EFFECT OF ARSENIC (As) ON GROWTH AND PHYSIOLOGY OF MAIZE (ZEA MAYS) IN VARYING SOILS
B. Hussain1, M. Sanaullah2, M. Iqbal1, F. Mahmood1, F. Azeem3, M. Shahid3, T. Shahzad1 and S. Hussain1*
1Department of Environmental Sciences & Engineering, Government College University Faisalabad, Pakistan
2Institute of Soil & Environmental Sciences, University of Agriculture Faisalabad, Pakistan.
3Department of Bioinformatics & Biotechnology, Government College University Faisalabad, Pakistan.
*Corresponding author’s email: sabirghani@gmail.com
ABSTRACT
Maize is one of the main fodders and food source and even staple food in different countries. The presence of arsenic (As) in the soil has been reported to affect the growth and productivity of agricultural crops. The plant uptake of As from soil might depend on the composition and type of soil texture. In this study, we assessed the effect of four levels (0, 25, 50 and 100 mg As kg-1 of soil) of As on As uptake as well as the growth and physiology of maize (Zea mays) grown in three soils varying in texture in a pot experiment conducted at Government college university Faisalabad in February-March, 2018. Results revealed that the growth parameters significantly decreased in response to increasing concentrations of As in all three types of soil. The most prominent effects of As treatments on maize were shown under sandy loam soil followed by loamy soil and then clayey soils. At highest level of As application, the maximum As concentration in plant shoot and root were found in sandy loam soil (45.1 and 93.4 mg kg-1) followed by that in loamy soil (39.9 and 86.2 mg kg-1) and clayey soils (34.9 and 81.1 mg kg-1). Similarly, the maximum Diethylenetriamine Pentaacetic Acid (DTPA) extractable As (2.4 mg kg-1) was detected in sandy loam soil, whereas, the lowest (2.1 mg kg-1) was observed in clayey soil. Antioxidant enzyme activities and protein contents were maximum in the soils amended with 25 mg as kg-1, and were significantly decreased in the soils containing higher levels of As. However, these values were relatively higher in the clayey soils followed by the loamy soil and least in the sandy loam soil. The reactive oxygen species (ROS) including hydrogen peroxide (H2O2), superoxide (O2-) and melondialdehyde (MDA) were found to increase with increasing level of As in the soils. The effects of As on ROS were also maximum in sandy loam soil followed by the loamy soil and minimum in clayey soil. Conclusively we can say that the presence of As significantly affected the growth and physiological parameters of the maize crop. However, the effects of As were more severe in sandy loam soil as compared to the other ones.
Keywords: Arsenic, Maize, Soil texture, DTPA extractable As, Growth, Antioxidants.
https://doi.org/10.36899/JAPS.2021.5.0338
Published online January 28, 2021
INTRODUCTION
Arsenic (As) is one of the oldest poisons in the world, metalloid in nature and was used in high profile murders in the Middle Ages and was famous as the “King of Poisons” as well as the “poison of kings” (Hughes et al., 2011). It was discovered by Alberts in 1250 AD. In the order of abundance, it is 12th most abundant element in human body, 20th most abundant element in earth’s crust and 14th most abundant element in sea water (Mandal and Suzuki, 2002). It occurs in inorganic forms [i.e As(-III), As (0), As (III), or As(V)] along with organic forms [for instance, monomethyl arsenic acid, arsenobetaine and arsenosugars]. Inorganic forms of Arsenic (As) are more mobile and toxic among which, As (III) is considered 60 times more lethal than As(V). As(V) is dominant in oxidized environments (Shakoor et al., 2015).
Arsenic is considered one of the emerging very toxic contaminants worldwide. The addition of Às in the environment is mainly because of different natural phenomena including geological/geothermal/volcanic events and weathering of rocks and minerals as well as because of various anthropogenic activities including agricultural activities (i.e. fertilizer, pesticides, herbicides, and seed treatments), industrial processes (e.g. tanneries, electroplating etc) and mining and smelting activities. Among the anthropogenic sources, the mining and smelting of As-containing ores, combustion of fossil fuels especially coal, landfilling by pulp, paper, tannery, and textile industries, discharge of chemical warfare agents, petroleum refining and pharmaceutical manufacturing are very common sources of As contamination in soils (States, 2015).
The presence of As in the soil environment is harmful because it results in contamination of food crops, fruits and vegetables grown on arsenic (As) contaminated earth. The As can, therefore, enter into food chain as well as commercially used human food products even in non-contaminated areas (Zhao et al., 2010). As per US Environmental Protection Agency (EPA), arsenic (As) is carcinogenic in nature and, according to the IARC (International Agency for Research on Cancer), Arsenic and its composites are ranked as a group one human carcinogen (Shahid et al., 2013). According to these reports, approximately 200 million people have been affected by As worldwide including Pakistan. The main health dangers include cancer of different body parts such as skin, lungs and bladder, and many other neurological, haematological, renal, and respiratory illnesses (Yadav et al., 2017). Arsenic also has a hazardous impact on plants resulting in low biomass production, chlorosis, necrosis, leaf senescence, defoliation, stunted growth, chlorophyll degradation and low yield (Ansari et al., 2013). The reduced plant growth is due to the production of reactive oxygen species (ROS) such as superoxide and hydrogen peroxide. These ROS result in oxidative stress toward biomolecules such as lipids and protein and reduction in antioxidant enzymes such as ATP, SOD etc. (Keshavkant et al., 2012). These ROS not only have a negative impact on the metabolism of starch and sugars in plant cells but also affect the DNA by replacing phosphorous group and the protein functioning through binding of As (III) by sulfhydryl groups, ultimately resulting into the death of plant cell (Talukdar et al., 2013; Shahid et al., 2014; Singh et al., 2016; Singh et al., 2017; Shahid et al., 2017). However, during the recent years, use of different amendments including the biochar and dicalcium phosphate has been reported to reduce the toxicity of different heavy metals including arsenic, copper, nickel and lead (Brennan et al., 2014; Turan, 2019; Turan, 2020). Arsenic has also been reported as a contaminant in the most of the groundwater water resources existing in Pakistan (Shakoor et al., 2015; Shahid et al., 2017; Shahid et al., 2018). Irrigating the agricultural crops with such contaminated groundwater contaminates the soils and might affect the growth, physiology and productivity of agricultural crops (Abbas et al., 2018). Hence, there is need to study the effects of arsenic on agricultural crops sown in varying agricultural soils in Pakistan.
Maize is one of the major agricultural crops which is intensively cultivated in various countries including the United States of America. China and Pakistan (Ashraf et al., 2016). In Pakistan, the maize is the 3rd largest crop and is grown on 1130 thousand hectares with annual yield of 4695 thousand tonnes (GOP, 2014-15). The arsenic might be affecting the growth and physiology of this crop in varying agricultural fields of Pakistan. Hence, the current study was designed to examine the effect of Arsenic (As) on physiological, biochemical, and morphological characteristics of maize grown on soils of varying texture i.e. clayey, loamy and sandy loam soils.
MATERIALS AND METHODS
Experimental details and treatments
Soil collection and characterization: Clayey, loamy and sandy loam soils were collected from Gujranwala (32.2439° N, 74.2975° E), Sahiwal (30.6522° N, 72.9691° E) and River Ravi near Sahiwal (30.9539° N, 73.2343° E), respectively. The soil samples were dried in air and sieved through 2.0 mm sieve in the research laboratory of the Department of Environmental Sciences & Engineering, Government College University, Faisalabad. Different physicochemical characteristics of soil were determined by adopting standard methods. The texture of all three soils was determined by the Technique of Gee and Bauder (1986) using the hydrometer. The soil pH and EC were measured by adopting procedure of Bigham (1996). The cation exchange capacity (CEC) and organic matter were measured by following the protocol described by Rhoades (1983) and Walkley-Black method (Jackson, 1962), respectively. The bioavailable As was determined through the method described by Mehlich (1984) and total As by the method of Hudson-Edwards et al., (2004) and available and total phosphorus by following the protocol of Olsen (1954) and Sommers and Nelson (1972), respectively. The CaCO3 contents in soil were determined by the method of Allison and Mooddie (1965). The Iron, Aluminium and Manganese were determined using DTPA method as described by Lindsay and Norvell, (1978). The measured physical and chemical characteristics of three type of soils are given in Table 1.
Soil spiking with As: Three-levels of Arsenic 25, 50 and 100 mg kg-1 of soil were set in all three soils by using Na2H4AsO5 salt. For this purpose, the dried and sieved soils were spiked with a solution of Na2H4AsO5 to obtain three As treatments for each soil (Table 2). A small amount of soil was taken for each treatment and As solution was mixed with it thoroughly in a plastic container. The spiked soil was then thoroughly mixed with other soils in specific proportions to obtain the required levels.
Pot experiment: After spiking with As, the soils were kept in dark at 25 °C temperature for 7 days. Before incubation, soil was moistened to 65 % water holding capacity (WHC). After that 3 kg soil was put in each plastic pot. Total 36 pots were used for this study which comprised of four levels of arsenic (0, 25, 50 and 100 mg As kg-1 of soil) and three types of soil (Sandy loam soil, loamy soil and clayey soil) with three replicate for each. The pots were arranged according to a completely randomized design in an open wire house of Government College University Faisalabad. The maize seeds of Monsanto 6103 hybrid variety were obtained from the market and 4 (four) maize seeds were sown in each pot. The balanced plant fertilizer [Grow Fertilizer (18-18-18)] (Shahbaz et al., 2018) was applied to plants after two weeks of germination. After 60 days of germination the plants were harvested. Before harvest, plants height was measured with the help of measuring tape. Other growth parameters including shoot fresh weight, root fresh weight was measured after the harvesting of the plants.
Measurements of physical and biological parameters of maize plant: The Hiscox and Isrraelstam (1979) protocol was used to determine the chlorophyll a (Chl-a) and chlorophyll b (Chl-b) contents in the leaves of maize plant after 60 days of germination. In order to measure Chl-a and Chl-b, a homogenized mixture was prepared by using one-gram fresh leaf and methanol chloroform water in the ratio 12:5:3 and the contents of Chl-a (at 664.5 nm) and Chl-b (647.4 nm) were estimated using a UV/Visible Spectrophotometer (Hitachi, Berkshire, UK).
Table 1: Physicochemical properties of soils used in study.
|
Soil properties
|
Soil texture
|
| |
Sandy Loam
|
Loamy
|
Clayey
|
|
Physical characteristics
|
|
|
|
|
Sand (%)
|
67.1
|
36.9
|
13.9
|
|
Silt (%)
|
20.2
|
35.7
|
15.7
|
|
clay (%)
|
12.7
|
27.4
|
70.4
|
|
Saturation percentage
|
22.1
|
29.9
|
36.1
|
|
Chemical characteristics
|
|
|
|
|
Soil organic matter (%)
|
0.19
|
0.58
|
0.69
|
|
Soil pH
|
7.23
|
7.85
|
7.90
|
|
Cation Exchange capacity [Cmol (+) kg-1]
|
5.90
|
21.9
|
39.9
|
|
Electrical conductivity (dS m-1)
|
0.22
|
0.49
|
0.59
|
|
Iron (g kg-1)
|
7.23
|
11.4
|
15.9
|
|
Aluminium (g kg-1)
|
0.39
|
0.69
|
0.97
|
|
Manganese (g kg-1)
|
0.13
|
0.19
|
0.25
|
|
Calcium carbonate (%)
|
8.9
|
13.2
|
20.8
|
|
Available Phosphorus (mg kg-1)
|
2.99
|
4.85
|
7.94
|
|
Total Arsenic (mg kg-1)
|
1.15
|
1.95
|
2.95
|
|
Water soluble Arsenic (mg kg-1)
|
0.31
|
0.15
|
0.07
|
Table 2: Three level of As 25, 50 and 100 mg kg-1 of soil set in all three soils by using Na2H4AsO5 salt
|
Treatments
|
Clay
|
Loam
|
Sandy Loam
|
|
Control
|
NAC
|
NAC
|
NAC
|
|
25 mg Kg-1 of Soil
|
LAC
|
LAC
|
LAC
|
|
50 mg Kg-1 of Soil
|
MAC
|
MAC
|
MAC
|
|
100 mg Kg-1 of Soil
|
HAC
|
HAC
|
HAC
|
Treatments realized in the pot experiment. NAC=CONTROL, LAC =25mg Kg-1, MAC =50 mg Kg-1 and HAC =100 mg Kg-1
Soil & plant analyses: A sharp scissor was used to harvest the maize plants near the soil surface after 60 days of germination. The plants were divided to shoots and roots biomass. Soil was taken from each pot and it was air dried. The pH of the soil samples was measured with a standardized pH meter by using a soil suspension (1:5 soil/de-ionized water) well shaken for 1 hour before measuring the pH. The Atomic Absorption Spectrophotometer (AAS) was used to measure the DTPA extractable As of the soils by following the standard procedure as already described by Hudson-Edward et al., (2004).
The harvested plant shoots and roots biomass were rinsed with tap water to remove any dust particles followed by oven drying at 80°C for 48 hours. After getting constant weight, dry shoot weights and dry root weights of the samples were determined. The plant biomass was ground to 0.5 mm by using grinding mill (IKAWerke, MF 10 Basic, Staufen, Germany). After grinding, a diacid mixture of HNO3 and HCLO4 at the ratio 2:1 was used to digest the plant biomass as prescribed by Jones and Case (1990) and this digested plant biomass was used on AAS to measure the As contents.
Determination of reactive oxygen species such as malondialdehyde (MDA), hydrogen peroxide (H2O2) and O2- in maize leaves was carried out by using standard protocols of Jambunathan (2010), Velikova et al., (2000) and Auclair and Voisin (1985) respectively. The MDA contents were determined by using a sample of fresh leaf measuring 500 mg following the procedure as described in Shahbaz et al. (2018) after 60 days of germination.
Similarly, the method recommended by Velikova et al. (2000) was used to determine the contents of H2O2 on spectrophotometer by adopting the protocols as described in Shahbaz et al. (2018). Similarly, the Auclair and Voisin (1985) procedure was followed to determine the concentrations of O2- in maize leaves at 25 °C and 7.0 pH by following the procedure as mentioned in Shahbaz et al. (2018).
The antioxidants enzymes activities were determined in 500 mg fresh maize leaves. A supernatant solution was obtained from the maize leaves and the activities of antioxidant enzymes were determined as already described by Shahbaz et al. (2018). The procedure reported by Roth and Gilbert (1984) was followed to measure the activity of superoxide dismutase (SOD) in maize leaves on spectrophotometer at 420 nm by using one mL reaction mixture. The reaction mixture was prepared adopting the method as described in Shahbaz et al. (2018). Similarly, the activities of CAT and APX were estimated following the procedures of Cakmak and Marschner (1992) and Nakano and Asada (1981), respectively, as described in Shahbaz et al. (2018). An extraction coefficient at 40 mM-1 cm-1 was used for the determination of specific activity of APX in the reaction mixture.
A protein-dye binding procedure as recommended by Bradford (1976) was adopted to measure the protein contents as described in Shahbaz et al. (2018).
Statistical analysis: Three replicates for each treatment were used in this pot study under complete randomized design (CRD). Two-way ANOVA, with As treatment levels and soil types as two factors, was used to interpret the results of study by using Statistic 8.1 ® (Analytical Software, Tallahassee, USA) and LSD test recommended by Steel et al., 1997 was adopted to measure the significant variance among the treatments with means P < 0.5.
RESULTS
Treatment effects on Growth, Physiological parameters and Biological Compounds of maize plant: The heights of plants ranged from 53.9 to 74.4 cm in clay, 75.0 to 50.5 cm in loam and 73.6 to 42.7 cm in sandy loam soil (Fig. 1). The plant height reduced by 8%, 17%, 30%; 13%, 20%, 33% and by 19%, 28% and 42% in clayey, loamy and sandy loam soil respectively as compared with control at low, medium and high level of As. The most significant stress of As on plant height was noted in sandy loam soil where the highest concentrations of As was used (Fig.1).

Fig. 1: The plant height (a), reactive water contents (RWC) (b), root fresh weight (c), root dry weight (d), shoot fresh weight (e), shoot dry weight (f), Chl-a contents (g) and Chl-b contents (h) in maize at various level of Arsenic. The means with same letters(s) are Non-Significant.
The shoot fresh and dry weights were in the range from 9.7 to 14.3 and 1.7 to 3.5, 8.7 to 13.7 and 1.6 to 3.6, 8.1 to 12.9 and 1.3 to 2.9 g pot-1, respectively, in clayey soil, loamy soil and sandy loam soil (Fig. 1). The fresh and dry shoot weights reduced by 16%, 23% & 33%, and 24%, 43 % & 51% in clayey soil, 17%, 24% & 36%, and 24%, 42% & 49%, respectively, in loamy soil and by 19%, 26 % & 37%, and 30%, 40% & 53% in sandy loam soil as compared with control at low, medium and high levels of As, respectively.
The fresh and dry weights of roots were found in the range from 8.3 to 5.8 and 2.1 to 1.6 g pot-1 in clay, 7.8 to 5.4 and 2.1 to 1.5g pot-1 in loam and 7.5 to 4.6 and 1.9 to 1.2 g pot-1 in sandy loam (Fig. 1). The fresh and dry root weight reduced by 14%, 22% & 30%, and 14 % 18 % & 24 % in clayey soil, 16%, 23% & 32%, and 14%, 19% & 24%, respectively, in loamy soil and by 21%, 27% & 36% and 18%, 24% & 37% respectively in sandy loam soil from control at low, medium and contents of As respectively.
The contents of chl-a, chl-b, were in the range of 2.7 to 1.4 and 2.1 to 1.1 mg g-1 fresh weight, 2.6 to 1.1 and 1.9 to 0.9 mg g-1fresh weight and 2.4 to 0.9 and 1.9 to 0.6 mg g-1 FW respectively in clayey soil, loamy soil and sandy loam soil in response to low, medium and high As application (Fig. 1). The chl-a, chl-b reduced by 11%, 28% & 46%, and 11%, 34 % & 49% respectively in clayey soil, 15%, 32% & 56%, and 12%, 34% & 55%, respectively in loamy soil and by 8 %, 37 % & 63 %, and 12 %, 39 % & 66 %, respectively, in sandy loam soil as from control at low, medium and concentrations of As in maize leaves. Maximum reduction in chl-a and chl-b was noted in sandy loam soil at high level of As.
The low, medium and high As concentrations in three different textured soils significantly reduce the RWC as illustrated in Fig. 1. The RWC was in range from 88.2 to 69%, 87 to 66% and 86 to 62% in clayey soil, loamy soil and sandy loam soil, respectively. The RWC contents reduced by 7%, 13% and 22% in clay soil, 8%, 17% and 24% in loamy soil and by 8%, 18% and 28% in sandy loam soil as compared to control at low, medium and high levels of As, respectively. The maximum reduction in RWC was detected in sandy loam soil where the higher As was applied in contrast to the plants grown in control without As.
As concentrations in plant tissues and DTPA-As: Arsenic contents in plant shoots and roots of maize ranged from 0.03 to 34.9 and 0.05 to 81.0; 0.05 to 39.7 and 0.06 to 86.2; 0.06 to 45.0 and 0.08 to 93.3 mg kg-1 of DW of the biomass of maize, in clayey soil, loamy soil and sandy loam soil respectively (Fig. 2). The DTPA-As was found in the ranges of 0.08 to 2.08; 0.03 to 2.21 and 0.03 to 2.38 mg Kg-1 of soil (Fig. 2). The soil textures significantly influenced As uptake and retention in plant biomass. For example, the shoot As content in the plants grown in the soil having 25 mg As kg-1 soil were found to be 7.6, 8.9 and 9.9 mg kg-1 of the plant in clayey, loamy and sandy loam soils, respectively. The shoot As content in the plants grown in the soil containing 50 mg As kg-1 soil were found to be 17.0, 18.9 and 20.5 mg kg-1 of the plant in clayey, loamy and sandy loam soils, respectively. The shoot As content in the plants grown in the soil with 100 mg As kg-1 soil were found to be 34.9, 39.7 and 45.0 mg kg-1 of the plant in clayey, loamy and sandy loam soils, respectively. Similarly, the root As content in the plants grown in the soil containing 25 mg As kg-1 soil were found to be 18.7, 20.5 and 21.9 mg kg-1 of the plant in clayey, loamy and sandy loam soils, respectively. The root As content in the plants grown in the soil containing 50 mg As kg-1 soil were found to be 37.3, 42.1 and 46.2 mg kg-1 of the plant in clayey, loamy and sandy loam soils, respectively. The root As content in the plants grown in the soil containing maximum level of As was found to be 81.1, 86.2 and 93.4 mg kg-1 of the plant in clayey soil, loamy soil and sandy loam soil, respectively.
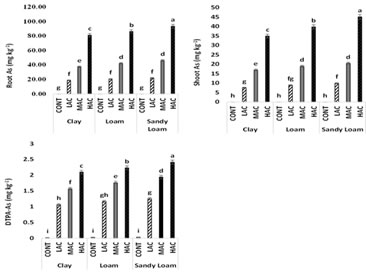
Fig. 2: The effect of various level of As on root As (a) and shoot As (b) in maize and DTPA As (c) in soil. Means having same letter(s) were non-significant p < 0.05).
The DTPA-extractable As contents in the soils spiked with low As concentration were found to be 1.06, 1.16 and 1.25 mg kg-1, at medium As concentrations were found to be 1.56, 1.76 and 1.94 mg kg-1and at highest level were found to be 2.09, 2.23 and 2.41 mg kg-1 of the soil in clayey soil, loamy soil and sandy loam soil, respectively.
Reactive oxygen species (ROS) and antioxidants enzymes: The presence of As in the soil significantly affected the concentrations of reactive oxygen species in maize plants (Fig. 3). The level of ROS increased up to variable extents in different treatments. The levels of MDA, H2O2, and O2- were ranging from 11.7 to 43.8, 13.2 to 52.1 and 6.0 to 32.3 (nmol min-1g-1 FW) in the clay soil, 14.1 to 48.2, 13.5 to 55.9 and 7.1-36.6 (nmol-1 min-1g-1 FW) in the loamy soil and 15.4 to 60.8, 13.2 to 52.1and 9.5 to 45.2 (nmol min-1 g-1 FW) in the sandy loam soil in response to low, medium and high As application respectively (Fig. 3). The level of ROS was significantly increased in all the As containing treatments. The contents of MDA increased by 58%, 118%, 217%; 67%, 127%, 228% and 72%, 148% and 244%; H2O2 by 53%, 114%, 212%; 52%, 119%, 213% and 57%, 139% and 226 and O2- by 41%, 101%, 201%; 46%, 111%, 241% and 60%, 131% and 288 % in clay, loam and sandy loam soil by low, medium and high As level respectively. Generally, the lowest levels of ROS were observed in clayey soil at low As level and the highest levels were observed in the sandy loam soil at high level of As.

Fig. 3: The impact of different level of arsenic on concentrations of antioxidant enzymes [CAT (a), SOD (b), APX (c)], Reactive Oxygen Species (ROS) [H2O2 (d), MDA (e), O2- (f)], and protein (g) in maize.
The activities of catalase, superoxide dismutase and ascorbate peroxidase and protein in fresh maize leaves ranged from 17.4 to 25.9, 23.9 to 70.1, 0.11 to 0.55 (µmole min-1 mg-1 protein) and 850.3 to 420.7 (µg g-1 FW) respectively in clay soil. They were 16.3 to 50.9, 22.9–67.7, 0.05–0.47 (µmole min-1 mg-1 protein) and 835.7 to -422.7 (µg g-1 FW) on loamy soil whereas they were 15.6 to 47.3, 22.0 to 60.4, 0.42 to 0.05 (µmole min-1 mg-1 protein) and 822.7 to 490 (µg g-1 FW) respectively in sandy loam soil. Application of all treatments in soil significantly reduced the activities of catalase, ascorbate peroxidase and superoxide dismutase and the contents of protein in maize leaves (Table 2). Interestingly, considerably the abnormal reduction in the activities of ascorbate peroxidase, superoxide dismutase and catalase and protein contents in maize leaves have been observed in sandy loam soil at the high level of As followed by loamy soil where the minimum activities were found in control treatment. Though, the As toxicity significantly affected the activities of antioxidant enzymes such as CAT by 311%, 237%, 149%; 312%, 230%, 141% and 303%, 210% and 110%; SOD by 296%, 221, 172%; 274%, 222%, 133% and 275%, 214% and 133%; APX by 146%, 93%, 28%; 133%, 82%, 15% and 131%, 72% and -5% and the contents of protein by -12%, -25%, -49%; -13%, -27%, -49% and -13%, -31% and -60% in Clayey, Loamy, and Sandy Loam soils respectively at low, medium and high level of As over control (Fig. 3).
DISCUSSION
In this study, the soil texture was found to significantly affect the impact of As on growth and physicochemical parameters of maize plant at all levels of As. Under the set conditions, the highest significant negative impacts of As on the maize plants were observed in sandy loam soil followed by the loam soil and then the clay soil. According to the results of this study, the maximum reduction in the plant height as well as the plant biomass of maize were recorded in the plants grown in sandy loam soil at highest level of As. This highest impact of As on plants in sandy loam soil might be due to the greater mobility and bioavailability of As in sandy loam soil which has relatively lower level of Fe and Al hydroxide(s), organic matter and clay contents as compared to loam and clayey soil (Inskeep et al., 2001). The reduction in plant growth parameters in the presence of As stress is in line with the findings of different researchers who already studied the impact of As on the growth of different crops (Ansari et al., 2013; Singh et al., 2016; Niazi et al., 2017). The As stress might have affected the metabolism of plant cells and the metabolic energy should have been used for the production of antioxidants and phytochelatins to cope with As stress resulting into a decrease in plant height (Ansari et al., 2013; Singh et al., 2016; Niazi et al., 2017). The As stress has also been reported to result in tissue loss and penetrability, reduction in enzymatic activities, decrease in mitotic activity, reduced cell elongation and reduced turgor of plant cell at higher level of toxicity, which results in decreased plant biomass (Gomes et al., 2013). The similar reasons for decrease in plant height and biomass in response to As stress have also been invoked by other researchers in different plants such as wheat, rice, broad bean, velvet grass and Arabidopsis thaliana (Ansari et al., 2013; Shahid et al., 2015; Khalid et al., 2017). The impact of As on the growth of the maize crop is also comparable with the impacts of different other metal ions on the growth and physiology of various other crops (Khan et al., 2019; Shahbaz et al., 2019). For example, Shahbaz et al., (2019) reported the loss of wheat biomass due to nickel and Khan et al., (2019) reported the loss of barley biomass due to Pb stress.
The elevated concentration of As significantly reduced the chl-a and chl-b and RWC contents in all three types of soils with maximum decrease in sandy loam soil at the high level of As. Our results appear to be well substantiated with the findings of other researchers who reported negative impact of As on chl-a and chl-b contents in other crops including B. napus and B. juncea (Niazi et al., 2017), Zea mays L (Anjum et al., 2017; Mehmood et al., 2017), Vigna mungo L. (Srivastava et al., 2017), Boehmeria nivea L (Mubarak et al., 2016) and Oryza sativa L (Dwivedi et al., 2012).
The metals stress results in lipid and nucleic acid peroxidation and loss of oxidizing protein which increases the production of ROS in plant (Turan et al., 2018). The application of As at various levels significantly increased the contents of ROS and negatively affected the activities of antioxidants in maize crop. Though the most significant impact was observed in sandy loam soil at the highest level of As treatment (Fig. 1). The content of ROS such as MDA, H2O2, and O2-increased by 244, 226 and 288 %, respectively, as compared with control in sandy loam soil at highest level of As. Reportedly, the As toxicity enhanced the concentrations of ROS in different plants such as Boehmeria nivea L (Mubarak et al., 2016), Oryza sativa L (Singh et al., 2017). Similarly, increase in ROS in response to As addition has also been reported in many edible food crops such as wheat (Ghosh et al., 2013), rice (Choudhury et al., 2011), Spinacia oleracea (Shahid et al., 2017) and Pisum sativum (Rafiq et al., 2017). The cell functions of plants are distressed due to unnecessary increase in the contents of ROS in plants due to the damage of metals on nucleic acids, oxidizing proteins, and lipids peroxidation (Abderrahim et al., 2015).
The oxidation processes of lipids and molecules are also significantly affected by metal stress in plants due to inhibition of oxidation chain reactions (Kumar et al., 2012). The activities of antioxidant enzymes such as CAT, SOD, and APX were increased by applying As with maximum increase at As level of 25 mg kg-1 of As but significantly reduced by increasing the level of As in all three soils. However, the maximum change in activities of antioxidants was observed in sandy loam soil at the high contents of As. The protein contents were also reduced by 60% at the high level of As in sandy loam soil. Similar results have already been reported in other crops such as in Cicer arietinum L (Tripathi et al., 2017), Vigna mungo L (Srivastava et al.,2017) and Boehmeria nivea L (Mubarak et al., 2016), whereas, reduced antioxidant activities of enzymes have also been reported in rice (Upadhyay et al.,2016) and Brasssica juncea L (Kanwar et al., 2015). Reduction in protein under As stress is due to the suppression of both nitrate and nitrite reductase enzymes and, ROS results in oxidation of protein and create free carbonyl groups, which results in hindering and altering the activities of protein (Rajjou et al., 2008). Consequently, proteins become more vulnerable to proteolytic attacks (Moller et al., 2007). Similar observations were also reported in other crops like rice (Singh et al., 2009) and maize (Stoeva et al., 2003).
The enhanced concentration of As in soil elevated the concentration of As in plant parts in all three types of soil as compared to control. The accumulation of As was more in the roots as compared to leaves in all types of soils. This order of accumulation of As in plant parts is mostly likely due to the fact that roots are directly exposed to As and As enters through the apoplastic tissue of cell and also due to reduction in transpiration and decreased As fluxes from root to shoot (Silva et al., 2018). The maximum uptake of As in root (93.4 mg kg-1) and shoot (44.03 mg kg-1) as well as the DTPA extractable As in soil (2.41 mg kg-1) was observed in sandy loam soil followed by the loam soil and the least was in clayey soil. This might be due to the maximum mobility and availability of As in the sandy loam soil as compared to other types of soil. Our observations are in line with the findings of other researchers (Quazi et al., 2013; Piracha et al., 2016) also reported the same order of As uptake in soil with different textures. The relatively lower DTPA extractable As in clayey soil was due to the fact that the clayey soil retained more As due to the high clay particle, organic matter, CEC and CaCO3 which might have adsorbed the As making it less available to be extracted with DTPA (Quazi et al., 2013; Masindi et al., 2014; Piracha et al., 2016; Uddin et al., 2017). The soil characteristics such as texture, organic matter content, Fe and Al (hydro) oxides, CEC and clay particles have already been found to significantly alter the fate of As in soil (Taggart et al., 2009; Simmler et al., 2016; Azeem et al., 2017; Strawn 2018).
Conclusions: On the basis of the results of the present study, it can be concluded that As significantly affects the growth and physiology of the maize crop. However, the toxic effects of As are influenced by the soil texture with the severe effects in the sandy loam soil as compared to the loamy and clayey soils with least effects in clayey soil. Moreover, the defence system of plant was also significantly affected by As in all textural soils but the plants grown on course textured soil (sandy loam) showed highest reduction in the enzymatic activities and maximum increase in ROS contents and least impact was observed in fine texture soil (clayey soil). The maximum negative impact of As on plant growth, biomass, and physiological parameters were noted in plants grown in sandy loam soil and least was in the plants grown in clayey soil. The least concentrations of DTPA As was noted in clayey soil due to the greater retention sites. It was suggested that textures of soil are important factors to control the mobility, bioavailability and uptake of As by the plant.
Acknowledgements: We acknowledge the help provided through the research funding by Government College University Faisalabad
Acronyms:
As. Arsenic.
LAC Low arsenic level
MAC Medium arsenic level
HAC High arsenic level
ROS Reactive Oxygen Species
ATP Adenosine triphosphate
SOD. Super Oxide Dismutase
CAT Catalase
APX Ascorbate peroxidase
MDA Malondialdehyde
H2O2 Hydrogen peroxide
REFERNCES
- Abbas, G., B. Murtaza, I. Bibi, M. Shahid, N.K. Niazi, M.I. Khan and M. Hussain (2018). Arsenic uptake, toxicity, detoxification, and speciation in plants: physiological, biochemical, and molecular aspects. Int. J. Environ. Res. Public Health. 15(1): 59.
- Abderrahim, F., E. Huanatico, R. Segura, S. Arribas, M.C. Gonzalez and L. Condezo-Hoyos (2015). Physical features, phenolic compounds, betalains and total antioxidant capacity of coloured quinoa seeds (Chenopodium quinoa Wild.) from Peruvian Altiplano. Food Chem. 183: 83-90.
- Allison, L.E. and C.D. Moodie (1965). Carbonate. Methods of Soil Analysis: Part 2 Chemical and Microbiological Properties. 9: 1379-1396.
- Anjum, S.A., M. Tanveer, S. Hussain, U. Ashraf, I. Khan and L. Wang (2017). Alteration in growth, leaf gas exchange, and photosynthetic pigments of maize plants under combined cadmium and arsenic stress. Water Air Soil Pollut. 228(1): 13.
- Ansari, M.K.A., H.B. Shao, S. Umar, A. Ahmad, S.H. Ansari, M. Iqbal and G. Owens (2013). Screening Indian mustard genotypes for phytoremediating arsenic‐contaminated soils. Clean (Weinh). 41(2): 195-201.
- Ashraf, U., M.N. Salim, A. Sher, S.U.R. Sabir, A. Khan, S. Pan and X. Tang (2016). Maize growth, yield formation and water-nitrogen usage in response to varied irrigation and nitrogen supply under semi-arid climate. Turkish J. Field Crop. 21(1): 88-96.
- Auclair, C. and E. Voisin (1985). Nitroblue tetrazolium reduction. Handbook of methods for oxygen radical research. 123 p.
- Azeem, W., M. Ashraf, S.M. Shahzad, M. Imtiaz, M. Akhtar and M.S. Rizwan (2017). Phosphate-arsenate relations to affect arsenic concentration in plant tissues, growth, and antioxidant efficiency of sunflower (Helianthus annuus L.) under arsenic stress. Environ. Sci. Pollut. Res. 24(31): 24376-24386.
- Bigham, J.M., U. Schwertmann and G. Pfab (1996). Influence of pH on mineral speciation in a bioreactor simulating acid mine drainage. J. Appl. Geochem. 11(6): 845-849.
- Bradford, M.M. (1976). A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72(1-2): 248-254.
- Brennan, A., E.M. Jiménez, M. Puschenreiter, J.A. Alburquerque and C. Switzer (2014). Effects of biochar amendment on root traits and contaminant availability of maize plants in a copper and arsenic impacted soil. Plant Soil. 379(1-2): 351-360.
- Cakmak, I. and H. Marschner (1992). Magnesium deficiency and high light intensity enhance activities of superoxide dismutase, ascorbate peroxidase, and glutathione reductase in bean leaves. J. Plant Physiol. 98(4): 1222-1227.
- Choudhury, B., S. Chowdhury and A.K. Biswas (2011). Regulation of growth and metabolism in rice (Oryza sativa L.) by arsenic and its possible reversal by phosphate. J. Plant Interact. 6(1): 15-24.
- Dwivedi, S., A. Mishra, P. Tripathi, R. Dave, A. Kumar, S. Srivastava and R.D. Tripathi (2012). Arsenic affects essential and non-essential amino acids differentially in rice grains: inadequacy of amino acids in rice-based diet. Environ. Int. 46: 16-22.
- Gee, G. W. and J.W. Bauder (1986). Particle‐size analysis. Methods of soil analysis: Part 1 Physical and mineralogical methods. 5: 383-411.
- Ghosh, S., J. Saha and A,K. Biswas (2013). Interactive influence of arsenate and selenate on growth and nitrogen metabolism in wheat (Triticum aestivum L.) seedlings. Acta Physiol. Plant. 35(6) : 1873-1885.
- Gomes, M.P., M. Carvalho, G.S. Carvalho, T.C.L.L.S.M. Marques, Q.S. Garcia, L.R.G Guilherme and A.M. Soares (2013). Phosphorus improves arsenic phytoremediation by Anadenanthera peregrina by alleviating induced oxidative stress. Int. J. Phytoremediation. 15(7): 633-646.
- Government of Pakistan, Economic survey of Pakistan (2014-15) In: Ministry of Food, Agric. and Livestock Division (Economic Wing), Islamabad. 17-23 p.
- Hiscox, J.D. and G.F. Israelstam (1979). A method for the extraction of chlorophyll from leaf tissue without maceration. Can. J. Bot. 57(12): 1332-1334.
- Hudson-Edwards, K.A., S.L. Houghton and A. Osborn (2004). Extraction and analysis of arsenic in soils and sediments. Trends Analyt. Chem. 23(10-11): 745-752.
- Hughes, M.F., B.D. Beck, Y. Chen, A.S. Lewis and D.J. Thomas (2011). Arsenic exposure and toxicology: a historical perspective. Toxicol. Sci. 123(2): 305-332.
- Jackson, M.L. (1962). Soil chemical analysis, constable and Co. Ltd. London. 497 p.
- Inskeep, W.P., T.R. McDermott, S. Fendorf and W. Frankenberger (2001). Arsenic (V)/(lll) cycling in soils and natural waters: Chemical and microbiological processes. Environmental chemistry of arsenic. 183.
- Jambunathan, N. (2010). Determination and detection of reactive oxygen species (ROS), lipid peroxidation, and electrolyte leakage in plants. In Plant stress Humana Press. Tolerance. 291-297 p.
- Jones Jr, J. B. and V.W. Case (1990). Sampling, handling, and analyzing plant tissue samples. Soil testing and plant analysis. 3: 389-427.
- Kanwar, M.K. and R. Bhardwaj (2015). Arsenic induced modulation of antioxidative defense system and brassinosteroids in Brassica juncea L. Ecotoxicol. Environ. Saf. 115: 119-125.
- Keshavkant, S., J. Padhan, S. Parkhey and S.C. Naithani (2012). Physiological and antioxidant responses of germinating Cicer arietinum seeds to salt stress. Russ. J. Plant Physiol. 59(2): 206-211.
- Khalid, S., M. Shahid, N.K. Niazi, B. Murtaza, I. Bibi, and C. Dumat (2017). A comparison of technologies for remediation of heavy metal contaminated soils. J. Geochem. Explor. 182: 247-268.
- Khan, I., M. Iqbal, and F. Shafiq (2019). Phytomanagement of lead-contaminated soils: critical review of new trends and future prospects. Int. J. Environ. Sci. Technol: 1-16.
- Kumar, H., D. Sharma and V. Kumar (2012). Nickel-induced oxidative stress and role of antioxidant defense in barley roots and leaves. Int. J. Environ. Biol. 2(3): 121–128.
- Lindsay, W.L. and W. Norvell (1978). Development of a DTPA soil test for zinc, iron, manganese, and copper 1. Soil Sci. Soc. Am. J. 42(3): 421-428.
- Mandal, B.K. and T.K. Suzuki (2002). Arsenic round the world: a review. Talanta. 58(1): 201-235.
- Masindi, V., M.W. Gitari, H. Tutu and M. De Beer (2014). Application of magnesite–bentonite clay composite as an alternative technology for removal of arsenic from industrial effluents. Toxicol. Environ. Chem. 96(10): 1435-1451.
- Mehlich, A. (1984) Mehlich No 3 soil test extractant: A modification of Mehlich No 2 extractant. Commun Soil Sci. Plant. Anal. 15:1409-1416.
- Mehmood, T., I. Bibi, M. Shahid, N.K. Niazi, B. Murtaza, H. Wang and G. Murtaza (2017). Effect of compost addition on arsenic uptake, morphological and physiological attributes of maize plants grown in contrasting soils. J. Geochem. Explor. 178: 83-91.
- Moller, I.M., P.E. Jensen and A. Hansson (2007). Oxidative modifications to cellular components in plants. Annu. Rev. Plant Biol. 58: 459-481.
- Mubarak, H., N. Mirza, L.Y. Chai, Z.H. Yang, W. Yong, C.J. Tang and W. Nasim (2016). Biochemical and Metabolic Changes in Arsenic Contaminated Boehmeria nivea L. Biomed. Res. Int. 2016.
- Nakano, Y. and K. Asada (1981). Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol. 22(5): 867-880.
- Niazi, N.K., I. Bibi, A. Fatimah, M. Shahid, M.T. Javed, H. Wang and M.B. Shakoor (2017). Phosphate-assisted phytoremediation of arsenic by Brassica napus and Brassica juncea: morphological and physiological response. Int. J. Phytoremediation. 19(7): 670-678.
- Olsen, S.R. (1954). Estimation of available phosphorus in soils by extraction with sodium bicarbonate .US Department of Agriculture. (No. 939).
- Piracha, M.A., M. Ashraf, S.M. Shahzad, A.R. Siddiqui and S. Nazeer (2016). Arsenic behavior in different textured soils amended with phosphate rock and farm yard manure. J. Environ. Agric. 1(1): 55-67.
- Quazi, S., D. Sarkar and R. Datta (2013). Human health risk from arsenical pesticide contaminated soils: a long-term greenhouse study. J. Hazard. Mater. 262: 1031-1038.
- Rafiq, M., M. Shahid, G. Abbas, S. Shamshad, S. Khalid, N.K. Niazi and C. Dumat (2017). Comparative effect of calcium and EDTA on arsenic uptake and physiological attributes of Pisum sativum. Int. J. Phytoremediation. 19(7): 662-669.
- Rajjou, L., Y. Lovigny, S.P. Groot, M. Belghazi, C. Job and D. Job (2008). Proteome-wide characterization of seed aging in Arabidopsis: a comparison between artificial and natural aging protocols. J. Plant Physiol. 148(1): 620-641.
- Rhoades, J. D. (1983). Cation exchange capacity. Methods of Soil Analysis: Part 2 Chemical and Microbiological Properties. 9: 149-157.
- Roth Jr, E.F. and H.S. Gilbert (1984). The pyrogallol assay for superoxide dismutase: absence of a glutathione artifact. Anal. Biochem. 137(1): 50-53.
- Shahbaz, A.K., M. Iqbal, A. Jabbar, S. Hussain, and M. Ibrahim (2018). Assessment of nickel bioavailability through chemical extractants and red clover (Trifolium pratense L.) in an amended soil: Related changes in various parameters of red clover. Ecotoxicol. Environ. Saf. 149: 116-127.
- Shahbaz, A.K., P.M.A. Ramzani, R. Saeed, V. Turan, M. Iqbal, K. Lewińska and M. Fatima (2019). Effects of biochar and zeolite soil amendments with foliar proline spray on nickel immobilization, nutritional quality and nickel concentrations in wheat. Ecotoxicol. Environ. Saf. 173: 182-191.
- Shahid, M., C. Dumat, B. Pourrut, J. Silvestre, C. Laplanche and E. Pinelli (2014). Influence of EDTA and citric acid on lead-induced oxidative stress to Vicia faba roots. J. Soils Sediments. 14(4): 835-843.
- Shahid, M., S. Khalid, G. Abbas, N. Shahid, M. Nadeem, M. Sabir and C. Dumat (2015). Heavy metal stress and crop productivity. In Crop production and global environmental issues Springer, Cham.1-25 p.
- Shahid, M., N.K. Niazi, C. Dumat, R. Naidu, S. Khalid, M.M. Rahman and I. Bibi (2018). A meta-analysis of the distribution, sources and health risks of arsenic-contaminated groundwater in Pakistan. Environ. Pollut. 242: 307-319.
- Shahid, M., M. Rafiq, N.K. Niazi, C. Dumat, S. Shamshad, S. Khalid and I. Bibi (2017). Arsenic accumulation and physiological attributes of spinach in the presence of amendments: an implication to reduce health risk. Environ. Sci. Pollut. Res. 24(19): 16097-16106.
- Shahid, M., T. Xiong, M. Castrec-Rouelle, T. Leveque and C. Dumat (2013). Water extraction kinetics of metals, arsenic and dissolved organic carbon from industrial contaminated poplar leaves. J. Environ. Sci. 25(12): 2451-2459.
- Shakoor, M.B., N.K. Niaz, I. Bibi, M.M. Rahman, R. Naidu, Z. Dong and M. Arshad (2015). Unraveling health risk and speciation of arsenic from groundwater in rural areas of Punjab, Pakistan. Int. J. Environ. Res. Public Health. 12(10): 12371-12390.
- Silva, J.R.R., A.R. Fernandes, M.S. Junior, C.R.C. Santos and A.K.S. Lobato (2018). Tolerance mechanisms in Cassia alata exposed to cadmium toxicity–potential use for phytoremediation. Photosynthetica. 56(2): 495-504.
- Simmler, M., E. Suess, I. Christl, T. Kotsev and R. Kretzschmar (2016). Soil-to-plant transfer of arsenic and phosphorus along a contamination gradient in the mining-impacted Ogosta River floodplain. Sci. Total Environ. 572: 742-754.
- Singh, A.P., G. Dixit, A. Kumar, S. Mishra, N. Kumar, S. Dixit and O.P. Dhankher (2017). A protective role for nitric oxide and salicylic acid for arsenite phytotoxicity in rice (Oryza sativa L.). Plant Physiol. Biochem. 115: 163-173.
- Singh, A.P., G. Dixit, A. Kumar, S. Mishra, P.K. Singh, S. Dwivedi and O.P. Dhankher (2016). Nitric oxide alleviated arsenic toxicity by modulation of antioxidants and thiol metabolism in rice (Oryza sativa L.). Front. Plant Sci. 6: 1272.
- Singh, H.P., S. Kaur, D.R. Batish, V.P. Sharma, N. Sharma and R.K. Kohli (2009). Nitric oxide alleviates arsenic toxicity by reducing oxidative damage in the roots of Oryza sativa (rice). Nitric oxide. 20(4): 289-297.
- Sommers, L.E. and D.E. Nelson (1972). Determination of total phosphorus in soils: a rapid perchloric acid digestion procedure. Soil Sci. Soc. Am. J. 36(6): 902-904.
- Srivastava, S., P. Sinha and Y.K. Sharma (2017). Status of photosynthetic pigments, lipid peroxidation and anti-oxidative enzymes in Vigna mungo in presence of arsenic. J. Plant Nutr. 40(3): 298-306.
- States, J.C. (2015). Arsenic: Exposure Sources, Health Risks, and Mechanisms of Toxicity. John Wiley & Sons.
- Steel, R.G. (1997). Principles and procedures of statistics a biometrical approach (No. 519.5 S8).
- Stoeva, N. and T. Bineva (2003). Oxidative changes and photosynthesis in oat plants grown in As-contaminated soil. Bulg. J. Plant Physiol. 29(1-2): 87-95.
- Strawn, D.G. (2018). Review of interactions between phosphorus and arsenic in soils from four case studies. Geochem. Trans. 19(1): 1-13.
- Taggart, M.A., R. Mateo, J.M. Charnock, F. Bahrami, A.J. Green and A.A. Meharg (2009). Arsenic rich iron plaque on macrophyte roots–an ecotoxicological risk? Environ. Pollut. 157(3): 946-954.
- Talukdar, D. (2013). Arsenic-induced changes in growth and antioxidant metabolism of fenugreek. Russ. J. Plant Physiol. 60(5): 652-660.
- Tripathi, P., P.C. Singh, A. Mishra, S. Srivastava, R. Chauhan, S. Awasthi and R.D. Tripathi (2017). Arsenic tolerant Trichoderma sp. reduces arsenic induced stress in chickpea (Cicer arietinum). Environ. Pollut. 223: 137-145.
- Turan, V., P.M.A. Ramzani, Q. Ali, F. Abbas, M. Iqbal, A. Irum and W.U.D. Khan (2018). Alleviation of nickel toxicity and an improvement in zinc bioavailability in sunflower seed with chitosan and biochar application in pH adjusted nickel contaminated soil. Arch. Agron. Soil Sci. 64 (8): 1053–1067.
- Turan, V. (2019). Confident performance of chitosan and pistachio shell biochar on reducing Ni bioavailability in soil and plant plus improved the soil enzymatic activities, antioxidant defense system and nutritional quality of lettuce. Ecotoxicol. Environ. Saf. 183: 109594.
- Turan, V. (2020). Potential of pistachio shell biochar and dicalcium phosphate combination to reduce Pb speciation in spinach, improved soil enzymatic activities, plant nutritional quality, and antioxidant defense system. Chemosphere. 245: 125611.
- Uddin, M.K. (2017). A review on the adsorption of heavy metals by clay minerals, with special focus on the past decade. Chem. Eng. J. 308: 438-462.
- Upadhyay, A.K., N.K. Singh, R. Singh and U.N. Rai (2016). Amelioration of arsenic toxicity in rice: comparative effect of inoculation of Chlorella vulgaris and Nannochloropsis sp. on growth, biochemical changes and arsenic uptake. Ecotoxicol. Environ. Saf. 124: 68-73.
- Velikova, V., I. Yordanov and A. Edreva (2000). Oxidative stress and some antioxidant systems in acid rain-treated bean plants: protective role of exogenous polyamines. Plant Sci. 151(1): 59-66.
- Yadav, P., B. Singh, V.K. Garg, S. Mor and V. Pulhani (2017). Bioaccumulation and health risks of heavy metals associated with consumption of rice grains from croplands in Northern India. Hum. Ecol. Risk Assess. 23(1): 14-27.
- Zhao, F.J., J.L. Stroud, T. Eagling, S.J. Dunham, S.P. McGrath and P.R. Shewry (2010). Accumulation, distribution, and speciation of arsenic in wheat grain. Environ. Sci. Technol. 44(14): 5464-5468.
|





