg L-1 ZnO-NPs; T7= 17.00 mM NaCl+50 mg L-1 ZnO-NPs; T8= 34.00 mM NaCl +50 mg L-1 ZnO-NPs; T9= 51.00 mM NaCl+50 mg L-1 ZnO-NPs; T10= 68.00 mM NaCl +50 mg L-1 ZnO-NPs.
Table 2: Influence of ZnO-NPs on tuber protein, calcium and iron in three potato cultivars under NaCl-induced salt stress in vivo
|
Cultivars
|
Spunta
|
Nicola
|
Hermes
|
|
ZnO-NPs
NaCl mM
|
0.0 mg L-1 ZnO-NPs
|
50 mg L-1 ZnO-NPs
|
0.0 mg L-1 ZnO-NPs
|
50 mg L-1 ZnO-NPs
|
0.0 mg L-1 ZnO-NPs
|
50 mg L-1 ZnO-NPs
|
|
Tuber protein (%) per 0.5 g dw
|
|
00.00
|
1.410±0.0007 b
|
1.391±0.008 d
|
1.142±0.001 d
|
1.140±0.001 d
|
1.126±0.001 e
|
1.159±0.006 c
|
|
17.00
|
1.415±0.0014 b
|
1.491±0.001 c
|
1.1469±0.001 d
|
1.149±0.001 d
|
1.136±0.0008 d
|
1.366±0.011 a
|
|
34.00
|
1.377±0.001 c
|
1.377±0.010 d
|
1.1822±0.001 c
|
1.207±0.006 c
|
1.356±0.0013 a
|
1.388±0.0037 a
|
|
51.00
|
1.514±0.010 a
|
1.521±0.009 a
|
1.4132±0.001 a
|
1.220±0.007 b
|
1.295±0.0043 b
|
1.233±0.0057 b
|
|
68.00
|
1.376±0.001 c
|
1.375±0.001 d
|
1.291±0.009 b
|
1.311±0.002 a
|
1.229±0.0095 c
|
1.260±0.0143 b
|
|
LSD( p ≤ 0.05)
|
0.0083
|
0.0133
|
0.0071
|
0.0097
|
0.0086
|
0.0543
|
|
Tuber calcium (%) per 0.5 g dw
|
|
00.00
|
0.372±0.002a
|
0.381±0.006a
|
0.353±0.006a
|
0.346±0.011b
|
0.416±0.005a
|
0.411±0.001a
|
|
17.00
|
0.346±0.006b
|
0.341±0.001c
|
0.290±0.005d
|
0.294±0.005c
|
0.314±0.004c
|
0.361±0.005d
|
|
34.00
|
0.363±0.007a
|
0.367±0.001b
|
0.303±0.005c
|
0.294±0.005d
|
0.362±0.003b
|
0.361±0.005c
|
|
51.00
|
0.337±0.007b
|
0.339±0.005c
|
0.342±0.001b
|
0.360±0.005a
|
0.369±0.018b
|
0.344±0.005b
|
|
68.00
|
0.308±0.008c
|
0.321±0.010d
|
0.308±0.008c
|
0.303±0.005cd
|
0.327±0.001c
|
0.327±0.004d
|
|
LSD( p ≤ 0.05)
|
0.0125
|
0.0129
|
0.0108
|
0.0120
|
0.0159
|
0.0084
|
|
Tuber iron (ppm)
|
|
00.00
|
9.85±0.045a
|
9.79±0.085a
|
11.326±0.094a
|
11.516±0.076b
|
10.503±0.240b
|
10.55±0.287a
|
|
17.00
|
9.683±0.104b
|
6.066±0.058b
|
10.93±0.065b
|
11.72±0.070a
|
10.55±0.0866a
|
9.426±0.087b
|
|
34.00
|
5.05±0.050c
|
6.066±0.058b
|
5.146±0.136c
|
6.486±0.070c
|
8.676±0.0862c
|
9.426±0.087b
|
|
51.00
|
3.856±0.055d
|
3.95±0.10c
|
2.526±0.176d
|
4.303±0.035d
|
6.306±0.325d
|
8.30±0.090c
|
|
68.00
|
2.836±0.060e
|
3.303±0.100d
|
2.04±0.062e
|
2.38±0.155e
|
1.666±0.090e
|
3.75±0.219d
|
|
LSD( p ≤ 0.05)
|
0.1209
|
0.1602
|
0.2102
|
0.1659
|
0.3514
|
0.3570
|
Data are represented as mean ± Standard deviation from three replicates.
Means sharing the same letters, within a column for a parameter, do not differ significant at p ≤ 0.05
Table 3: Influence of ZnO-NPs on number of tubers and tuber weight of potato cultivars under salt stress in vivo.
|
Cultivars
|
Spunta
|
Nicola
|
Hermes
|
|
ZnO-NPs NaCl mM
|
0.0 mg L-1 ZnO-NPs
|
50 mg L-1 ZnO-NPs
|
0.0 mg L-1 ZnO-NPs
|
50 mg L-1 ZnO-NPs
|
0.0 mg L-1 ZnO-NPs
|
50 mg L-1 ZnO-NPs
|
|
Number of tubers/plants
|
|
00.00
|
12.33±0.577a
|
12.66±0.577a
|
8.00±1.00a
|
10.00±1.732a
|
8.00±2.645a
|
7.33±1.154ab
|
|
17.00
|
8.00±1.00bc
|
8.33±0.577b
|
7.33±0.577a
|
7.33±1.154b
|
5.33±1.154ab
|
5.66±0.577bc
|
|
34.00
|
9.33±0.577b
|
11.00±1.00a
|
7.66±1.527a
|
10.66±1.527a
|
4.66±0.577ab
|
8.00±1.00a
|
|
51.00
|
6.33±0.577d
|
6.66±0.577bc
|
6.00±1.00ab
|
6.33±0.577b
|
5.00±2.00ab
|
4.66±0.577c
|
|
68.00
|
7.00±1.00cd
|
5.66±2.5c
|
4.33±1.154b
|
5.00±1.00b
|
3.66±1.527b
|
4.66±1.527c
|
|
LSD( p ≤ 0.05)
|
1.409
|
2.348
|
1.992
|
2.301
|
3.151
|
1.878
|
|
Tuber weight (g)
|
|
00.00
|
17.33±0.702b
|
20.1±1.212b
|
16.53±0.680a
|
18.76±0.251b
|
16.53±0.472a
|
16.43±0.450b
|
|
17.00
|
18.7±0.20b
|
19.93±0.602b
|
14.85±0.259b
|
17.06±0.115c
|
11.9±0.30c
|
14.3±1.053c
|
|
34.00
|
16.7±0.80b
|
18.9±0.529b
|
14.03±0.550bc
|
16.9±1.044c
|
14.3±0.793b
|
18.4±0.321a
|
|
51.00
|
21.56±2.315a
|
25.7±2.06a
|
17.5±0.854a
|
20.47±0.296a
|
17.13±0.115a
|
12.4±1.250d
|
|
68.00
|
14.03±0.896c
|
15.86±0.709c
|
13.73±0.378c
|
16.00±1.389c
|
11.63±0.568c
|
17.53±0.351ab
|
|
LSD( p ≤ 0.05)
|
2.204
|
2.135
|
1.063
|
1.451
|
0.9204
|
1.433
|
Data are represented as mean ± Standard deviation from three replicates.
Means sharing the same letters, within a column for a parameter, do not differ significant at p ≤ 0.05
DISCUSSION
This study demonstrated that salt stress caused a significant reduction in the growth of callus in vitro as well as potato tuber number, tuber weight, tuber protein, tuber calcium, and tuber iron in vivo. Potato cultivars also differed significantly in response to salinity. However, in the current study the application of ZnO-NPs helped mitigate the adverse effects of salt stress in potato cultivars. Salinity affects plant growth through osmotic stress and mineral ion toxicity (Ghosh et al., 2001; Munns and Tester 2008). An increase in salt concentration in the rhizosphere soil disturbs the osmotic balance resulting in ‘physiological/secondary drought’, which restricts plant water uptake (Farooq et al., 2017). In this study, salt stress reduced the fresh and dry weight of callus. However, application of ZnO-NPs enhanced the callus fresh and dry weight of potato cultivars as ZnO-NPs are absorbed by plants to a greater extent and have high bioavailability owing to smaller particle sizes and low water solubility (Prasad et al., 2012), which enhance the chlorophyll contents, photosynthesis, and biomass production (García-López et al., 2019; Noohpisheh et al., 2020). Application of ZnO-NPs improved the tuber protein content under salt stress condition. Adequate Zn modulates the enzymes involved in the protein biosynthesis and thus enhance the protein content (Zhang et al., 2017; Rehman et al., 2018 b). Tuber protein contents were higher in salt-stressed plants due to lower tuber weight. There was great genotypic variation among the potato cultivars regarding protein content as cv. Spunta had the highest protein content under normal and salt stress conditions, irrespective of ZnO-NPs application.
Salt stress reduced the calcium and iron concentrations in tubers of all cultivars as salinity causes nutritional imbalances owing to decreased nutrient uptake and transport to the shoot. In this regard, Mirvat et al., (2006) reported that salt stress caused significant Zn imbalances in plant cells, which disturbs the normal plant metabolism. The application of ZnO-NPs helped in improving the ionic homeostasis and enhanced the calcium and iron uptake (Hezareh et al., 2020). The ZnO-NPs application enhanced the Zn availability which helped in better root growth and nutrient uptake and translocation (Rehman et al., 2019). Moreover, the application of Zn improves the calcium accumulation in roots (Rehman et al., 2019) and edible plant parts (Rehman et al., 2018 d) as was observed in this study. Zinc application caused significant improvement in calcium and iron uptake under salt stress by improving the structural integrity of root cell membranes and reducing the excessive uptake of the Na+ under salt stress (Aktas et al., 2006; Noohpisheh et al., 2020).
Salt stress drastically reduced the yield and yield contributing traits of potato cultivars. The application of ZnO-NPs substantially improved the number of tubers per plant and potato tuber weight. This improvement was possible due to the involvement of Zn in carbohydrate metabolism, IAA metabolism, and ribosomal function (Rehman et al., 2018a). Furthermore, an increase in tuber weight and number with ZnO-NPs was the possible outcome of improved photosynthesis, plant growth, and nutrient uptake (Rehman et al., 2018c).
The use of nanodevices and nanomaterials offers an attractive option of micronutrient application in plants (Usman et al., 2020) including Zn. Zinc as nanomaterial may help improve plant growth and nutrient acquisition, under less than optimum conditions, than the traditional approaches as cited by Reynolds (2002) in accordance with the present study. The use of ZnO NPs may also help cope with the salinity-induced oxidative stress (Sheykh et al., 2009).
Conclusion: Salt stress significantly reduced the potato growth and yield; however, tuber protein concentration was higher at a high salinity level. Application of ZnO-NPs proved effective in ameliorating the salinity induced harmful effect on yield and quality of potato. The tested potato cultivars differed significantly for their response to salt stress. In this regard, the cultivar Spunta performed better under normal and salt stress conditions.
Acknowledgement: This project was funded by the Deanship of Scientific Research (DSR), King Abdulaziz University, under grant no. G-115-253-1439. The authors, therefore, acknowledge with thanks DSR for technical and financial support. The authors also thank Prof. Fouad H. Ali, SCU, Egypt for his constant advice on this research
Conflict of Interest statement: The authors declare that they have no conflict of interest. We will be responsible for any kind of conflict.
Author Contributions Statement: E. S. Alghamdi conceived the research, designed the figures and tables and participated in the discussion results, reviewed several draft of the manuscript, read and approved the manuscript; M. Farooq performed the experiment analyzed data, statistical analysis, wrote the manuscript and made language editing; EMR Metwali performed the experiments, planned the whole work, conducted provided lab facilities, helped in write up, statistical analysis and submitted the MS.
REFERENCES
- Aghaei, K., A.A. Ehsanpour, and S. Komatsu (2009). Potato responds to salt stress by increased activity of antioxidant enzymes. J. Integr. Plant Biol. 51:1095–1103.
- Aghaei, K., A.A. Ehsanpour, and S. Komatsu (2008). Proteome analysis of potato under salt stress. J. Proteom. Res. 7: 4858-4868.
- Ahmed, H.A.A., N.K. Şahin, G. Akdoğan, C. Yaman, D. Köm and S. Uranbey (2020). Variability in salinity stress tolerance of potato (Solanum tuberosum) varieties using in vitro screening. Ciência e Agrotecnologia, 44., e004220. Epub July 15, 2020.https://dx.doi.org/10.1590/1413-7054202044004220
- Akhtar, S.S., M.N. Anderson, and F. Liu (2015). Biochar mitigates salinity stress in potato. J. Agron. Crop. Sci. 201:368–378.
- Aktas, H., K. Mabak, L. Osturk, and S. Cakmak (2006). The effect of zinc on the growth and shoot concentrations of sodium and potassium in pepper plants under salinity stress. Turk. J. Agric. For. 30:407–412.
- AOAC (1995). Official methods of analysis 16th Association of Official Analytical Chemists. Washington DC, USA.
- Batelli, G., Massarelli, I., Van Oosten, M., Nurcato, R., Vannini, C., Raimondi, G., Leone, A., Zhu, J.K., Maggio, A. and S. Grillo (2012). Asg1 is a stress-inducible gene which increases stomatal resistance in salt stressed potato. J. Plant Physiol. 169:1849-1857.
- Bündig, C., T.H. Vu, P. Meise, S. Seddig, A. Schum, and T. Winkelmann (2017). Variability in osmotic stress tolerance of starch potato genotypes (Solanum tuberosum ) as revealed by an in vitro screening: role of proline, osmotic adjustment and drought response in pot trials. J. Agron. Crop Sci. 203:206-218.
- Chapman, H.D., and P.F. Pratt (1961). Methods of Analysis for Soils, Plants and Waters. University of California, Riverside, CA, USA.
- Duncan, D.B. (1955). Multiple range and multiple F tests. Biometrics 11:1–42.
- Faran, M., M. Farooq, A. Rehman, A. Nawaz, M. K. Saleem, N. Ali, and K.H.M. Siddique (2019). High intrinsic seed Zn concentration improves abiotic stress tolerance in wheat. Plant Soil 437:195–213.
- Farooq, M., N. Gogoi, M. Hussain, S. Barthakur, S. Paul, N. Bharadwaj, H.M. Migdadi, S.S. Alghamdi, and K.H.M. Siddique (2017). Effects, tolerance mechanisms and management of salt stress in grain legumes. Plant Physiol. Biochem. 118:199-217.
- Fidalgo, F., A. Santos, I. Santos, and R. Salema (2004). Effects of long-term salt stress on antioxidant defence systems, leaf water relations and chloroplast ultrastructure of potato plants. Ann. Appl. Biol. 145:185–192.
- Gambuti, A., A. Rinaldi, R. Romano, N. Manzo, and Moio L (2016). Performance of a protein extracted from potatoes for fining of white musts. Food Chem. 190:237–243.
- García-López, J.I., G. Niño-Medina, E. Olivares-Sáenz, R.H. Lira-Saldivar, E.D. Barriga-Castro, R. Vázquez-Alvarado, P.A. Rodríguez-Salinas, and F. Zavala-García (2019). Foliar application of zinc oxide nanoparticles and zinc sulfate boosts the content of bioactive compounds in habanero peppers. Plants 30:8. doi: 10.3390/plants8080254.
- Gąsiorowska, B., A. Płaza, E. Rzążewska, A. Cybulska, and R. Górski (2018). The potato tuber content of microelements as affected by organic fertilisation and production system. Environ. Monitoring Assessment. 190:522.
- Ghosh, S.C., K. Asanuma, A. Kusutani, and M. Toyota (2001). Effect of salt stress on some chemical components, and yield of potato. Soil Sci. Plant Nutr. 47:467–475.
- Gowayed, M.H., H.S. Al-Zahrani, and E.M. Metwali (2017). Improving the salinity tolerance in potato (Solanum tuberosum) by exogenous application of silicon dioxide nanoparticles. Int. J. Agric. Biol. 19:183–194.
- Gu, R., Q. Liu, D. Pei, and X. Jiang (2004). Understanding saline and osmotic tolerance of Populus euphratica suspended cells. Plant Cell Tissue Organ Cult. 78:261–265.
- Hezaveh, T.A., F. Rahmani, H. Alipour and L Pourakbar (2020). Effects of foliar application of ZnO nanoparticles on secondary metabolite and micro-elements of camelina (Camelina sativa) under salinity stress. J. Stress Physiol. Biochem. 16: 54–69.
- Hijmans, R.J. (2003). The effect of climate change on global potato production. Am. J. Potato Res. 80: 271-279.
- Hussein, M.M., and N. Abou-Baker (2008). The contribution of nano-zinc to alleviate salinity stress on cotton plants. Royal Soc. Open Sci. 5:171809
- Jaarsma, R., R.S.M. de Vries, and A.H. de Boer (2013). Effect of salt stress on growth, Na+ accumulation and proline metabolism in potato (Solanum tuberosum) cultivars. PLoS One 8: e60183. 10.1371/journal.pone.0060183
- Jackson, M.L. (1973) Soil Chemical Analysis. Prentice-Hall Inc., Englewood Cliffs, NJ, USA.
- Khatri, P., J.S. Rana, A. Sindhu, and P. Jamdagni (2019). Effect of additives on enhanced in-vitro shoot multiplication and their functional group identification of Chlorophytum borivilianum Sant. Et Fernand. SN Appl. Sci. 1:1105. https://doi.org/10.1007/s42452-019-1118-8
- Kim DH, Gopal J, Sivanesan I (2017) Nanomaterials in plant tissue culture: the disclosed and undisclosed. RSC Adv. 7:.36492-36505.
- Kumlay AM, Ercisli S (2015) Callus induction, shoot proliferation and root regeneration of potato (Solanum tuberosum) stem node and leaf explants under long-day conditions. Biotechnol. Biotechnol. Equip. 29:1075-1084.
- Levy D, Coleman WK, Veilleux, R.E (2013) Adaptation of potato to water shortage: irrigation management and enhancement of tolerance to drought and salinity. Am J Potato Res 90:186-206.
- Liao, X., Z. Su, G. Liu, L. Zotarelli, Y. Cui, and C. Snodgrass (2016). Impact of soil moisture and temperature on potato production using seepage and center pivot irrigation. Agric. Water Manag. 165:.230-236.
- Liu, R., and R. Lal (2015). Potentials of engineered nanoparticles as fertilizers for increasing agronomic productions. Sci. Total Environ. 514:131–139.
- Lutts, S., M. Almansouri, and J.M. Kinet (2004). Salinity and water stress have contrasting effects on the relationship between growth and cell viability during and after stress exposure in durum wheat callus. Plant Sci. 167:9–18.
- Mahmoud, A.W.M., E.A. Abdeldaym, S.M. Abdelaziz, M.B.I. El-Sawy, and S.A. Mottaleb (2020). Synergetic effects of zinc, boron, silicon, and zeolite nanoparticles on confer tolerance in potato plants subjected to salinity. Agronomy 10:19.
- Martínez-Ballesta, M.C., R. Dominguez-Perles, D.A. Moreno, B. Muries, C. Alcaraz-López, E. Bastías, C. García-Viguera, M. Carvajal (2010). Minerals in plant food: effect of agricultural practices and role in human health. A review. Agron. Sustain. Dev. 30:295–309.
- Mirvat, E.G., M.H. Mohamed, and M.M. Tawfik (2006). Effect of phosphorus fertilizer and foliar spraying with zinc on growth, yield and quality of groundnut under reclaimed sandy soils. J. App. Sci. Res. 2:491–496.
- Munns, R., and M. Tester (2008). Mechanisms of salinity tolerance. Ann. Rev. Plant. Biol. 59:651-681.
- Murashige, T., and F. Skoog (1962). A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant 15:473–479.
- Noohpisheh, Z., H. Amiri, A. Mohammadi, and S. Farhadi (2020). Effect of the foliar application of zinc oxide nanoparticles on some biochemical and physiological parameters of Trigonella foenum-graecum under salinity stress. Plant Biosystems https://doi.org/10.1080/11263504.2020.1739160
- Prasad, T., P. Sudhakar, Y. Sreenivasulu, P. Latha, V. Munaswamy, K. Raja-Reddy, T.S. Sreeprasad, P.R. Sajanlal, and T. Pradeep (2012). Effect of nanoscale zinc oxide particles on the germination, growth and yield of peanut. J. Plant Nut. 35:905–927.
- Rehman, A., M. Farooq, M. Asif, and L. Ozturk (2019). Supra‐optimal growth temperature exacerbates adverse effects of low Zn supply in wheat. J. Plant Nutri. Soil Sci. 182:656–666.
- Rehman, A., M. Farooq, M. Naveed, A. Nawaz, and B. Shahzad (2018b). Seed priming of Zn with endophytic bacteria improves the productivity and grain biofortification of bread wheat. Eur. J. Agron. 94:98–107.
- Rehman, A., M. Farooq, M. Naveed, L. Ozturk, and A. Nawaz (2018c). Pseudomonas-aided zinc application improves the productivity and biofortification of bread wheat. Crop Pasture Sci. 69:659–672.
- Rehman, A., M. Farooq, A. Nawaz, A.M. Al‐Sadi, K.S. Al-Hashmi, F. Nadeem, A. Ullah (2018d). Characterizing bread wheat genotypes of Pakistani origin for grain zinc biofortification potential. J. Sci. Food Agric. 98:4824–4836.
- Rehman, A., M. Farooq, L. Ozturk, M. Asif, and K.H. Siddique (2018a). Zinc nutrition in wheat-based cropping systems. Plant Soil 422:283–315.
- Reynolds, G.H. (2002) Forward to the future nanotechnology and regulatory policy. Pacific Res. Inst. 24:1–23.
- Rossi, M., F. Cubadda, L. Dini, M.L. Terranova, F. Aureli, A. Sorbo, and D. Passeri (2014). Scientific basis of nanotechnology, implications for the food sector and future trends. Trends Food Sci. Technol. 40:127–
- Sen, I., and B. Pendam (2014). Determination of heavy metals in five major ingredients of herbal medicines. Int. J. Pharm. Sci. Res. 5:4310–4314.
- Seo, S., S. Karboune, and A. Archelas (2014) Production and characterization of potato patatin–galactose, galactooligosaccharides, and galactan conjugates of great potential as functional ingredients. Food Chem. 158:480–489.
- Sheykh, B.N., G.T.A. Hasanzade, M.M. Baghestani, and B. Zand (2009). Study the effect of zinc foliar application on the quantitative and qualitative yield of grain corn under water stress. Electr. J. Crop Prod. 2:59–74.
- Usman, M., M. Farooq, A. Wakeel, A. Nawaz, S.A. Cheema, H. Rehman, I. Ashraf, and M. Sanaullah (2020). Nanotechnology in agriculture: current status, challenges and future opportunities. Sci. Total Environ. 137778
- Wegener, C.B., G. Jansen, and H.U. Jürgens (2015). Bioactive compounds in potatoes: Accumulation under drought stress conditions. Foods Health Dis. 5:108–116.
- Zhang, D.Q., T.H. Mu, H.N. Sun, J.W. Chen, and M. Zhang (2017). Comparative study of potato protein concentrates extracted using ammonium sulfate and isoelectric precipitation. Int. J. Food Prop. 20:2113–212.
|





