EVIDENCE OF ANTI-GOUT ACTIVITY OF A STANDARDIZED TRADITIONAL HERBAL MEDICINE
A. Shaukat*, K. Hussain, N. I. Bukhari, N. Shehzadi, S. Naheed, S. Siddique, F. Saghir and S. Iftikhar
Punjab University College of Pharmacy, University of the Punjab, Allama Iqbal Campus Lahore-54000, Pakistan
*Corresponding author’s E-mail: Ayishashaukat@gmail.com
ABSTRACT
A five-herb containing traditional homemade medicine is extensively used to treat gout but the literature is devoid of scientific evidence. Therefore, the present study aimed to investigate anti-gout and anti-inflammatory activities of a standardized traditional medicine. Reversed phase HPLC method was developed for the simultaneous determination of chlorogenic acid, caffeic acid, vanillin and ferulic acid, and applied to standardize the traditional medicine. Completely randomized design (CRD) was followed in the current study. In vivo anti-gout activity was evaluated using potassium oxonate induced hyperuricemia model and the study was divided into two parts, protective and curative study. In former, male Wistar rats were randomly segregated into 4 groups (n=6) and treated for 21 days while in later, rats from toxic group were randomly segregated into 3 groups (n=3), two treated and one control, and treatment was continued for 7 days. Serum uric acid level, xanthine oxidase activity of rat-liver homogenate and renal histopathology were used to assess anti-gout activity. The medicine was also evaluated for anti-inflammatory activity using carrageenan induced hind paw edema model. Data were analyzed by one-way ANOVA, and a p value ≤ 0.05 was regarded as significantly different. The medicine contained 0.2967 mg/g chlorogenic acid, 0.2698 mg/g caffeic acid, 0.2122 mg/g vanillin and 0.2160 mg/g ferulic acid. The medicine significantly reduced uric acid level in medicine-treated groups in both protective and curative studies (p≤0.05). The medicine also exhibited significant xanthine oxidase inhibition in rat-liver homogenate in protective and curative study (p≤0.05). Kidney histopathological examination indicated mild interstitial cells infiltration in treatment groups as compared to toxic group. The medicine also exhibited significant anti-inflammatory activity indicated by 49.8% reduction of paw edema volume (p≤0.05). The findings of this study support the traditional use of the herbal medicine, and the developed HPLC method may be useful to produce standardized anti-gout medicine.
Keywords: Gout, HPLC, standardization, polyherbal, xanthine oxidase, potassium oxonate, allopurinol.
https://doi.org/10.36899/JAPS.2021.6.0388
Published online March 31, 2021
INTRODUCTION
Gout is an inflammatory disorder which is characterized by swelling and inflammation of synovial joints, raised uric acid level in plasma and sodium urate crystals deposition in joints and kidneys. It may occur due to some pathological condition or as a comorbidity of heart and kidney diseases or intake of certain medicines (Kramer and Curhan, 2002). Serum uric acid level is an independent predictor of mortality in populations at high risk of coronary artery disease (Grassi et al., 2013). Higher serum uric acid level may result in deposition of sodium urate crystals in joints (Schumacher, 1996). Monosodium urate crystals induced release of lysosomal enzymes from leucocytes in the absence of phagocytosis (Sabina et al., 2011). The deposition of these crystals results in the release of microcrystals from preformed crystals which interact with monocytes, neutrophils, fibroblasts and macrophages. These inflammatory microcrystals also stimulate secretion and synthesis of proinflammatory cytokines and interleukins, responsible of acute inflammatory response and gout attack (Guerne et al., 1989). These microcrystals can also specifically induce COX-2 expression in monocytes; a new factor involved in monosodium urate crystals induced inflammation (Pouliot et al., 1998). Mononucleated and multinucleated macrophages are also involved in the development of gout tophi resulting in the production of proinflammatory cytokines and gout flares (Schweyer et al., 2000).
The risk of gout can be reduced either by reducing biosynthesis or increasing excretion of uric acid from the body (Umamaheswari et al., 2007). The treatment of gout was suggested by using combination of relevant treatment, such as uric acid lowering by using selective enzyme inhibitors in uric acid biosynthesis, inhibition of inflammatory responses by using anti-inflammatory agents and modification of dietary life style (Cannella and Mikuls, 2005). Allopurinol is one of the most widely used xanthine oxidase inhibitor (Fields et al., 1996; Pacher et al., 2006); however, superoxide generation leading severe hypersensitivity reaction, gastrointestinal irritation, renal toxicity, bone marrow suppression and severe liver function abnormalities are reported due to use of XOI (Umpierrez et al., 1998; Kong et al., 2000). Hence, due to high cost and serious adverse effects on utilization of synthetic medicines (Strazzullo and Puig, 2007), different attempts have been made to search safe alternative therapy from natural source for curing gout (Kong et al., 2004; Zhu et al., 2004).
A folklore polyherbal medicine comprising five ingredients (seeds of Tribulus terrestris, Carthamus tinctorius, Cucumis melo and Punica granatum, and seed-less dried fruit of Vitis vinifera) is being extensively used and promoted through social media in Pakistan and India for curing gout. Though, the literature supports the use of such herbs in treating hyperuricemia, nephrolithiasis, osteoarthritis and inflammation (Wang et al., 2004; Joshi et al., 2005; Arasaratnam et al., 2010; Arora et al., 2011; Ravishankar and Priya, 2012; Singh and Kaushik, 2017). However, we could not find any scientific evidence for effectiveness of this polyherbal remedy, which necessitates to investigate its anti-gout and anti-inflammatory potential using appropriate experimental models.
Herbal formulations are inconsistent in terms of chemical constituents due to various genetic, growing, harvesting and environmental factors. Hence, for reproducible claim, a herbal product needs to be standardized prior to pharmacological or clinical evaluation. Herbal standardization can be achieved using a set of diverse analytical activities including the use of markers. Eight different types of analytical markers were reported for standardization of herbal products (Li et al., 2008). Hence caffeic acid, chlorogenic acid, vanillin and ferulic acid were selected as markers because of their presence in different ingredients of the selected remedy (Wang et al., 2010; Chhatre et al., 2014; Ben et al., 2015; Shao et al., 2016; Mallek et al., 2018). These compounds possess xanthine oxidase inhibitory and anti-inflammatory potential hence can be used as pharmacologically active markers to be used as analytical standards in HPLC method development and validation (Murakami et al., 2007; Wang et al., 2007; Compaore et al., 2012). The literature contained several types of in vivo models for evaluation of anti-gout and anti-inflammatory potential (Schiltz et al., 2002; Sabina and Rasool, 2008; Singh et al., 2009; Martin et al., 2009; Meng et al., 2014; Chen et al., 2016). In the present study, hyperuricemia induction by oxonic acid salt of potassium in rat models were utilized to investigate the herbal remedy for anti-gout activity and evaluated nephroprotective effect of remedy on experimental rats. Postassium oxonate inhibits uricase which converts uric acid to allantoin, hence used to induce hyperuricemia in experimental animals. Thus, in the current study, we developed a rat model with potassium oxonate induced hyperuricemia. Inflammation is relieved through various mechanisms, hence, hind paw oedema model (carrageenan induced) in albino rats was selected as in vivo model to investigate remedy in the current study. Keeping this in view, we have selected the described models to evaluate pharmacological activity of the polyherbal remedy. The results of the current study may give scientific evidence to traditional use of the standardized polyherbal remedy.
MATERIALS AND METHODS
Herbs: Seed of Tribulus terrestris, Carthamus tinctorius, Cucumis melo and Punica granatum, and dried-fruit of Vitis vinifera were obtained from the local herbal medicine market (Papar Mandi, Lahore, Pakistan). All the ingredients were cleaned from dirt, if any, and seeds were removed from Vitis vinifera fruit.
Chemicals: The materials utilized in the current study were caffeic acid, chlorogenic acid, ferulic acid, vanillin, xanthine oxidase (Sigma Aldrich), xanthine (Bioworld, Biofine Plus Research Chemicals), allopurinol (Toshima, Tokyo, Japan), potassium oxonate (Alfa Aesar), diclofenac sodium, carrageenan, methanol, acetonitrile, tetrahydrofuran, potassium dihydrogen phosphate, acetic acid, sodium acetate, sodium chloride and sodium hydroxide (Merck, Germany).
Instruments: Beckman Coulter AU480(USA), Electronic centrifuge, refrigerator centrifuge machine (Sigma 2-16 Kc, Germany), homogenizer (Wisestir Homogenizer Stirrer-HS-30E, Korea), double beam UV-Vis spectrophotometer(Shimadzu), liquid chromatographic system (Agilent Technologies, 1200 series, Germany) having an isocratic pump (G1310A), an auto-sampler (G1329A), thermostatically controlled column (G1316A) and diode array detector (G1315B), were used in the current study.
Preparation of anti-gout remedy: Anti-gout remedy was prepared by scaling-up the traditional method. All the experimentation was carried out from March-June, 2019, at Pharmaceutical Chemistry Research Laboratory, Punjab University College of Pharmacy, University of the Punjab, Lahore. Briefly, one hundred grams of Tribulus terrestris seeds, 50 g seeds of each of Carthamus tinctorius, Cucumis melo and Punica granatum, and 50 g dried seedless-fruit of Vitis vinifera were mixed and crushed gently. The resulting mixture was divided into 7 equal parts. One part was soaked in 300 mL water for 12 h and boiled for 20 min. The decoction was strained and concentrated by heating to 150 mL (Shaukat et al., 2020).
Standard solutions preparation: The standard stock solutions (1 mg/mL) of markers (chlorogenic acid, caffeic acid, vanillin, ferulic acid) were prepared in methanol. Chlorogenic acid (14 µL), caffeic acid (12 µL), vanillin (33 µL) and ferulic acid (11 µL), from stock solutions were mixed, making the volume 1 mL with mobile phase to give mixed standard stock solution. A range of mixed working solutions were obtained by dilution of mixed standard solution with mobile phase. These solution contained 0.14, 0.28, 0.56, 1.12, 2.24, 3.36, 6.72 and 12.04 µg/mL chlorogenic acid, 0.12, 0.24, 0.48, 0.96, 1.92, 2.88 and 5.76 µg/mL caffeic acid, 0.33, 0.66, 1.32, 2.64, 5.28, 7.92 and 15.84 µg/mL vanillin and 0.11, 0.22, 0.44, 0.88, 1.76, 2.64, 5.28 µg/mL ferulic acid (ICH, 2005).
Sample preparation: Herbal medicine (1 g) was dissolved in 2N HCl (10 mL) and kept at 100ºC for 1 h to hydrolyze glycosidic bonds. Then, aglycone part was extracted by partitioning with equal volume of ethyl acetate (Ewais et al., 2016). The solvent layer was collected and evaporated in vacuo at 40oC and concentration of 20 mg/mL was prepared for HPLC analysis.
Development of method: Mixed working standard solution containing 14 µg/mL chlorogenic acid, 12 µg/mL caffeic acid, 33 µg/mL vanillin and 11 µg/mL ferulic acid were analysed in triplicate. Sample(20 µL) elution was done by mobile phase (isocratic) comprising aqueous acetate buffer having pH 3.6: methanol: acetonitrile: tetrahydrofuran (65:20:10:5) at 0.8 mL/min flow rate through HPLC column (Agilent TC-C18; 250 × 4.6 mm), maintained at 35ºC with photodiode array detector operating at detection beam of 330 nm and reference beam of 360 nm. The peaks of the standards were identified in the sample by comparison of retention time. System suitability parameters (peak asymmetry, tailing factor, theoretical plates number (N), capacity factor (k´), height equivalent to theoretical plate (HETP), and resolution) were assessed using the mixed standard solution chromatogram (ICH, 2005).
Method validation: Linearity of the method was assessed analyzing the mixed standard solutions in triplicate. The mixed working standard solution contained chlorogenic acid (0.14-12.04 µg/mL), caffeic acid (0.12-5.76 µg/mL), vanillin (0.33-15.84 µg/mL) and ferulic acid (0.11-5.28 µg/mL). The plot between concentration and peak area of each standard was constructed and linearity was evaluated visually and applying the linear regression. The data points correlation was evaluated by coefficient of determination (r2= 0.9999-1.0000). Beer’s range was determined from linear portion of the concentration versus peak area. The recovery was determined spiking 20 mg of ethyl acetate extract of the medicine obtained with 1 mL of each of the three mixed working standard solutions containing caffeic acid (0.24, 0.48 and 0.96 µg/mL), chlorogenic acid (0.28, 0.56 and 1.12 µg/mL), ferulic acid (0.22, 0.44 and 0.88 µg/mL) and vanillin (0.66, 1.32 and 2.64 µg/mL). The analysis of spiked and un-spiked samples was done in triplicate and standards were quantified using the respective calibration curves. The calculated amount was then compared with the spiked amount to assess recovery (ICH, 2005).
For intraday precision and accuracy, each of the three mixed working standard solution used for recovery was analyzed six times within a day, while for interday precision and accuracy, each of three mixed working standard solution was analyzed once daily for six successive days. Accuracy was determined by quantification of each standard from respective standard curve constructed on each day, whereas the RSD of the six readings was taken as precision. LOD and LOQ were determined statistically analyzing five mixed working standard solutions containing 0.24-2.88 µg/mL caffeic acid, 0.28-3.36 µg/mL chlorogenic acid, 0.22-2.64 µg/mL ferulic acid and 0.66-7.92 µg/mL vanillin in quintuplicate. Standard curves of each standard were constructed to determine mean slope and standard deviation of intercepts. Robustness of method was determined by altering column temperature (±2⁰C), pH (±0.1) and detection wavelength (±2 nm) (ICH, 2005).
Pharmacological studies
Animals and grouping: Forty eight male Wistar rats, weight 190 ± 10 g, aged 15 weeks, acquired from Animal House, Punjab University College of Pharmacy, Lahore, Pakistan, were housed to acclimatize at room temperature with constant humidity for one week period. Rodent pellet-diet was provided and water was supplied adlibitum to experimental animals. These animals were kept in separate cages at the Animal House, following the regulations and guidelines of the Bioethics Committee, University of the Punjab, Lahore. The experimental procedures utilized in the current study were authorized and approved by the Bioethics Committee, Vide reference No. D/1304/02, Punjab University, Lahore, Pakistan.
The experiments were laid-down following completely randomized design. The in vivo anti-gout activity was determined by method described by Nguyen et al. (2017) with some modifications. Anti-gout activity protocol was designed into two parts, protective and curative. For the former (protective study), thirty male Wistar rats were segregated into four groups, each having six rats, except group-II which had twelve rats. The group-I served as control (uncompromised and untreated), group-II as toxic (Potassium oxonate treated), group-III as herbal medicine treated and group-IV as allopurinol treated, respectively. In curative study, nine rats separated from group-II (toxic group) after protective study period were segregated into three groups (n=3) as group-I served as vehicle control, group-II served as herbal medicine treated and group-III served as allopurinol treated.
The in vivo anti-inflammatory activity of herbal medicine was determined following the method reported in literature (Muhammad et al., 2012). Eighteen, male, Wistar rats, (independent samples) were segregated into three groups (each n=6), termed as group-I (vehicle control group), group-II (herbal medicine) and group-III (diclofenac sodium).
Preparation and administration of dose: A dose of 792 mg/kg was selected for in vivo anti-gout and anti-inflammatory studies of herbal medicine and it was determined on basis of following formula i.e. Animal equivalent dose (mg/kg)=(human equivalent in mg/kg)(Km of human/ Km of rat) (Nair and Jacob, 2016). Doses of herbal medicine, potassium oxonate, allopurinol and diclofenac sodium were prepared in a mixture of water and tween 80(95:5 V/V). For anti-gout activity, treatments given to each group were described as follows; group-I was administered vehicle, group-II was given potassium oxonate (250 mg/kg), group-III was given herbal medicine (792 mg/kg) and group-IV received allopurinol (10 mg/kg). To induce hyperuricemia, uricase inhibitor (potassium oxonate) was administered by oral gavage one hour prior to administration of test samples, in all treatment groups (II, III and IV) with the exception of control group (group-I) and the study was continued for 21 days for protective study design.
For curative study design, treatments given to each group were explained as follows; group-I was administered vehicle, group-II was given herbal medicine (792 mg/kg) and group-III was given allopurinol (10 mg/kg) by oral gavage for a period of 7 days.
For in vivo anti-inflammatory activity, group-wise treatments were as follows; group-I received vehicle, group-II received herbal medicine (792 mg/kg) and group-III received diclofenac sodium (10 mg/kg). There is oedema induction in hind paw of Wistar rats by subplantar injection of 1.0 % carrageenan (0.1 mL) immediately 1 h after the administration of test drug. Swelling in hind-paw was measured using Plethysmometer immediately before drug administration and later at 1, 2, 3, 4 and 24 h. Percent oedema inhibition was calculated in comparison with control group using following formula:
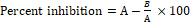
Where A represents oedema volume of control group and B as oedema volume of treatment group.
Collection of blood and tissues: One hour after last dose administration of the treatment in protective and curative study, the blood samples were withdrawn in plain tubes by cardiac puncture of experimental rats under diethyl ether anesthesia. Then euthanization of animals was done under diethyl ether anesthesia to harvest liver tissue which was washed with 0.9% normal saline. Liver homogenate was prepared by mincing liver tissue (150 mg) in a sterile petridish, kept on ice pack. Then the minced tissue was suspended in 50 millimolar phosphate buffer (3 mL; pH 7.4) and then homogenized for 5 min followed by centrifugation (3000 g for 10 min) to collect supernatant. Kidney sample of each experimental animal was preserved in buffered neutral formalin (10% V/V).
Biochemical analysis and histology:To collect serum, centrifugation of blood sample was done for 10 min at 2700 rpm and resulting samples were quantified for uric acid by Beckman Coulter AU480. For histopathology, kidney samples were treated by routine techniques of dehydration in alcohol and paraffin embedding method. Kidney tissue sections (4-5 µm thickness) were made and stained with the hematoxylin and eosin stains followed by histopathological examination of slides under light microscope.
Xanthine oxidase activity of liver homogenate:A reaction mixture containing 0.5 mL supernatant, phosphate buffer solution (3.5 mL) having pH 7.5 and 1 mM potassium oxonate(1 mL), was incubated at 37ºC for 15 min and then 0.250 mM xanthine(1 mL) was added and contents were further incubated(37ºC) for 30 min. The reaction was arrested by 0.5 M HCl (0.5 mL) followed by determination of absorbance at 290 nm. The xanthine oxidase activity was shown as nanomoles of uric acid produced per min/mg of protein content (Mo et al., 2007).
Statistical analysis:The results were mentioned as mean ± SD for validation studies and mean ± SE for in vivo studies. Data were analyzed by ANOVA (one-way) with PostHoc multiple comparisons with Bonferoni utilizing SPSS 22.0 (IBM SPSS statistics). A p value ≤ 0.05 was regarded as significantly different (Field, 2013).
RESULTS
Development of method:Mixed working standard solution containing 14 µg/mL chlorogenic acid, 12 µg/mL caffeic acid, 33 µg/mL vanillin and 11 µg/mL ferulic acid were analysed in triplicate. Sample(20 µL) was eluted by an isocratic mobile phase consisting of aqueous acetate buffer having pH 3.6: methanol: acetonitrile: tetrahydrofuran (65:20:10:5) at 0.8 mL/min flow rate through HPLC column (Agilent TC-C18), maintained at 35ºC with photodiode array detector operated at detection beam of 330 nm and reference beam of 360 nm. The peaks of the standards were identified in the sample by comparison of retention time. The HPLC chromatogram of mixed standard is given in Figure 1. System suitability parameters (peak asymmetry, tailing factor, theoretical plates number (N), capacity factor (k´), height equivalent to theoretical plate (HETP), and resolution) were assessed using the mixed standard solution chromatogram. These parameters are given in Table 1.
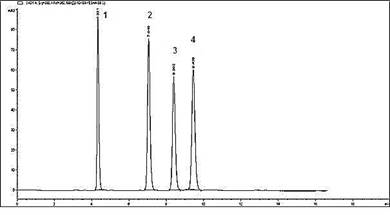
Figure 1. HPLC chromatogram of the markers (1) chlorogenic acid (2) caffeic acid (3) vanillin (4) ferulic acid
Table 1. System suitability parameters calculated from chromatogram of mixed standards
|
Parameters
|
Chlorogenic acid
|
Caffeic acid
|
Vanillin
|
Ferulic acid
|
Acceptable limit
|
|
k´
|
3.32
|
6.04
|
7.39
|
8.43
|
K ≥ 2
|
|
As
|
1.0
|
1.0
|
1.0
|
1.0
|
T ≤ 2
|
|
N
|
4779.7
|
12709.4
|
18028.9
|
22808.2
|
N ˃ 2000
|
|
HETP (µm)
|
52.3
|
19.6
|
13.8
|
10.9
|
The smaller the value, the higher the column efficiency
|
|
Rs Chlorogenic acid and caffeic acid
|
13.62
|
|
|
|
Rs≥ 1.5
|
|
Rs Chlorogenic acid and vanillin
|
20.35
|
|
|
|
Rs≥ 1.5
|
|
Rs Chlorogenic acid and ferulic acid
|
25.59
|
|
|
|
Rs≥ 1.5
|
k´ is capacity factor, As is peak asymmetry, N is number of theoretical plates, HETP is height equivalent to theoretical plate and Rs is resolution
Method validation:The developed analytical method was linear in the range investigated as chlorogenic acid (0.14-12.04 µg/mL), caffeic acid (0.12- 5.76 µg/mL), vanillin (0.33- 15.84 µg/mL) and ferulic acid (0.11- 5.28 µg/mL). The results of recovery, precision and accuracy of all the four standards are given in Table 2. These results depicted that the method exhibited good recovery, hence method was reliable. Intra-day and inter-day accuracy values of analytical markers were 90.4% to 114.94% and 91.5% to 114.9% with RSD less than 5%, which showed that the method was reproducible and repeatable. LOD of caffeic acid, chlorogenic acid, vanillin and ferulic acid were 0.10, 0.31, 0.082 and 0.073 µg/mL, respectively, while LOQ were found to be 0.32, 0.95, 0.25 and 0.22 µg/mL, respectively. These values determined in the present study were lesser than those reported previously which indicated the higher sensitivity of the method (Li et al., 2004). Hence, the method can be used to quantify these markers even at sub-microgram level. Furthermore, the method was robust as slight variation in mobile phase, detection wavelength and column temperature did not affect the accuracy.
Table 2. Recovery, Intra-day, inter-day accuracy and precision of the HPLC method for determination of chlorogenic acid, caffeic acid, vanillin and ferulic acid (n = 6)
|
Concentration
(µg/ml)
|
Recovery (%) ± SD
|
Intraday
|
Interday
|
|
Accuracy
|
Precision
|
Accuracy
|
Precision
|
|
Chlorogenic acid
|
|
0.28
|
95.0±0.0073
|
102.39
|
2.52
|
114.67
|
4.13
|
|
0.56
|
98.4±0.0042
|
110.49
|
0.46
|
113.62
|
1.55
|
|
1.12
|
99.3±0.0036
|
91.42
|
1.97
|
114.54
|
1.62
|
|
Caffeic acid
|
|
0.24
|
106.0±0.0041
|
114.90
|
1.24
|
113.76
|
2.23
|
|
0.48
|
98.90±0.0015
|
101.5
|
0.32
|
104.11
|
1.47
|
|
0.96
|
101.1±0.0027
|
114.94
|
1.95
|
114.94
|
0.55
|
|
Vanillin
|
|
0.66
|
96.8±0.0040
|
114.4
|
2.89
|
114.48
|
2.38
|
|
1.32
|
97.3±0.0040
|
90.47
|
1.38
|
91.55
|
2.78
|
|
2.64
|
100.9±0.004
|
114.92
|
0.15
|
114.92
|
1.04
|
|
Ferulic acid
|
|
0.22
|
100.08±0.002
|
114.73
|
0.72
|
114.7
|
0.67
|
|
0.44
|
102.5±0.0012
|
92.38
|
3.18
|
93.91
|
3.66
|
|
0.88
|
99.4±0.0012
|
112.83
|
0.24
|
114.71
|
0.94
|
Determination of markers in herbal medicine:The peaks of marker compounds were identified by comparing retention time and their area was used to quantify these compounds in the herbal medicine. The amount of marker compounds (chlorogenic acid, caffeic acid, vanillin and ferulic acid) quantified in herbal medicine were found to be 0.2967 mg/g, 0.2698 mg/g, 0.2122 mg/g and 0.2160 mg/g, respectively (Figure 2).
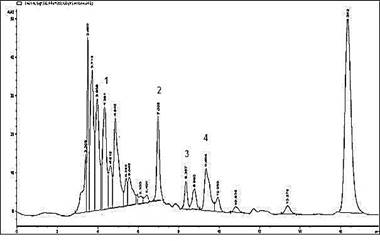
Figure 2. HPLC chromatogram of herbal medicine (1) chlorogenic acid (2) caffeic acid (3) vanillin (4) ferulic acid
Pharmacological studies:The results of protective anti-gout activity of herbal medicine assessed by level of serum uric acid and enzyme activity of liver homogenate are given in Figure 3. Administration of potassium oxonate by oral gavage resulted in significant (P≤0.05) hyperuricemia as indicated by raised serum uric acid level (4.8 mg/dL) in toxic group as compared to control group (0.6 mg/dL). The treatment with the herbal medicine (792 mg/kg) for 21 days duration significantly reduced serum uric acid level (1.46 mg/dL) and xanthine oxidase activity (0.1 nM/min/mg protein) of the liver homogenate. Allopurinol (10 mg/kg) showed significant (P≤0.05) reduction in serum uric acid level (0.93 mg/dL) and xanthine oxidase activity (0.06 nM/min/mg protein). The results indicated that herbal medicine (792 mg/kg) inhibited xanthine oxidase activity by 75.79% as compared to control group. Similarly, allopurinol (10 mg/kg) inhibited xanthine oxidase activity by 85.07%, exhibiting slightly more potent activity as compared to herbal medicine.
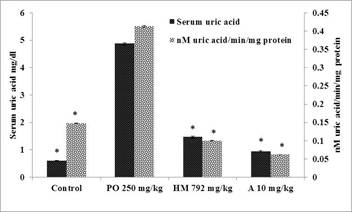
Figure 3. Protective study for determination of serum uric acid and nM uric acid/min/mg protein in rat liver in control, PO (Potassium oxonate 250 mg/kg) group, and treated groups with HM (herbal medicine 792 mg/kg); A (allopurinol 10 mg/kg). Each bar represents mean of six±SE, * significantly different than the toxic group, p≤0.05 from potassium oxonate, control, herbal medicine 792 mg/kg and allopurinol 10 mg/kg groups, respectively.
The results of curative role of herbal medicine assessed by level of serum uric acid and enzyme activity of liver homogenate are given in Figure 4. Group-II treated with the herbal medicine (792 mg/kg) for 7 days duration exhibited significant lowering of serum uric acid level (0.53 mg/dL) and xanthine oxidase activity (0.045 nM/min/mg protein) of liver homogenate. These results showed that administration of herbal medicine cured hyperuricemia in toxic group rats. Allopurinol (10 mg/kg) showed significant reduction of serum uric acid level (0.42 mg/dL) and xanthine oxidase activity (0.043 nM/min/mg protein) of liver homogenate.
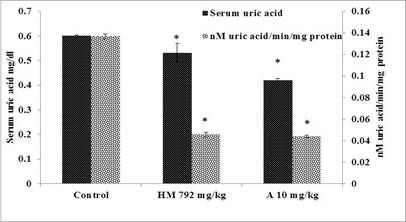
Figure 4.Curative study for determination of serum uric acid and nM uric acid/min/mg protein in rat liver in control, and treated groups with HM (herbal medicine); A (allopurinol). Each bar represents mean of six±SE, * significantly different than the control group, p≤0.05 from control, herbal medicine 792 mg/kg and allopurinol 10 mg/kg groups, respectively.
The histopathological examination of kidney-slides in different treatment groups indicated histological changes in the renal cells of toxic group due to renal damage caused by hyperuricemia resulting in cellular infiltration, glomerular damage and tubulointestinal nephritis. Mild cellular infiltration with improvement in renal histomorphological structure was observed in herbal medicine treatment group (Figure 5).
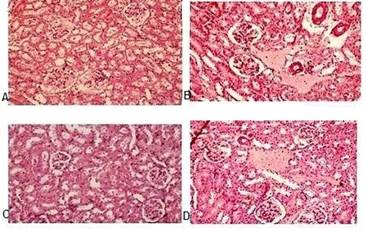
Figure 5. Microscopic images by light microscopy of kidney sections after Haematoxylin and Eosin staining (A) Vehicle control group; (B) Potassium oxonate 250mg/kg (toxic group); (C) HM (herbal medicine 792 mg/kg) treated; (D) Allopurinol (10 mg/kg) treated (positive control)
The anti-inflammatory potential of herbal medicine was assessed employing carrageenan-induced rat hind paw oedema model. On subplantar injection of carrageenan (1% w/v; 0.1 mL), there is marked increase in hind paw volume with maximal effect after 3 h. Group-II and group-III experimental animals treated with herbal medicine and diclofenac sodium caused significant reduction (p≤0.05) in oedema volume as compared to control group(Figure 6). Percent oedema inhibition by herbal medicine (792 mg/kg) and diclofenac sodium (10 mg/kg) was 26.45% and 20% in first hour and was maximum (49.8% and 46.78%) in the fourth hour of study.

Figure 6.Effect of herbal medicine (HM 792 mg/kg) and diclofenac sodium (DS 10 mg/kg) on carrageenan induced paw oedema volume in rats, asterick’s indicated values are statistically significant at p≤0.05 over control.
DISCUSSION
In the current study, a traditionally used polyherbal anti-gout remedy was standardized by a reversed-phase HPLC method using four phenolic compounds (chlorogenic acid, caffeic acid, vanillin and ferulic acid) as pharmacologically active analytical standards. The methods reported in the literature for quantification of different phenolic compounds required expensive reagents, instruments, techniques and could not be utilized for routine quality control (Aramendia et al., 2000; Alpar et al., 2018; Cittan and Celik, 2018; Paniagua et al., 2019). Moreover, there was no study regarding simultaneous quantification of these four markers using isocratic mobile phase. Hence, a simple and sensitive analytical method was developed and validated to standardize polyherbal anti-gout remedy. Optimum chromatographic conditions were obtained after running different mobile phases using reversed phase C18 column to achieve the best resolution. Detection wavelength of 330 nm resulted in an acceptable response and enabled the detection of these four phenolic compounds used in this study.
Pharmacological evaluation demonstrated that at dose of 792 mg/kg, polyherbal remedy exhibited significant reduction of serum uric acid level in oxonate induced hyperuricemia rats in both protective and curative study design. At dose (792 mg/kg) the remedy also notably inhibited hepatic xanthine oxidase activity in both study group rats. Hence, the hypouricemic effect of polyherbal remedy is caused by its inhibitory potential on xanthine oxidase (a key enzyme in the biosynthetic pathway of uric acid). Literature reported significant xanthine oxidase inhibitory potential of chlorogenic acid (Ozyurek et al., 2009; Meng et al., 2014), caffeic acid (Sud’Ina et al., 1993; Masuda et al., 2014) and ferulic acid (Wang et al., 2007; Nile et al., 2016), hence the marker compounds quantified in the polyherbal remedy contributed to significant pharmacological activity.
The anti-hyperuricemic property of polyherbal remedy observed in the study could also be explained by enzyme inhibitory effect of different flavonoids (Beiler and Martin, 1951; Iio et al., 1985; Nagao et al., 1999; Van et al., 2002; Lin et al., 2015), found in the ingredients of the remedy (Saleh et al., 1982; Schubert et al., 1999; Kim et al., 2007; Gill et al., 2011; Waghulde et al., 2011; Liu et al., 2012). Therefore, phenolic compounds and flavonoids found in the ingredients of the remedy contributed to significant anti-gout activity of the herbal remedy.
The herbal remedy also displayed notable anti-inflammatory activity in carrageenan-induced rat hind-paw oedema in vivo model. The phenolic compounds quantified in the remedy also exhibited significant anti-inflammatory potential by inhibiting release of cytokines, leukotrienes and pro-inflammatory mediators (interleukin-1β, TNF-α and interleukin-6) responsible for inflammation (Ozaki, 1992; Lim et al., 2008; Chao et al., 2009; Makni et al., 2011; Srikanth et al., 2013; Cunha et al., 2016; Gawlik et al., 2017). Hence, significant anti-inflammatory potential of herbal medicine was due to inhibition on release of these inflammatory mediators by phenolic compounds found in the herbal remedy.
Conclusion: The HPLC method developed and validated for simultaneous quantification of four standards is simple, sensitive and reliable, hence may be used to standardize anti-gout remedy. This study experimentally demonstrates that herbal medicine exhibits anti-hyperuricemic and anti-inflammatory activities. Hence, our findings gave scientific evidence for the traditional use of herbal remedy for treating gout.
Acknowledgements: Corresponding author would like to acknowledge Punjab University College of Pharmacy, Punjab University, Pakistan for provision of necessary research facilities during the study.
Conflict of interest: There is no conflict of interest.
REFERENCES
- Alpar, N., Y. Yardım and Z. Senturk (2018). Selective and simultaneous determination of total chlorogenic acids, vanillin and caffeine in foods and beverages by adsorptive stripping voltammetry using a cathodically pretreated boron-doped diamond electrode. Actuators. B. Chem. 257: 398-408.
- Aramendia, M. A., I.M. Garcia, F. Lafont, J. Lizaso, J.M. Marinas and F.J. Urbano (2000). Rapid determination of chlorogenic acid and related compounds in sunflower seeds by high‐performance liquid chromatography/atmospheric pressure chemical ionization mass spectrometry. Commun. Mass. Spectrom. 14(12): 1019-1022.
- Arasaratnam, V., S. Balakumar, A. Senthuran and R. Rajendraprasad (2010). A study of Tribulus terrestris extract on risk factors for urinary stone in normal subjects and urolithic patients. J. Natl. Sci. Found. 38(3): 189-193.
- Arora, R., M. Kaur and N.S. Gill (2011). Antioxidant activity and pharmacological evaluation of Cucumis melo agrestis methanolic seed extract. Res. J. Phytochemistry. 5(3): 146-155.
- Beiler, J.M. and G.J. Martin (1951). The inhibition of xanthine oxidase by flavonoids and related compounds. Biol. Chem. 192(2): 831-834.
- Ben Moumen, A., F. Mansouri, G. Richard, M. Abid, M.L. Fauconnier, M. Sindic and H. Serghini Caid (2015). Biochemical characterisation of the seed oils of four safflower (Carthamus tinctorius) varieties grown in north-eastern of Morocco. J. Food. Sci. Tech. 50(3): 804-810.
- Cannella, A.C. and T.R. Mikuls (2005). Understanding Treatments for Gout. Am. J. Manag. Care. 11(15): 451-458.
- Chao, P.C., C.C. Hsu and M.C. Yin (2009). Anti-inflammatory and anti-coagulatory activities of caffeic acid and ellagic acid in cardiac tissue of diabetic mice. Metab.6(1): 1-8.
- Chen, W.J., Y. Wu, X. Zhao, S. Liu, F.R. Song, Z.Y. Liu and Z.Q. Liu (2016). Screening the anti-gout traditional herbs from TCM using an in vitro method. Chem. Lett. 27(11): 1701-1707.
- Chhatre, S., T. Nesari, G. Somani, D. Kanchan and S. Sathaye (2014). Phytopharmacological overview of Tribulus terrestris. Rev. 8(15): 45-51.
- Cittan, M. and A. Çelik (2018). Development and validation of an analytical methodology based on liquid chromatography–electrospray tandem mass spectrometry for the simultaneous determination of phenolic compounds in olive leaf extract. Chromatogr. Sci. 56(4): 336-343.
- Compaore, M., C.E. Lamien, A. Lamien-Meda, L. Vlase, M. Kiendrebeogo, C. Ionescu and O. G. Nacoulma (2012). Antioxidant, xanthine oxidase and lipoxygenase inhibitory activities and phenolics of Bauhinia rufescens (Caesalpiniaceae). Nat. Prod. Res. 26(11): 1069-1074.
- Cunha, F.V.M., B. de Sousa Gomes, B. de Sousa Neto, A.R. Ferreira, D.P. de Sousa and F. de Assis Oliveira (2016). Ferulic acid ethyl ester diminished Complete Freund’s Adjuvant-induced incapacitation through antioxidant and anti-inflammatory activity. N-S. Arch. Pharmacol. 389(1): 117-130.
- Ewais, E.A., M.M. Abd El-Maboud, M.H. Elhaw and M.I. Haggag (2016). Phytochemical studies on Lycium schweinfurthii schweinfurthii (Solanaceae) and Isolation of five Flavonoids from leaves. J. Med. Plants. 4: 288-300.
- Fields, M., C.G. Lewis and M.D. Lure (1996). Allopurinol an inhibitor of xanthine oxidase reduces uric acid levels and modifies the signs associated with copper deficiency in rats fed fructose. Radic. Biol. Med.20(4): 595-600.
- Field, A. (2013). Discovering statistics using IBM SPSS statistics. sage. 4th Ed. Sage Publications; London. 818 p
- Gawlik-Dziki, U., D. Dziki, M. Swieca and R. Nowak (2017). Mechanism of action and interactions between xanthine oxidase inhibitors derived from natural sources of chlorogenic and ferulic acids. Chem.225(2017): 138-145.
- Gill, N.S., J. Bajwa, K. Dhiman, P. Sharma, S. Sood, P.D. Sharma and M. Bali (2011). Evaluation of therapeutic potential of traditionally consumed Cucumis meloAsian. J. Plant. Sci. 10(1): 86-91.
- Grassi, D., L. Ferri, G. Desideri, P. Di Giosia, P. Cheli, R. Del Pinto and C. Ferri (2013). Chronic hyperuricemia, uric acid deposit and cardiovascular risk. Pharm. Des. 19(13): 2432-2438.
- Guerne, P.A., R. Terkeltaub, B. Zuraw and M. Lotz (1989). Inflammatory microcrystals stimulate interleukin‐6 production and secretion by human monocytes and synoviocytes. Rheum. 32(11): 1443-1452.
- International Conference on Harmonization guidelines ICH Q2 (R1) (2005). Validation of analytical procedures: Text and methodology. International Conference on Harmonization, International Federation of Pharmaceutical Manufacturers and Associations, Geneva. Retrieved fromhttp://www.ich.org/fileadmin/Public_Web_Site/ICH_Products/Guidelines/Quality/Q2_R1/Step4/Q2_R1__Guideline.pdf.
- Iio, M., A. Moriyama, Y. Matsumoto, N. Takaki and M. Fukumoto (1985). Inhibition of xanthine oxidase by flavonoids. Biol. Chem.49(7): 2173-2176.
- Joshi, V.S., B.B. Parekh, M.J. Joshi and A.B. Vaidya (2005). Herbal extracts of Tribulus terrestris and Bergenia ligulata inhibit growth of calcium oxalate monohydrate crystals in vitro. Cryst. Growth. 275(1-2): 1403-1408.
- Kim, E.O., J.H. Oh, S.K. Lee, J.Y. Lee and S.W. Choi (2007). Antioxidant properties and quantification of phenolic compounds from safflower (Carthamus tinctorius) seeds. Food. Sci. Biotechnol. 16(1): 71-77.
- Kong, L.D., Y. Zhang, X. Pan, R.X. Tan and C.H.K. Cheng (2000). Inhibition of xanthine oxidase by liquiritigenin and isoliquiritigenin isolated from Sinofranchetia chinensis. Mol. Life. Sci.57(3): 500-505.
- Kong, L.D., C. Yang, F. Ge, H.D. Wang and Y.S. Guo (2004). A Chinese herbal medicine Ermiao wan reduces serum uric acid level and inhibits liver xanthine dehydrogenase and xanthine oxidase in mice. Ethnopharmacol.93(2-3): 325-330.
- Kramer, H. M. and G, Curhan (2002). The association between gout and nephrolithiasis: the National Health and Nutrition Examination Survey III, 1988-1994. J. Kidney. Dis.40(1): 37-42.
- Li, S., Q. Han, C. Qiao, J. Song, C.L. Cheng and H. Xu (2008). Chemical markers for the quality control of herbal medicines: an overview. Chin. Med. 3(1): 1-16.
- Li, X.P., J. Yu, J.Y. Luo, H.S. Li, F.J. Han, X.G. Chen and Z.D. Hu (2004). Simultaneous determination of chlorogenic acid, caffeic acid, ferulic acid, protocatechuic acid and protocatechuic aldehyde in chinese herbal preparation by RP-HPLC. Pharm. Bull.52(10): 1251-1254.
- Lim, E.J., H.J. Kang, H.J. Jung, Y.S. Song, C.J. Lim and E.H. Park (2008). Anti-angiogenic, anti-inflammatory and anti-nociceptive activities of vanillin in ICR mice. Ther. 16: 132-136.
- Lin, S., G. Zhang, Y. Liao, J. Pan and D. Gong (2015). Dietary flavonoids as xanthine oxidase inhibitors: Structure–affinity and structure–activity relationships. Agr. Food. Chem. 63(35): 7784-7794.
- Liu, T., J. Zhao, L. Ma, Y. Ding and D. Su (2012). Hepatoprotective effects of total triterpenoids and total flavonoids from Vitis vinifera L against immunological liver injury in mice. Based. Complement. Alternat. Med. 2012: 1-8.
- Makni, M., Y. Chtourou, H. Fetoui, E.M. Garoui, T. Boudawara and N. Zeghal (2011). Evaluation of the antioxidant, anti-inflammatory and hepatoprotective properties of vanillin in carbon tetrachloride-treated rats. J. Pharmacol.668(1-2): 133-139.
- Mallek-Ayadi, S., N. Bahloul and N. Kechaou (2018). Chemical composition and bioactive compounds of Cucumis melo seeds: Potential source for new trends of plant oils. Process. Saf. Environ. 113: 68-77.
- Martin, W.J., P.M. Herst, E.W. Chia and J.L. Harper (2009). Sesquiterpene dialdehydes inhibit MSU crystal-induced superoxide production by infiltrating neutrophils in an in vivo model of gouty inflammation. Radic. Biol. Med. 47(5): 616-621.
- Masuda, T., Y. Shingai, C. Takahashi, M. Inai, Y. Miura, S. Honda and A. Masuda (2014). Identification of a potent xanthine oxidase inhibitor from oxidation of caffeic acid. Free. Radic. Biol. Med. 69(2014): 300-307.
- Meng, Z.Q., Z.H. Tang, Y.X. Yan, C.R. Guo, L. Cao, G. Ding and Z.L. Yang (2014). Study on the anti-gout activity of chlorogenic acid: Improvement on hyperuricemia and gouty inflammation. J. Chin. Med. 42(6): 1471-1483.
- Mo, S.F., F. Zhou, Y.Z. Lv, Q.H. Hu, D.M. Zhang and L.D. Kong (2007). Hypouricemic action of selected flavonoids in mice: structure–activity relationships. Pharm. Bull.30(8): 1551-1556.
- Muhammad, N., M. Saeed and H. Khan (2012). Antipyretic, analgesic and anti-inflammatory activity of Viola betonicifolia whole plant. Complement. Altern. Med.12(1): 1-8.
- Murakami, Y., A. Hirata, S. Ito, M. Shoji, S. Tanaka, T. Yasui and S. Fujisawa (2007). Re-evaluation of cyclooxygenase-2-inhibiting activity of vanillin and guaiacol in macrophages stimulated with lipopolysaccharide. Res. 27(2): 801-807.
- Nagao, A., M. Seki and H. Kobayashi (1999). Inhibition of xanthine oxidase by flavonoids. Biosci. Bioch. 63(10): 1787-1790.
- Nair, A.B. and S. Jacob (2016). A simple practice guide for dose conversion between animals and human. Basic. Clin. Pharm. 7(2): 27-31.
- Nguyen, T.D., P.T. Thuong, I.H. Hwang, T.H.K. Hoang, M.K. Nguyen, H.A. Nguyen and M. Na (2017). Anti-hyperuricemic, anti-inflammatory and analgesic effects of Siegesbeckia orientalis Resulting from the fraction with high phenolic content. BMC. Complement. Altern. Med. 17(1): 1-9.
- Nile, S.H., E.Y. Ko, D.H. Kim and Y.S. Keum (2016). Screening of ferulic acid related compounds as inhibitors of xanthine oxidase and cyclooxygenase-2 with anti-inflammatory activity. Bras. Farmacogn.26(1): 50-55.
- Ozaki, Y. (1992). Antiinflammatory effect of tetramethylpyrazine and ferulic acid. Pharm. Bull.40(4): 954-956.
- Ozyurek, M., B. Bektaşoglu, K. Guçlu and R. Apak (2009). Measurement of xanthine oxidase inhibition activity of phenolics and flavonoids with a modified cupric reducing antioxidant capacity (CUPRAC) method. Chim. Acta.636(1): 42-50.
- Pacher, P., A. Nivorozhkin and C. Szabo (2006). Therapeutic effects of xanthine oxidase inhibitors: renaissance half a century after the discovery of allopurinol. Rev. 58(1): 87-114.
- Paniagua-Garcia, A. I., M. Hijosa-Valsero, J. Garita-Cambronero, M. Coca and R. Diez-Antolinez (2019). Development and validation of a HPLC-DAD method for simultaneous determination of main potential ABE fermentation inhibitors identified in agro-food waste hydrolysates. J. 150: 104-147.
- Pouliot, M., M.J. James, S.R. McColl, P.H. Naccache and L.G. Cleland (1998). Monosodium urate microcrystals induce cyclooxygenase-2 in human monocytes. 91(5): 1769-1776.
- Ravishankar, K. and P.S.V.V. Priya (2012). Evaluation of diuretic effect of ethanolic seed extracts of Macrotyloma uniflorum and Cucumis melo in rats. J. Pharma. Bio. Sci.3(3): 251-255.
- Sabina, E.P., S. Nagar and M. Rasool (2011). A role of piperine on monosodium urate crystal induced inflammation-an experimental model of gouty arthritis. Inflammation. 34(3): 184-1
- Sabina, E.P. and M. Rasool (2008). An in vivo and in vitro potential of Indian ayurvedic herbal formulation Triphala on experimental gouty arthritis in mice. Pharmacol. 48(1): 14-20.
- Saleh, N.A., A.A. Ahmed and M.F. Abdalla (1982). Flavonoid glycosides of Tribulus pentandrus and terrestris. Phytochemistry. 21(8): 1995-2000.
- Schiltz, C., F. Liote, F. Prudhommeaux, A. Meunier, R. Champy, J. Callebert and T. Bardin (2002). Monosodium urate monohydrate crystal–induced inflammation in vivo: quantitative histomorphometric analysis of cellular events. Rheum. 46(6): 1643-1650.
- Schubert, S.Y., E.P. Lansky and I. Neeman (1999). Antioxidant and eicosanoid enzyme inhibition properties of pomegranate seed oil and fermented juice flavonoids. Ethnopharmacol.66(1): 11-17.
- Schumacher Jr, H.R. (1996). Crystal-induced arthritis: an overview. J. Med. 100(2): 46S-52S.
- Schweyer, S., B. Hemmerlein, H.J. Radzun and A. Fayyazi (2000). Continuous recruitment, co-expression of tumour necrosis factor-α and matrix metalloproteinases, and apoptosis of macrophages in gout tophi. J. Pathol. 437(5): 534-539.
- Shao, D., L. Zhang, S. Du, W. Yokoyama, J. Shi, N. Li and J. Wang (2016). Polyphenolic content and color of seedless and seeded shade dried chinese raisins. Sci. Technol. Res. 22(3): 359-369.
- Shaukat, A., K. Hussain, N.I. Bukhari, N. Shehzadi, S. Naheed, F. Saghir, S. Iftikhar and O. Javed (2020). In vitro anti-gout and anti-inflammatory activity of traditionally used polyherbal anti-gout remedy. Int. J. Biosci. 16(5): 327-335.
- Singh, H.P., P. Utreja, A.K. Tiwary and S. Jain (2009). Elastic liposomal formulation for sustained delivery of colchicine: in vitro characterization and in vivo evaluation of anti-gout activity. J. 11(1): 54-64.
- Singh, M. and M.L. Kaushik (2017). A Review on Treatments of Gout for Future Aspects. Research and Reviews. Pharm. Sci. 8(3): 1-6.
- Sud'Ina, G.F., O.K. Mirzoeva, M.A. Pushkareva, G. Korshunova, N.V. Sumbatyan and S.D. Varfolomeev (1993). Caffeic acid phenethyl ester as a lipoxygenase inhibitor with antioxidant properties. Lett. 329(1-2): 21-24.
- Srikanth, D., V.H. Menezes, N. Saliyan, U.P. Rathnakar, S.D. Acaharya, A.K. Shenoy and A.L. Udupa (2013). Evalution of anti-inflammatory property of vanillin in carrageenan induced paw edema model in rats. J. Bioassays.2(1): 269-271.
- Strazzullo, P. and J.G. Puig (2007). Uric acid and oxidative stress: relative impact on cardiovascular risk. Nutr. Metab. Cardiovasc. Dis. 17(6): 409-414.
- Umamaheswari, M., K. AsokKumar, A. Somasundaram, T. Sivashanmugam, V. Subhadradevi and T.K. Ravi (2007). Xanthine oxidase inhibitory activity of some Indian medical plants. Ethnopharmacol. 109(3): 547-551.
- Umpierrez, A., J. Cuesta-Herranz, M. De Las Heras, M. Lluch-Bernal, E. Figueredo and J. Sastre (1998). Successful desensitization of a fixed drug eruption caused by allopurinol. Allergy. Clin. Immunol. 101(2): 286-287.
- Van Hoorn, D.E., R.J. Nijveldt, P.A. Van Leeuwen, Z. Hofman, L. M'Rabet, D.B. De Bont and K. Van Norren. (2002). Accurate prediction of xanthine oxidase inhibition based on the structure of flavonoids. J. Pharmacol.451(2): 111-118.
- Waghulde, H., S. Kamble, P. Patankar, B. Jaiswal, S. Pattanayak, C. Bhagat and M. Mohan (2011). Antioxidant activity, phenol and flavonoid contents of seeds of Punica granatum (Punicaceae) and Solanum torvum (Solanaceae). 1: 193-202.
- Wang, R.F., W.D. Xie, Z. Zhang, D.M. Xing, Y. Ding, W. Wang and L.J. Du (2004). Bioactive Compounds from the seeds of Punica granatum (Pomegranate). Nat. Prod. 67(12): 2096-2098.
- Wang, R., Y. Ding, R. Liu, L. Xiang and L. Du (2010). Pomegranate: constituents, bioactivities and pharmacokinetics. Veg. Cereal. Sci. Biotech. 4(2): 77-87.
- Wang, F., L. Yang, K. Huang, X. Li, X. Hao, J. Stockigt and Y. Zhao (2007). Preparation of ferulic acid derivatives and evaluation of their xanthine oxidase inhibition activity. Prod. Res. 21(3): 196-202.
- Zhu, J.X., Y. Wang, L.D. Kong, C. Yang and X. Zhang (2004). Effects of Biota orientalis extract and its flavonoid constituents, quercetin and rutin on serum uric acid levels in oxonate-induced mice and xanthine dehydrogenase and xanthine oxidase activities in mouse liver. Ethnopharmacology.93(1): 133-140.
|





