EFFECTS OF DIETARY SUPPLEMENTATION OF SYNBIOTICS ON GROWTH, INTESTINAL BARRIER FUNCTION AND CECAL MICROORGANISMS OF CHERRY VALLEY DUCKS
R. X. Wang, Z. W. Chen, R. Chen, Q. Liu* and S. Zhuang*
College of Animal Science and Technology, Nanjing Agricultural University, Nanjing, 210095, P. R. China
*Corresponding author’s email: zhuangsu@njau.edu.cn; liuayang@njau.edu.cn
ABSTRACT
The present study was set out to explore the effects of diets supplemented with synbiotics, as a possible antibiotic substitute, on growth performance, intestinal barrier function and cecal microorganisms of Cherry Valley ducks. Briefly, a total of 540 Cherry Valley ducks (one-day-old) were randomly categorized into 3 groups for 42 days: control group (basal diets free from the antibiotic), antibiotic group (basal diets supplemented with 40 mg/kg bacitracin zinc), and synbiotic group (basal diets supplemented with 1 000 mg/kg synbiotic). Each group consisted of 6 replicates of 30 ducks each. In synbiotic group, average-day-gain (ADG) throughout the trial period (p<0.05) and average-day-feed-intake (ADFI) during the starter and grower periods (p<0.05) were increased as compared with the control group. Moreover, dietary synbiotics enhanced ileal villus height to crypt depth (V/C), decreased ileal crypt depth compared with the control group (p<0.05). In addition, the synbiotics increased numbers of Intraepithelial lymphocytes (IELs) and goblet cells in the small intestine(p<0.05), and secretory IgA (sIgA) concentration in the duodenum and jejunum (p<0.05) of 42-day-old ducks as compared with the control group. Furthermore, synbiotics supplementation increased the mRNA expression of jejunal mucosal occluding (OCLN) and the population of Bifidobacterium in cecal chyme (p<0.05). In conclusion, diets supplemented with synbiotics could improve growth performance and intestinal morphology, enhance intestinal barrier function, and modulate the cecal microorganisms in Cherry Valley ducks.
Key words: synbiotic, Cherry Valley ducks, growth performance, intestinal barrier function, cecal microorganisms
https://doi.org/10.36899/JAPS.2022.2.0437
Published first online August 13, 2021
INTRODUCTION
Antibiotics have been extensively applied to animals’ feed for decades because of superior growth promoting effect. However, the application of antibiotics caused a series of problems such as increasing bacterial resistance, spreading drug resistant pathogens, endangering the environment and human health (Barton, 2000; Huyghebaer et al., 2011; Allen et al., 2013). In 2006, the European Union has given a lead by issuing a ban on prohibiting antibiotics supplemented to animal feed, as a growth promoter (Liu et al., 2017a). Also, Chinese government has banned the application of antibiotics apart from Chinese herbal medicine in animal feed from July 1, 2020 (Tian et al., 2020). However, the restrict of antibiotic has caused many problems in poultry production, such as reducing feed conversion rate and increasing morbidity (Huyghebaer et al., 2011). Thus, searching for the alternative of antibiotic has received growing interests.
Probiotics are live microorganisms, which could preserve the natural intestinal and have beneficial effects on host animals when delivered via the feed (Fuller, 1989), which can also strengthen intestinal mucosal barrier and enhance intestinal immune responses (Hassanpour et at., 2013). Prebiotics are selective fermented ingredients that change the compositions and activity of the gastrointestinal microflora which are beneficial for animal or human being health. Synbiotic is a kind of biological preparation which contains both probiotics and prebiotics, playing a dual role of probiotics and prebiotics (Collins and Gibson, 1999; Schrezenmeir and Vrese, 2001). Previous studies have demonstrated that dietary supplemented with probiotics (Clostridium butyricum or Bacillus subtilis), prebiotic (xylooligosaccharides or chitooligosaccharides) could promote growth performance, improve intestinal morphology, increase the number of beneficial gut microorganisms and enhance intestinal barrier function of poultry (Maesschalcket al., 2015; Zhuang et al., 2015; Ma et al., 2018; Li et al., 2019). Moreover, studies have shown that synbiotic used as an antibiotic substitute, could exert positive effect on antioxidant capacity, gut microorganisms, intestinal morphology, immune function, and growth performance (Min et al., 2016; Chen et al., 2018), and appeared to have more beneficial effects than either probiotic or prebiotic alone supplemented in the diets of broilers and pigs (Awad et al., 2009; Tanner et al., 2016; kridtayopas et al., 2019). However, rare research has been performed on the synergetic effects of probiotics (Clostridium butyricum, Bacillus subtilis) and prebiotics (xylooligosaccharides and chitooligosaccharides) in duck production. Consequently, this research set out to investigate the effect of dietary supplemented with the synbiotic on growth performance, gut mucosal barrier function and cecal microorganisms of Cherry Valley ducks.
MATERIALS AND METHODS
Procedures related to animals were in accordance with requirements of Animal Care and Use Committee of Nanjing Agricultural University.
Experimental design: A total of 540 one-day-old Cherry Valley ducks were randomly assigned into 3 groups, with 6 replicate pens per group, and 30 ducks per pen. Ducks in the control group were fed with basal diets free from antibiotics, while ducks in the experimental groups were fed with basal diets inclusion of bacitracin zinc (40 mg/kg, Antibiotic group) or synbiotics (1 000 mg/kg, Synbiotic group), respectively. One gram of the synbiotic consisted of 100 mg xylooligosaccharides, 50 mg chitooligosaccharides, 3×1010 CFU of Bacillus subtilis, and 2×109 CFU of Clostridium butyricum. The experiment period lasted for 42 days, and divided into starter and grower period, with day 15 being the demarcation. The ingredient compositions and nutrient contents of basal diet is presented in Table 1. Ducks were offered diets and water ad libitum and arranged on a plastic slatted floor in a house, where the temperature was remained at 32-33℃ in the first three days and then lowered by 2-3℃ weekly to a final temperature of 25℃.
Table 1. Composition and nutrient level of basal diet (air drying basis).
|
Composition
|
1-14 days
|
15-42 days
|
|
Ingredient’s composition/%
|
|
Corn
|
50.00
|
48.50
|
|
Rice bran
|
6.40
|
7.30
|
|
Wheat middlings
|
7.50
|
14.65
|
|
Soybean meal
|
24.00
|
14.00
|
|
Corn gluten meal
|
5.00
|
8.00
|
|
Animal fat
|
2.00
|
2.50
|
|
Lysine 55%
|
0.15
|
0.20
|
|
Methionine 99%
|
0.15
|
0.15
|
|
Limestone
|
1.50
|
1.40
|
|
Calcium Hydrophosphate
|
1.50
|
1.50
|
|
Sodium chloride
|
0.30
|
0.30
|
|
Premix1
|
1.00
|
1.00
|
|
Attapulgite
|
0.50
|
0.50
|
|
Calculated Nutrient composition
|
|
Apparent metabolizable energy (MJ/kg)
|
12.16
|
12.54
|
|
Crude protein (%)
|
19.79
|
17.44
|
|
Calcium (%)
|
0.97
|
0.91
|
|
Available phosphorus (%)
|
0.37
|
0.36
|
|
Lysine (%)
|
0.99
|
0.80
|
|
Methionine (%)
|
0.45
|
0.44
|
1Premix provided the followings for per kilogram of diet: vitamin A, 10 000 IU; vitamin D, 2 000 IU; vitamin E, 20 IU; vitamin K, 0.5 mg; vitamin B12, 0.04 mg; nicotinic acid, 60 mg; D-pantothenie, 11 mg; cholinechloride, 600 mg; riboflavin, 4.0 mg; pyridoxine, 2.5 mg; folic acid, 0.6 mg; biotin, 0.2 mg; Mn, 80 mg; Fe, 80 mg; Zn, 60 mg; Cu, 8 mg; thiamine, 3 mg; I, 0.4 mg; Se, 0.2 mg.
Growth performance: Feed consumption was documented every day, body weight was recorded on a replicate basis at day 14 and 42. Based on these given data, average-day-feed-intake (ADFI), average-day-gain (ADG) and feed conversion ratio (F: G) were obtained.
Sample’s collection: One male duck per replicate (close to the replicate average body weight) was picked and weighed after feed deprivation for 12 h at 42 days of age. The ducks were euthanized, and the intestinal tissues from the different segments of small intestine (approximately 2.0 cm long, each) were collected, flushed with ice-cold phosphate-buffered saline, and fixed in 10% formaldehyde solution for later determination. The duodenum, jejunum and ileum were opened along the long axis, and the mucosa was scraped gently using a sterile glass slide and deposited at -80℃ until analysis. Cecal chyme was obtained aseptically and deposited at -20℃ for further assessment of microorganisms and volatile fatty acids contents.
Intestinal morphology: The fixed intestinal segments were embedded in paraffin and sliced according to the procedures of Xing et al. (2017). The slices were stained using hematoxylin and eosin (H&E), Ten randomly selected intact villi from each slice were picked. The villus height and crypt depth in each were visually observed and measured under a light microscope (Nikon Corporation, Tokyo, Japan).
Intraepithelial lymphocytes and goblet cells: The prepared intestinal tissues were fixed, dehydrated, and embedded in paraffin. To observe the distribution of intraepithelial lymphocytes (IELs) and goblet cells, some sections were stained using H&E to observe the distribution of goblet cells and others with Periodic Acid Schiff (PAS) for IELs. All sections were observed under a light microscope (Olympus Optical Co., Japan). IELs and goblet cells of the three different intestinal segments were counted at five separate fields of intestinal intact villi and represented as cell numbers in 100 columnar epithelial cells.
Secretory IgA content: Double-antibody sandwich ELISA method was used to measure secretory IgA (sIgA) contents in duodenal, jejunal and ileal mucosa, using the commercial kits which were purchased from Nanjing Jiancheng Bioengineering Institute, China.
Tight junction proteins related genes expression in gut mucosa: Total RNA in the small intestinal mucosa was isolated using RNAiso reagent. The quantification of RNA was determined by a NanoDropND-2000 spectrophotometer according to OD260/280 reading. The reverse transcription kit (PrimeScript™ RT Master Mix (Perfect Real Time), 20 µL reaction system) was used to reverse-transcribe the isolated RNA into complementary DNA (cDNA). Subsequently, a TB Green Premix Ex Taq™ kit was used for real-time PCR process following the manufacturer’s instruction, which was done by the ABI StepOnePlusTM Real-Time PCR system (Applied Biosystems). The amplification procedure was as following: pre-denaturation at 95℃ for 5 mins, the cyclic reaction at 60℃ for 34 s followed by 40 cycles of 95℃ for 10 s, and melting stage of 95℃ for 15 s, 60℃ for 1 min, 95℃ for 15 s. The primers were synthesized by Sangon Biotechnology (Shanghai, China), and the sequences for zonulaoccludens-1 (ZO-1), claudin-1 (CLDN-1), occludin (OCLN), and β-actin (reference gene) were given in Table 2, which were referred to Wang et al. (2019). The relative expressions of these genes were analyzed by the method of 2−ΔΔCT (Livak and Schmittgen, 2001). All results of gene expression are reported as the fold difference between the experimental and control groups. The above kits were provided by Takara Biotechnology and purchased from Shanghai Haojia Technology Development Co., Ltd, China.
Absolute quantification of cecal microorganism: The population of Escherichia coli, Lactobacillus and Bifidobacterium in the cecal chyme were determined by real-time PCR. The cecal chyme from each sample was diluted with phosphate-buffered saline (1:4) and the total DNA were extracted using the hexadecyl trimethyl ammonium bromide (CTAB) method. The quantification of the isolated total DNA was determined by a NanoDropND-2000 spectrophotometer (NanoDrop Technologies, Wilmington, DE, USA). The qualitied supernatants were transferred to sterile tubes and deposited at -20°C until real-time PCR process. The primer sequences are listed in Table 2 based on the studies of Du et al. (2015) and Xing et al. (2015). Based on the 16S RNA sequences of Escherichia coli, Lactobacillus and Bifidobacterium in GenBank, multiple continuous standard templates were used to establish the standard curves of these bacteria. The expression of the three bacteria was analyzed by standard curve method.
Table 2. Primers sequences used for real-time PCR.
|
Genes1
|
Primer sequence
|
Product size (bp)
|
|
β-actin
|
F: TTGGTTTGTCAAGCAAGCGCGG
|
100
|
|
R: CCCCCACATACTGGCACTTT
|
|
CLDN-1
|
F: TCCATGCATGTGCTGTTGGC
|
145
|
|
R: CCTGCTGCAGTTGCAGTGTT
|
|
ZO-1
|
F: TGTAGCCACAGCAAGAGGTG
|
159
|
|
R: CTGGAATGGCTCCTTGTGGT
|
|
OCLN
|
F: CTCTGCTTCCTGGCCCAGTT
|
214
|
|
R: AGACGATGGA GGCGA TGAGC
|
|
Escherichia coli
|
F: GTTAATACCTTTGCTCATTGA
|
340
|
|
R: ACCAGGGTATCTAATCCTGT
|
|
Lactobacillus
|
F: TACATCCCAACTCCAGAACG
|
91
|
|
R: AAGCAACAGTACCACGACC
|
|
Bifidobacterium
|
F: CTCCTGGAAACGGGTGG
|
550
|
|
R: GGTGTTCTTCCCGATATCTACA
|
1 ZO-1, zonula occludens-1; OCLN, occluding; CLDN-1, claudin-1.
Volatile fatty acids (VFAs) contents of cecal chyme: The contents of VFAs in cecal chyme were determined by gas chromatography. One gram of sterile cecal contents were weighed and put into a sterile centrifuge tube. Ultrapure water (1mL) was added and vortex oscillation was performed for 5 min. Then, the centrifugation was done at 10 000×g for 5 min, and supernatants were taken to a fresh sterile tube. Crotonic acid-metaphosphoric acid solution (200 μL) was added per milliliter, then stored at -20℃ overnight after mixing. After thawing, the centrifugation was done at 12 000×g for 10 min. The supernatants were taken and filtered through a 0.22 μm aqueous phase needle filter, and then injected into Shimadzu gas chromatograph (GC 2014AFsc).
Statistical analysis: The data were analyzed by one-way ANOVA using SPSS statistics 25.0. The differences among groups were compared using Duncan’s multiple-range tests and considered significant at p<0.05. Data were represented as values of means and their total standard errors (SEM).
RESULTS
Growth performance: As indicated in Table 3, dietary inclusion of synbiotic or antibiotic increased ADG (p<0.05), whereas F:G ratio and ADFI were not significantly changed during the starter period (p>0.05). Synbiotic supplemented to diet significantly improved ADG (p<0.05) and ADFI (p<0.05) during the grower period and whole period. Additionally, the antibiotic group obtained similar ADG (p>0.05), and a significantly lower ADFI (p<0.05) compared with the control group during the grower period and whole periods. Dietary antibiotic significantly reduced the F:G ratio (p<0.05) during the whole period.
Table 3.Effect of synbiotics on growth performance of Cherry Valley ducks.
|
Items1
|
Control
|
Antibiotic
|
Synbiotic
|
SEM2
|
p-value
|
|
Starter period
|
|
ADFI (g/d)
|
40.51
|
42.57
|
43.51
|
0.54
|
0.056
|
|
ADG (g/d)
|
35.60c
|
39.02b
|
41.74a
|
0.69
|
<0.001
|
|
F: G
|
1.14
|
1.09
|
1.04
|
0.02
|
0.101
|
|
Grower period
|
|
ADFI (g/d)
|
145.03b
|
134.75c
|
150.89a
|
1.89
|
<0.001
|
|
ADG (g/d)
|
68.40b
|
66.58b
|
71.89a
|
0.77
|
0.008
|
|
F: G
|
2.12
|
2.03
|
2.10
|
0.02
|
0.132
|
|
Whole period
|
|
ADFI (g/d)
|
110.19b
|
104.00c
|
115.10a
|
1.30
|
<0.001
|
|
ADG (g/d)
|
57.47b
|
57.41b
|
61.84a
|
0.62
|
<0.001
|
|
F: G
|
1.92a
|
1.81b
|
1.86ab
|
0.02
|
0.025
|
a-c Means within a row with different superscripts differ significantly at p<0.05. The same as below.
1 ADG, average daily gain; ADFI, average daily feed intake; F: G, feed conversation ratio.
2 SEM, total standard error of means (n = 6). The same as below.
Intestinal morphology: The results of intestinal morphology are presented in Fig.1. Compared with the control group, dietary inclusion of synbiotic or antibiotics improved ileal V/C ratio (p<0.05). Dietary synbiotic reduced ileal crypt depth as compared with the control group (p<0.05), but there were no differences between the antibiotic and synbiotic groups (p>0.05). The jejunal crypt depth (p=0.058) had an increasing tendency in the antibiotic and synbiotic groups as compared with the control group.

Fig. 1. Effects of synbiotic on intestinal morphology of Cherry Valley ducks (40×).
Values with different letters (a, b) for each group indicate significant differences at p<0.05.
IELs, goblet cells and secretory immunoglobulin A: As shown in Fig.2, the synbiotics significantly enhanced IELs and goblet cell in the small intestine as compared with the control group (p<0.05). Simultaneously, dietary antibiotic enhanced the quantity of ileal goblet cells and IELs (p<0.05). Antibiotic and synbiotic could improve or have a tendency to improve the levels of sIgA (p<0.05 or p=0.058) in different intestinal sections (Table 4).
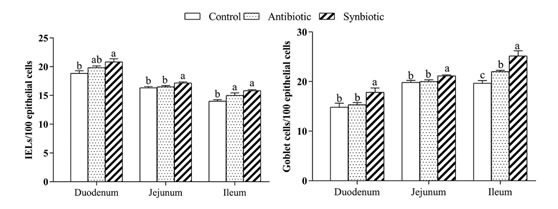
Fig. 2.Effects of synbiotics onIELs, goblet cells in the intestinal epithelium of Cherry Valley ducks.
Values with different letters (a, b, c) for each group indicate significant differences at p<0.05.
Table 4. Effects of synbiotics onsIgA contents in the intestinal mucosa of Cherry Valley ducks (µg/mg prot)1.
|
Items
|
Control
|
Antibiotic
|
Synbiotic
|
SEM
|
p-value
|
|
Duodenum
|
5.72b
|
6.54a
|
6.89a
|
0.17
|
0.006
|
|
Jejunum
|
5.14c
|
6.42a
|
5.86b
|
0.15
|
< 0.001
|
|
Ileum
|
7.72
|
8.60
|
7.96
|
0.16
|
0.058
|
1 SIgA, secretory immunoglobulin A.
The expression of tight junction protein related genes in intestinal mucosa: As exhibited in Table 5, the mRNA expression of jejunal OCLN in the synbiotic group was increased as compared with the other two groups (p<0.05). The duodenal ZO-1 mRNA expression levels in antibiotic group were higher than that in the control group (p<0.05), whereas no difference was observed between the synbiotic and antibiotic groups (p>0.05). No significant differences were found in the mRNA expression of OCLN and CLDN-1 in duodenum, CLDN-1 and ZO-1 in jejunum and tight junction proteins in ileum (p>0.05).
Table 5. Effects of synbiotics onmRNA expression of tight junction protein related-genes in the intestinal mucosa of Cherry Valley ducks.
|
Items1
|
Control
|
Antibiotic
|
Synbiotic
|
SEM
|
p-value
|
|
Duodenum
|
|
ZO-1
|
1.00b
|
2.16a
|
1.56ab
|
0.16
|
0.004
|
|
OCLN
|
1.00
|
1.11
|
1.54
|
0.12
|
0.185
|
|
CLDN-1
|
1.00
|
1.71
|
1.25
|
0.14
|
0.089
|
|
Jejunum
|
|
ZO-1
|
1.00
|
0.89
|
1.13
|
0.12
|
0.750
|
|
OCLN
|
1.00b
|
1.11b
|
2.40a
|
0.19
|
< 0.001
|
|
CLDN-1
|
1.00
|
1.15
|
1.54
|
0.16
|
0.376
|
|
Ileum
|
|
ZO-1
|
1.00
|
0.88
|
1.05
|
0.12
|
0.858
|
|
OCLN
|
1.00
|
1.22
|
1.09
|
0.16
|
0.865
|
|
CLDN-1
|
1.00
|
1.28
|
1.10
|
0.14
|
0.741
|
1 ZO-1, zonula occludens-1; OCLN, occluding; CLDN-1, claudin-1.
Microorganisms’ populations in the cecal chyme: Compared with the control group, the quantity of Bifidobacteria in the synbiotic or antibiotic group were significantly increased (p<0.05). No differences were found in copy numbers of Lactobacilli and Escherichia coli among three groups (p>0.05, Table 6).
Table 6.Effects of synbiotics on cecal microorganisms of Cherry Valley ducks (log10 (copy)/g chyme).
|
Items
|
Control
|
Antibiotic
|
Synbiotic
|
SEM
|
p-value
|
|
Escherichia coli
|
6.60
|
5.95
|
5.91
|
0.17
|
0.161
|
|
Lactobacillus
|
4.98
|
5.21
|
5.27
|
0.08
|
0.269
|
|
Bifidobacterium
|
10.15b
|
12.02a
|
12.10a
|
0.32
|
0.010
|
Volatile fatty acids contents in cecal chyme: The contents of volatile fatty acids (VFA) are given in Table 7. No differences of VFAs contents were found among three groups (p>0.05).
Table 7.Effects of synbiotics on the contents of VFAs in cecal chyme of Cherry Valley ducks (µmol/g).
|
Items
|
Control
|
Antibiotic
|
Synbiotic
|
SEM
|
p-value
|
|
Acetic acid
|
62.07
|
67.90
|
65.93
|
3.05
|
0.754
|
|
Propionic acid
|
24.35
|
31.96
|
29.02
|
2.34
|
0.432
|
|
Butyric acid
|
7.38
|
7.98
|
8.18
|
0.81
|
0.925
|
|
Isobutyric acid
|
0.73
|
0.52
|
0.36
|
0.08
|
0.254
|
|
Valeric acid
|
1.33
|
1.01
|
0.73
|
0.13
|
0.191
|
|
Isovaleric acid
|
0.84
|
0.46
|
0.48
|
0.10
|
0.211
|
|
Total volatile fatty acids
|
96.70
|
109.83
|
106.90
|
5.12
|
0.576
|
DISCUSSION
It was found in this study that dietary supplementation of the synbiotics, consisted of clostridium butyricum, Bacillus subtilis, xylooligosaccharides and chitooligosaccharides, increased the ADG throughout the period of experiment, and improved the ADFI during the grower and whole period of Cherry Valley ducks. These results were consistent with earlier studies on broilers, which discovered that synbiotics supplemented to the diets could enhance growth performance and suggested that was associated with regulation of gastrointestinal integrity and barrier function and promotion in digesting and absorbing nutrients (Cheng et al., 2017; Chen et al., 2018; Kridtayopas et al., 2019). Furthermore, values of these indexes in the synbiotic group were superior to those in the antibiotic group, except for F:G ratio during grower and entire periods in the present study, which suggested that the synbitic might be used as a substitute for antibiotic in meat duck production and also indicated that the effects of synbiotic on performance would be better than antibiotics under the condition of relatively good hygiene, such as on a plastic slatted floor in this study. Villus height, depth of crypt and V/C ratio are usually used to assess the digestion and absorption of nutrients in intestinum tenue, as well as are regarded as indicators of intestinal health (Xiong et al., 2015; Wen et al. 2019). Here, dietary supplementation with synbiotics lowered the ileal crypt depth, promoted V/C ratio. Awad et al. (2009) demonstrated that either synbiotics or probiotics could enhance V/C ratio and villus height in the intestinum tenue of broilers and suggested that feeding of direct-fed microbials increased epithelial cell turnover. Several studies have demonstrated that probiotics supplemented to poultry’s diets could promote growth performance, improve intestinal morphology, and enhance nutrients absorption and digestion (Jayaraman et al., 2013; Zhuang et al., 2015; Guo et al., 2016; Ma et al., 2018). Therefore, the incremental levels of profitable microbes in the gut might be the reasons for improving intestinal morphology and promoting growth performance of Cherry Valley ducks.
The intestinal barrier is a continuous single layer composed of closely arranged intestinal epithelial cells, which can separate the intestinal cavity from the internal environment and prevent the invasion of pathogenic antigens (Romero et al., 2015). The physical barrier and immune barrier are the main parts of the intestinal barrier (Liu et al., 2009). We evaluated the immune barrier by quantifying immune-related cells (IELs, goblet cells) and the concentration of sIgA in this study. IELs are firstly exposed to antigens and microorganisms in the mucosal immune system, which can secrete cytokines to mediate mucosal immune response (Patterson and Waston, 2018). Goblet cells can secrete mucus to lubricate and protect the intestinal mucosal epithelium, the formed mucus layer contains a large amount of secreted immunoglobulin, which is the most important antibody in digestive juice. SIgA is presented at mucosal surfaces and bound to pathogenic organisms, which is the most effective antibody to provide passive immune protection by blocking the binding to mucosa cells (Tohid et al., 2010). Our results indicated that dietary supplemented with synbiotics enhanced the population of IELs and goblet cells in the small intestine, and increased sIgA concentrations in duodenal and jejunal mucosa as well as promoted the intestinal immune barrier function of ducks. This was consistent with previous studies that conducted on dietary synbiotic supplementation to broilers diet (Calik et al., 2017; Liu et al., 2017b; Chen et al., 2018). Many reports indicated that probiotics could increase small intestinal sIgA level and lymphocyte populations and improve the immune status of broilers (Bai et al., 2018; Ekim et al., 2020). It has been reported that probiotics via feed entered the intestinal tract and diffused into gut-associated lymphoid tissues, such as Payl’s node and mesenteric lymph nodes (Duc et al., 2003), which could stimulate phagocytes to be highly activated, enhance the antigen presentation and antibacterial defense capabilities (Min et al., 2016). Combining the results of IELs, goblet cells and sIgA, it was suggested that dietary supplementation of synbiotics could maintain the intestinal health by improving the intestinal immune barrier of Cherry Valley ducks.
The epithelial cells are firmly linked by tight junctions, forming the mechanical barrier that prevents pathogens from penetration (Magalhaes et al., 2007). Tight junctions (TJs) are consisted of plenty of functional and structural proteins including claudin (CLDN), zonula accludens (ZO), and occluding (OCLN) (Wang et al., 2017). The reduction of CLDN-1, OCLN and ZO-1 expression in intestinal tissue can be translated to the failure of intestinal barrier function and the increase of intestinal permeability (Anderson, 2001; Gadde et al., 2017). Sheng et al., (2020) stated that a kind of synbiotic, containing B. infants and XOS, efficaciously upregulated the expression levels of colonic OCLN, ZO-1 and CLDN-1 in DSS-induced mice. Herein, mRNA expression of OCLN in jejunum mucosa was increased in the synbiotic group and ZO-1 in duodenal mucosa was increased in the antibiotic group. Dietary supplementation with probiotic (Clostridium butyricum and Bacillus subtilis) or prebiotic (xylooligosaccharides and chitooligosaccharides) alone could upregulate the expression of TJ proteins (Huang et al., 2019; Li et al., 2019; Musa et al., 2019; Yin et al., 2019). It was suggested that comparatively better behavior of synbiotic supplementation may be correlated with the additive efficacy of prebiotics and probiotics (Sheng et al., 2020).
The probiotics in the synbiotics not only increase the survive of probiotic microorganisms but also stimulate the activity of the host’s endogenous bacteria in the gastrointestinal tract (Vandenplas et al., 2013). While prebiotics were selectively fermented by profitable microbes in the hindgut, provided nourishment for existing flora (Roberfroid, 2007). Dibaji et al. (2014) indicated that dietary synbiotic increased populations of lactic acid bacteria and reduced Escherichia coli numbers of broilers. The synbiotic used in the current study consisted of Clostridium butyricum, Bacillus subtilis, xylooligosaccharides and chitooligosaccharides, which have been demonstrated having beneficial roles in modulating intestinal microbiota and improving immune function, thereby promoting growth performance of poultry (Maesschalcket al., 2015; Zhuang et al., 2015; Ma et al., 2018; Li et al., 2019). In the current study, dietary antibiotic (bacitracin zinc) or synbiotic led to a favorable regulation of the cecal microflora, with increases in copy numbers of Bifidobacteria and Lactobacilli, and a reduction in populations of Escherichia coli. The results were consistent with the earlier research by Li et al. (2009), who stated that synbiotics consisted of Lactobacillus, B.cereus and astragalus polysaccharides could increase the number of beneficial microbes (Bifidobacterium and Lactobacilli) and decrease populations of hazardous microorganisms (Escherichia coli) of chicks. It was demonstrated that dietary probiotics could be colonized in the epithelial wall of the intestine, which would competitively exclude the attachment of pathogens and promote the proliferation of probiotic (Jin et al., 1996; Mookiah et al., 2013). These microorganisms in hindgut can ferment the undigested feed components to produce VFAs, such as acetic acids, propionic acids, butyric acids. It is widely known that VFAs are the main fermentation product of intestinal microorganisms, which can lower pH values in the intestine, provide essential energy for intestinal epithelium, suppress the harmful bacteria, and stabilize the intestinal microflora indirectly (Pryde et al., 2002; Duncan et al., 2004; Louis et al., 2004; Le Roy et al., 2015; Yang et al., 2019). In this study, the contents of total VFA and the main VFAs (acetic, propanoic and butyric acid) in cecal chyme were numerically higher in antibiotic and synbiotic group than in the control group, which might be responded to the greater numbers of Lactobacillus and Bifidobacterium, and reduced copies of Escherichia coli. Based on the result, we can deduce that the diets inclusion of synbiotics could improve the gastrointestinal health by promoting the cecal microorganisms of Cherry Valley ducks.
Conclusions: The results of our study suggested that dietary supplementation of synbiotics consisted of chitoligosaccharides, xylooligosaccharide, Clostridium butyricum and Bacillus subtilis, could be used as an antibiotic substitute to improve the growth performance, stimulate the intestinal morphology, enhance the intestinal barrier function, and increase the population of beneficial microflora of Cherry Valley ducks.
Acknowledgements: This research was funded by the Department of Science and Technology of Jiangsu Province, P, R. China (CX (18)1004).
Authors’ contributions: R. X. Wang, S. Zhuang and Q. Liu participated in the design of this research, and they conducted the statistical analysis. R. X. Wang, Z. W. Chen and R. Chen carried out the study and collected the data. The first version of the manuscript was written by R. X. Wang. S. Zhuang and Q. Liu revised this manuscript. The content of submitted manuscript has been read and agreed by all authors.
REFERENCES
- Allen, H. K., U. Y. Levine, T. Looft, M. Bandrick and T. A. Casey (2013). Treatment, promotion, commotion: Antibiotic alternatives in food-producing animals. Trends Microbiol. 21(3): 114-119.
- Anderson, J. M. (2001). Molecular structure of tight junctions and their role in epithelial transport. News Physiol Sci. 16: 126-130.
- Awad, W. A., K. Ghareeb, S. Abdel-Raheem and J. Böhm (2009). Effects of dietary inclusion of probiotic and synbiotic on growth performance, organ weights, and intestinal histomorphology of broiler chickens. Poult Sci. 88(1): 49-56.
- Bai, K., C. Feng, L. Jiang, L. Zhang, J. Zhang, L. Zhang and T. Wang (2018). Dietary effects of Bacillus subtilis fmbj on growth performance, small intestinal morphology, and its antioxidant capacity of broilers. Poult Sci. 97(7): 2312-2321.
- Barton, M. D. (2000). Antibiotic use in animal feed and its impact on human health. Nutr Res Rev. 13(2): 279-299.
- Calik A., A. Ceylan, B. Ekim, S. G. Adabi, F. Dilber, A. G. Bayraktaroglu, T. Tekinay, D. Özen and P. Sacakli (2017). The effect of intra-amniotic and posthatch dietary synbiotic administration on the performance, intestinal histomorphology, cecal microbial population, and short-chain fatty acid composition of broiler chickens. Poult Sci. 96(1): 169-183.
- Chen, Y., C. Wen and Y. Zhou (2018). Dietary synbiotic incorporation as an alternative to antibiotic improves growth performance, intestinal morphology, immunity and antioxidant capacity of broilers. J Sci Food Agric. 98(9): 3343-3350.
- Cheng, Y., Y. Chen, X. Li, W. Yang, C. Wen, Y. Kang, A. Wang and Y. Zhou (2017). Effects of synbiotic supplementation on growth performance, carcass characteristics, meat quality and muscular antioxidant capacity and mineral contents in broilers. J Sci Food Agric. 97(11): 3699-3705.
- Collins, M. D. and G. R. Gibson (1999). Probiotics, prebiotics, and synbiotics: Approaches for modulating the microbial ecology of the gut. Am J Clin Nutr. 69(5): 1052-1057.
- Dibaji, S. M., A. Seidavi, L. Asadpour and M. D. S. Fernando (2014). Effect of a synbiotic on the intestinal microflora of chickens. J Appl Poultry Res. 23: 1-6.
- Du, E., L. Gan, Z. Li, W. Wang, D. Liu and Y. Guo (2015). In vitro antibacterial activity of thymol and carvacrol and their effects on broiler chickens challenged with Clostridium perfringens. J Anim Sci Biotechnol. 6: 58.
- Duc, L. H., H. A. Hong, N. Fairweather, E. Ricca and S. M. Cutting (2003). Bacterial spores as vaccine vehicles. Infect Immun. 71(5): 2810-2818.
- Duncan, S. H., P. Louis and H. J. Flint (2004). Lactate-utilizing bacteria, isolated from human feces, that produce butyrate as a major fermentation product. Appl Environ Microbiol. 70(10): 5810-5817.
- Ekim, B., A. Calik, A. Ceylan and P. Saçaklı (2020). Effects of Paenibacillus xylanexedens on growth performance, intestinal histomorphology, intestinal microflora, and immune response in broiler chickens challenged with Escherichia coli Poult Sci. 99(1): 214-223.
- Fuller, R (1989). Probiotics in man and animals. J Appl Bacteriol. 66(5), 365-378.
- Gadde, U., S. T. Oh, Y. S. Lee, E. Davis, N. Zimmerman, T. Rehberger and H. S. Lillehoj (2017). The effects of direct-fed microbial supplementation, as an alternative to antibiotics, on growth performance, intestinal immune status, and epithelial barrier gene expression in broiler chickens. Probiotics Antimicrob Proteins. 9(4): 397-405.
- Guo, M., G. Hao, B. Wang, N. Li, R. Li, L. Wei and T. Chai (2016). Dietary administration of Bacillus subtilis enhances growth performance, immune response and disease resistance in Cherry Valley ducks. Front Microbiol. 7: 1975.
- Hassanpour, H., A. K. Z. Moghaddam, M. Khosravi and M. Mayahi (2013). Effects of synbiotic on the intestinal morphology and humoral immune response in broiler chickens. Livest Sci. 153(1-3): 116-122.
- Huang, T., X. Y. Peng, B. Gao, Q. L. Wei, R. Xiang, M. G. Yuan and Z. H. Xu (2019). The effect of Clostridium butyricum on gut microbiota, immune response and intestinal barrier function during the development of necrotic enteritis in chickens. Front Microbiol. 10: 2309.
- Huyghebaert, G, R Ducatelle and Van Immerseel F (2011). An update on alternatives to antimicrobial growth promoters for broilers. Vet J. 187(2): 182-188.
- Jayaraman, S., G. Thangavel, H. Kurian, R. Mani, R. Mukkalil and H. Chirakkal (2013). Bacillus subtilis PB6 improves intestinal health of broiler chickens challenged with Clostridium perfringens-induced necrotic enteritis. Poult Sci. 92(2): 370-4.
- Jin, L. Z., Y. W. Ho, M. A. Ali, N. Abdullah, K. B. Ong and S. Jalaludin (1996). Adhesion of Lactobacillus isolates to intestinal epithelial cells of chicken. Lett Appl Microbiol. 22(3): 229-232.
- Kridtayopas, C., C. Rakangtong, C. Bunchasak and W. Loongyai (2019). Effect of prebiotic and synbiotic supplementation in diet on growth performance, small intestinal morphology, stress, and bacterial population under high stocking density condition of broiler chickens. Poult Sci. 98(10): 4595-4605.
- Le Roy, C. I., J. Štšepetova, E. Sepp, E. Songisepp, S. P. Claus and M. Mikelsaar (2015). New insights into the impact of Lactobacillus population on host-bacteria metabolic interplay. Oncotarget. 6(31): 30545–30556.
- Li, J., Y. Cheng, Y. Chen, H. Qu, Y. Zhao, C. Wen and Y. Zhou (2019). Dietary chitooligosaccharide inclusion as an alternative to antibiotics improves intestinal morphology, barrier function, antioxidant capacity, and immunity of broilers at early age. Animals (Basel). 9(8): 493.
- Li, S. P., X. J. Zhao and J. Y. Wang (2009). Synergy of Astragalus polysaccharides and probiotics (Lactobacillus and Bacillus cereus) on immunity and intestinal microbiota in chicks. Poult Sci. 88(3): 519-525.
- Liu, C., A. Li, Y. B. Weng, M. L. Duan, B. E. Wang and S. W. Zhang (2009). Changes in intestinal mucosal immune barrier in rats with endotoxemia. World J Gastroenterol. 15(46): 5843-5850.
- Liu, W., W. Wang, C. Ran, S. He, Y. Yang and Z. Zhou (2017a). Effects of dietary scFOS and Lactobacilli on survival, growth, and disease resistance of hybrid tilapia. Aquaculture. 470: 50-55.
- Liu, X. L., H. Fan, L. B. Chen, H. H. Li, J. Y. Qiao and W. J. Wang (2017b). Effects of cecropin and synbiotics on the intestinal mucosal morphology and intestinal mucosal immune cells of AA broilers. China Animal Husbandry & Veterinary Medicine. 44(11): 3187-3194.
- Livak, K. J. and T. D. Schmittgen (2001). Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT Methods. 25(4):402-408.
- Louis, P., S. H. Duncan, S. I. McCrae, J. Millar, M. S. Jackson and H. J. Flint (2004). Restricted distribution of the butyrate kinase pathway among butyrate-producing bacteria from the human colon. J Bacteriol. 186(7): 2099-2106.
- Ma, Y., W. Wang, H. Zhang, J. Wang, W. Zhang, J. Gao, S. Wu and G. Qi (2018). Supplemental Bacillus subtilis DSM 32315 manipulates intestinal structure and microbial composition in broiler chickens. Sci Rep. 8(1): 15358.
- Maesschalck, C. D., V. Eeckhaut, L. Maertens, L. D. Lange, L. Marchal, C. Nezer, S. D. Baere, S. Croubels, G. Daube, J. Dewulf, F. Haesebrouck, R. Ducatelle, B. Taminau and F. V. Immerseel (2015). Effects of xylo-oligosaccharides on broiler chicken performance and microbiota. Appl Environ Microbiol. 81(17): 5880-5888.
- Magalhaes, J. G., I. Tattoli and S. E. Girardin (2007). The intestinal epithelial barrier: how to distinguish between the microbial flora and pathogens. Semin Immunol. 19(2): 106-115.
- Min, Y. N., H. L. Yang, Y. X. Xu and Y. P. Gao (2016). Effects of dietary supplementation of synbiotics on growth performance, intestinal morphology, sIgA content and antioxidant capacities of broilers. J Anim Physiol Anim Nutr (Berl). 100(6): 1073-1080.
- Mookiah, S., C. C. Sieo, K. Ramasamy, N. Abdullah, Y. W. Ho (2013). Effects of dietary prebiotics, probiotic and synbiotics on performance, caecal bacterial populations and caecal fermentation concentrations of broiler chickens. J Sci Food Agric. 94(2): 341-348.
- Musa, B. B., Y. Duan, H. Khawar, Q. Sun, Z. Ren, M. A. Elsiddig Mohamed, I. H. R. Abbasi and X. Yang (2019). Bacillus subtilis B21 and Bacillus licheniformis B26 improve intestinal health and performance of broiler chickens with Clostridium perfringens-induced necrotic enteritis. J Anim Physiol Anim Nutr (Berl). 103(4): 1039-1049.
- Patterson, A. M. and A. J. M. Watson (2018). Intraepithelial cells are the first line of defense against enteric infection. Gastroenterology. 155: 1-3.
- Pryde, S. E., S. H. Duncan, G. L. Hold, C. S. Stewart and H. J. Flint (2002). The microbiology of butyrate formation in the human colon. FEMS Microbiol Lett. 217(2): 133-139.
- Roberfroid, M. (2007). Prebiotics: the concept revisited. J Nutr. 137(3 Suppl 2): 830S-837S.
- Romero, E. S., C. C. Alonso, C. C. Pardo, B. M. Casado and M. Vicario (2015). The intestinal barrier function and its involvement in digestive disease. Rev Esp Enferm Dig. 107(11): 686-696.
- Schrezenmeir, J. and M. D. Vrese (2001). Probiotics, prebiotics, and synbiotics-approaching a definition. Am J Clin Nutr. 73(2 suppl): 361s-364s.
- Sheng, K., S. He, M. Sun, G. Zhang, X. Kong, J. Wang and Y. Wang (2020). Synbiotic supplementation containing Bifidobacterium infantis and xylooligosaccharides alleviates dextran sulfate sodium-induced ulcerative colitis. Food Funct. 11(5): 3964-3974.
- Tanner, S. A., C. Lacroix, C. Del'Homme, C. Jans, A. Zihler Berner, A. Bernalier-Donadille and C. Chassard (2016). Effect of Bifidobacterium thermophilum RBL67 and fructo-oligosaccharides on the gut microbiota in Göttingen minipigs. Br J Nutr. 114(5): 746-755.
- Tian, Z., Y. Cui, Z. Du and X. Ma (2020). Advances in researches and applications of antibiotic alternatives in livestock breeding. Chinese Journal of Animal nutrition. 32(4): 1516-1525.
- Tohid, T., G. Hasan and T. Alireza (2010). Efficacy of mannanoligosaccharides and humate on immune response to Avian Influenza (H9) disease vaccination in broiler chickens. Vet Res Commun. 34(8): 709-717.
- Vandenpla, Y. S, E. D. Greef, T. Devreker, G. Veereman-Wauters and B. Hauser (2013). Probiotics and prebiotics in infants and children. Curr Infect Dis Rep. 15(3): 251-262.
- Wang, Y., B. Wang, Q. Liu, C. Fan, J. Li, Y. Zhou and S. Zhuang (2019). Palygorskite supplementation improves growth performance, oxidative status, and intestinal barrier function in Cherry Valley Ducks. J Poult Sci. 56(3): 186-194.
- Wang, Y., W. Du, K. Lei, B. Wang, Y. Wang, Y. Zhou and W. Li (2017). Effects of dietary Bacillus licheniformis on gut physical barrier, immunity, and reproductive hormones of laying hens. Probiotics Antimicrob Proteins, 9(3), 292-299.
- Wen, Z. S., M. Du, Z. Tang, T. Y. Zhou, Z. S. Zhang, H. H. Song, X. W. Xiang and X. Y. Han (2019). Low molecular seleno-aminopolysaccharides protect the intestinal mucosal barrier of rats under weaning stress. Int J Mol Sci. 20(22): 5727.
- Xing, S., B. Zhang, M. Lin, P. Zhou, J. Li, L. Zhang, F. Gao, G. Zhou (2017). Effects of alanyl-glutamine supplementation on the small intestinal mucosa barrier in weaned piglets. Asian-Australas J Anim Sci. 30(2): 236-245.
- Xing, Y., S. Wang, J. Fan, A. O. Oso, S. W. Kim, D. Xiao, T. Yang, G. Liu, G. Jiang, Z. Li, L. Li and B. Zhang (2015). Effects of dietary supplementation with lysine-yielding Bacillus subtilis on gut morphology, cecal microflora, and intestinal immune response of Linwu ducks. J Anim Sci. 93(7):3449-3457.
- Xiong, X., H. S. Yang, X. C. Wang, Q. Hu, C. X. Liu, X. Wu, D. Deng, Y. Q. Hou, C. M. Nyachoti, D. F. Xiao and Y. L. Yin (2015). Effect of low dosage of chito-oligosaccharide supplementation on intestinal morphology, immune response, antioxidant capacity, and barrier function in weaned piglets. J Anim Sci. 93(3): 1089–1097.
- Yang, C., L. Zhang, G. Cao, J. Feng, M. Yue, Y. Xu, B. Dai, Q. Han and X. Guo (2019). Effects of dietary supplementation with essential oils and organic acids on the growth performance, immune system, fecal volatile fatty acids, and microflora community in weaned piglets. J Anim Sci. 97(1): 133-143.
- Yin, J., F. Li, X. Kong, C. Wen, Q. Guo, L. Zhang, W. Wang, Y. Duan, T. Li, Z. Tan and Y. Yin (2019). Dietary xylo-oligosaccharide improves intestinal functions in weaned piglets. Food Funct. 10(5): 2701-2709.
- Zhuang, S., F. B. Jiang, Z. X. Jia and Y. Ran (2015). Clostridium butyricum can be used as a potential alternative for the antibiotic in Cherry Valley ducks. J Anim Plant Sci. 25(5): 1227-1232.
|





