CARDIOPROTECTIVE ACTIVITIES OF QUERCETIN AND RUTIN IN SPRAGUE DAWLEY RATS TREATED WITH 5-FLUOROURACIL
E. Sengul1*, V. Gelen2 and S. Gedikli3
1Department of Physiology, Faculty of Veterinary, Atatürk University, Erzurum, Turkey;
2Department of Physiology, Faculty of Veterinary, Kafkas University, Kars, Turkey;
3Department of Histology-Embriology, Faculty of Veterinary, Atatürk University, Erzurum, Turkey
*Corresponding Author’s emails: emin.sengul97@gmail.com; emin.sengul@atauni.edu.tr
ABSTRACT
In this study, the protective effects of Quercetin (Q) and Rutin in 5-fluorouracil (5-FU)-induced cardiotoxicity in rats were investigated and used eighty-piece Sprague Dawley male rats. Rats were divided randomly to eight groups; control, 5-FU, Q50+5-FU, Q100+5-FU, Q100, Rutin50+5-FU, Rutin100+5-FU, Rutin100. The control group was given intragastric (ig) corn oil for 14 days. 5-FU group was injected intraperitoneal (ip) with a single dose of 50 mg/kg. The doses of Q (50 and 100 mg/kg) and Rutin (50 and 100 mg/kg) were administered for 14 days. Rat of the 5-FU-administered groups were sacrificed under anesthesia 72 hours after 5-FU administration. The other groups were sacrificed on the day 15 after the administration. The blood samples were collected from anesthetized rats and sera were isolated from the clotted blood. The cardiac tissues were used for biochemical, histopathological and immunohistochemical analysis. Cardiac enzyme levels were significantly increased in the 5-FU group. Malondialdehyde (MDA) level was significantly higher than control in the 5-FU group and decreased in the Q100+5-FU and Rutin100+5-FU. Superoxide dismutase (SOD) and glutathione (GSH) activities were significantly decreased in the 5-FU group compared to the control, Q100+5-FU and Rutin100+5-FU. 5-FU groups had degeneration of cardiac cells and myofibril. The intensity of β-MHC positivity was higher in the 5-FU group compared to the other groups. In conclusion, this study demonstrates that Q (50 and 100 mg/kg) and Rutin (50 and 100 mg/kg) have protective effects on the 5-FU-induced cardiotoxicity in rats.
Keywords: 5-fluorouracil: Cardiotoxicity: Quercetin: Rutin: Rat.
https://doi.org/10.36899/JAPS.2021.2.0231
Published online October 03,2020
INTRODUCTION
The 5-fluorouracil (5-FU) is an agent with fluoropyrimidine antimetabolite, which is widely used in pancreatic and skin cancer treatment (Malet-Martino et al., 2002). 5-FU is converted to fluorodeoxyuridine monophosphate (FdUMP), fluorodeoxyuridine triphosphate (FdUTP) and fluorouridine triphosphate (FUTP) by a dihydropyrimidine dehydrogenase (DPD) enzyme, and these compounds are active metabolites of 5-FU (Miura et al., 2010). FdUMP and FdUTP cause degradation of DNA synthesis and repair, resulting in DNA damage (Miura et al., 2010; Nagasaki et al., 2010). Hepatotoxicity (Gelen et al., 2018a; Gelen et al., 2017; Ali, 2012), nephrotoxicity (Gelen et al., 2018a; Ali, 2012), gastrointestinal and hematologic toxicities (Gelen and Sengul, 2018b; Lévy et al., 1998) are some of the most common side effects of 5-FU.
Cardiotoxicity, which is well recognized for other antineoplastic agents, is a major complication of 5-FU, and the cardiotoxicity resulting from the use of 5-FU is characterized by angina, pulmonary edema, congestive heart failure, myocardial infarction, ventricular arrhythmias, myocardial ischemia and sudden death (Canale et al., 2006; Jensen and Sørensen, 2012; Polk et al., 2014; Sorrentino et al., 2012; Yuan et al., 2019). The symptom of initiation in 5-FU induced cardiotoxicity appear nearly 12 hrs following the infusion (Becker et al., 1999). Past studies have reported that cardiotoxicity cases have increased due to 5-FU (Polk et al., 2014). The 5-FU’s themselves, or its metabolites, are toxic for myocytes (Ensley et al., 1986) and 5-FU has direct toxic effects on vascular endothelium that have been associated with a decrease of endothelial nitric oxide (NO) synthase, and the reduction of NO synthase leads to coronary spasms and endothelium-independent vasoconstriction via protein kinase C (Chong and Ghosh., 2019; Çalık et al., 2012; Dechant et al., 2012; Tsibiribi et al., 2006). It has been reported that cardiotoxicity is induced the cell damage and apoptosis occur due to increased oxidative stress in cardiomyocytes (Rashid et al., 2014).
To prevent or treat the toxic effects of anticancer agents such as 5-FU, antioxidant compounds are commonly used (Gedikli and Şengül., 2019; Gelen and Şengül., 2020). Antioxidant compounds prevents oxidative stress and oxidative stress mediated tissue damage (Sengul et al., 2019; Rahal et al., 2014a). Quercetin (Q) and Rutin are natural flavonoids and have strong antioxidant effects; they are available in many fruits (Raw apricot %2.54, Apple % 0.17, Weet cherry % 0.18), vegetables (Raw broccoli %3.22, Red pepper %0.17, Red hot chillis pepper %0.22) and herbs (Raw plums % 1.21, Green tea % 10.18, Grape seeds % 10.16) (Gibellini et al., 2011; Manach et al., 1997; Rahal et al., 2014b; Atanassova and Bagdassaria., 2009; Muhammad et al., 2018). Absorption of both Q and rutin from the small intestine of the rat is very good. However, in previous studies, it is stated that rutin is absorbed slower than Q (Carbonaro and Grant., 2005). Several experimental studies have reported that these molecules have anti-ischemic, hypolipidemic, cytoprotective, anti-mutagenic, antioxidants, anti-inflammatories, anti-cancer, anti-proliferative and anti-viral effects (Guo et al., 2007; Jeong et al., 2009; Kelly, 2011; Russo et al., 2012; Yang et al., 2008). It has been suggested that Q and Rutin have prevented toxicity and tissue damage induced by some anticancer agents (Gelen et al., 2017; Şengül et al., 2017). The aim of the present study, in the context of 5-FU-induced cardiotoxicity in rats, was to investigate the possible cardioprotective effects of Q and Rutin.
MATERIALS AND METHODS
Chemicals: 5-FU, Q and Rutin were obtained from Sigma-Aldrich Chemical Company (St. Louis, MO). 5-FU, Q and Rutin doses were determined base on previous studie (Gelen et al., 2017).
Animals: In the study, 80 adult male Sprague Dawley rats were used, whose weights were between 220-250 g. Rats were provided from the Animal Laboratory at the Experimental Research Centre of Ataturk University, Erzurum, Turkey. All the animals were housed under standard environmental conditions and were allowed access to standard diet and ad libitum drinking water. Animal experiments were performed in accordance with the national guidelines for the use and care of laboratory animals and were approved by the Local Ethics Committee of Ataturk University for Animal Experiments (Protocol no: 2017/72).
Experimental Protocol: The rats were randomly divided into eight groups, and there were ten rats in each group. The eight groups are described below.
- Control: The rats were given only intragastric (i.g.) solvent (corn oil) for 14 days.
- 5-FU: The rats were given corn oil for 14 days and single-dose intraperitoneal (i.p.) 5-FU (50 mg/kg) were injected on the 11th day of the study.
- Q50+5-FU: The rats were administered 50 mg/kg doses of Q dissolved in corn oil for 14 days and injected the single dose (50 mg/kg) of 5-FU on the 11th
- Q100+5-FU: The rats were given i.g. 100 mg/kg doses of Q dissolved in corn oil for 14 days and injected with a single dose (50 mg/kg, i.p.) of 5-FU on the 11th
- Q100: The rats were given Q (100 mg/kg, i.g.) for 14 days.
- Rutin50+5-FU: The rats were given i.g. doses of 50 mg/kg of Rutin dissolved in corn oil for 14 days, and a single dose (50 mg/kg, i.p.) of 5-FU was administered on the 11th
- Rutin100+5-FU: The rats were given i.g. doses of 100 mg/kg of Rutin dissolved in corn oil for 14 days, and a single dose (50 mg/kg, i.p.) of 5-FU was administered on the 11th
- Rutin100: The rats were administered Rutin (100 mg/kg, i.g.) for 14 days.
On the 15th day of the experiment, the intracardiac blood samples were taken under sevoflurane (Sevorane liquid 100%, Abbott Laboratory, Istanbul, Turkey) anesthesia from rats, and all the animals were sacrificed.
Sample Collection: The blood samples were transferred into test tubes without an anticoagulant and allowed to clot at room temperature for 15 min. Afterwards, the bloods were centrifuged at 3000 rpm for 10 min at 4◦C, and serum was obtained for the analysis of the cardiac enzymes. The whole hearts were isolated immediately from the animals. The hearts were washed with cold physiological saline and stored at -20◦C.
Cardiac Enzymes Analysis: The sera samples were immediately processed using an auto-analyzer to determine cardiac troponin I (cTnI), lactate dehydrogenase (LDH), alanine transaminase (ALT), aspartate transaminase (AST), creatine kinase (CK) and creatine kinase-MB (CK-MB) parameters.
Analysis of Oxidant and Antioxidants: The cardiac tissues were homogenized in a Teflon-glass homogenizer using a buffer of 1.15% KCl to obtain a 1:10 (w/v) homogenate. The malondialdehyde (MDA) levels in the heart homogenate were measured using the thiobarbituric acid reaction (Placer et al., 1966). The MDA levels were expressed as µmol/g of tissue. The generation of superoxide radicals produced by xanthine and xanthine oxidase, following the reaction of nitro blue tetrazolium and the formation of formazan dye, was used to measure superoxide dismutase (SOD) activity (Sun et al., 1988). SOD activity was measured as the level of inhibition of absorbance at 560 nm and is expressed as U/mg protein. The cardiac glutathione (GSH) content was measured at 412 nm (Sedlak and Lindsay, 1968) and is expressed as µmol/g.
Histopathological Analysis: At the end of the experiment, all rats were sacrificed, and their hearts were removed. After processing the samples with the rutin histological method, heart tissue sections stained with Crossman’s modified Mallory triple staining were examined under a light microscope. For myocardial immunohistochemistry, β-myosin heavy chain (β-MHC) staining was performed. β-MHC positivity was scored as follows: none = -; weak = +; moderate = ++; strong = +++; very strong = ++++.
Statistical Analysis: The all data were analyzed with one-way ANOVA using the SPSS package program. Post-hoc Tukey’s test was used to compare of the parameters among the groups and data were expressed as the mean ± standard deviation (SD). P value less than 0.05 was considered as significant.
RESULTS AND DISCUSSION
Cardiotoxicity Indices: As compared to the other groups, the serum activities of LDH, AST, ALT, CK, CK-MB and cTnI were significantly increased in the 5-FU group (Table 1, a,b, b,c: p<0.01; a,c: p<0.05 n=10). The treatment with both doses of Q and Rutin prevented to increase in the serum activities of these enzymes (Table 1, a,b; b,c: p<0.01; a,c: p<0.05; n=10).
Table 1. The cardiac enzyme parameters in the all groups (n=10).
|
Groups
|
LDH
(U/L)
Mean±SD
|
AST (U/L)
Mean±SD
|
ALT (U/L)
Mean±SD
|
CK-MB
(U/L)
Mean±SD
|
CK
(U/L)
Mean±SD
|
cTnI
( ng/ml)
Mean±SD
|
|
Control
|
771±126a
|
144±45a
|
41±13a
|
749±104a
|
820±270a
|
5±0.28a
|
|
5-FU
|
2688±104b
|
242±38b
|
70±12b
|
1650±119b
|
2443±315b
|
8±0.41b
|
|
Q50+5-FU
|
970±113a
|
146±38a
|
32±17a
|
949±206a
|
1341±164c
|
5±0.1a
|
|
Q100+5-FU
|
771±154a
|
140±42a
|
33±4a
|
755±218a
|
940±14a
|
4±0.34c
|
|
Q100
|
785±85a
|
155±43a
|
46±11a
|
627±219a
|
749±145a
|
5±0.15a
|
|
Rutin50+5-FU
|
921±125a
|
175±25a
|
34±9a
|
1103±104a
|
1428±114c
|
7±0.63b
|
|
Rutin100+5-FU
|
896±86a
|
158±33a
|
37±10a
|
864±98a
|
1388±96c
|
4±0.21c
|
|
Rutin100
|
807±104a
|
156±32a
|
44±14a
|
658±101a
|
869±108a
|
5±0.22a
|
There is a statistical difference between values that are expressed in different letters on the same column.
a,b; b,c: p<0.01 a,c; p<0.05
Oxidative Stress Findings: To assess the effects of Q and Rutin on 5-FU-induced oxidative stress, we analyzed MDA levels, SOD and GSH activities (Figure 1 and 2). In the 5-FU group, MDA levels were higher than all the other groups. The 100 mg/kg dose of the Q and Rutin significantly prevented the increase in MDA, whereas the 50 mg/kg dose of the Q and Rutin was unable to significantly prevent the increase in MDA level (Figure 1A and 2A). The cardiac SOD activity of the 5-FU treated rats was significantly lower than in the control, Q100+5-FU, Q100, Rutin100+5-FU and Rutin100 groups. The SOD activity in the Q50+5-FU and Rutin50+5-FU groups was lower in comparison with the control (Figure 1B and 2B). The GSH levels in the 5-FU group were significantly lower than the control. In the Q100+5-FU and Rutin100+5-FU groups, GSH levels were like the control (Figure 1C and 2C).
Histopatholagical and Immunhistochemical Examination: The myocardial tissues of the control group displayed normal cardiac muscle bundles, without any infarction and degeneration (Figure 3A), whereas the heart tissue of the 5-FU treatment group had significantly degenerated cardiac myofibril and abundant fibrous tissue deposition (Figure 3B). The Rutin100 and Q100 groups had similar histological structures and morphology as compared to the control group (Figure 3C and 3D). For the Rutin50+5-FU and Q50+5-FU treatments in the cardiac myofibers degeneration and collagen fibers deposition was reduced (Figure 3E and 3F). In the Rutin100+5-FU group tissues, a small number of degenerative myofibers were seen, and these tissues exhibited a nearly normal, non-inflammed structure according to the Q100+5-FU group tissues (Figure 3G and 3H).
The intensity of β-MHC positivity was higher in the 5-FU group sections as compared to the control group sections (Table 2, Figure 4A, 4C and 4D). Furthermore, immunopositivity of β-MHC was significantly decreased in the Rutin100+5-FU groups as compared with the Rutin50+5-FU, Q50+5-FU and Q100+5-FU groups (Table 2, Figure 4G, 4H, 4I, 4J).
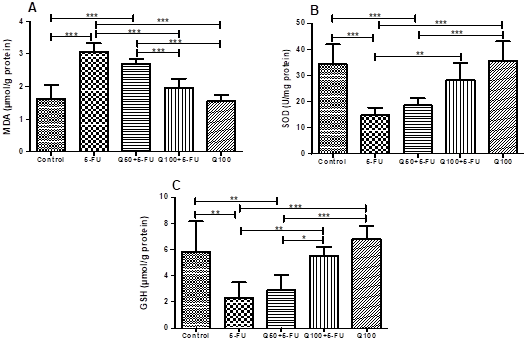
Figure 1. Effects of Q on the oxidative stress parameters in
cardiac tissues of the experimental groups. (MDA (A), SOD (B) and GSH (C), n=10,
***: p<0.001, **: p<0.01, *: p<0.05).
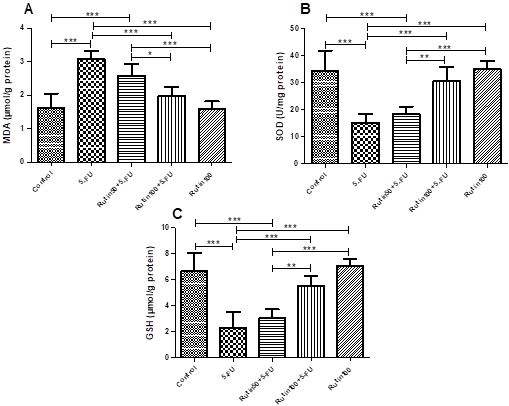
Figure 2. Effects
of Rutin on the oxidative stress parameters in cardiac tissues of the experimental
groups. (MDA (A), SOD (B) and GSH (C), n=10, ***: p<0.001, **: p<0.01, *:
p<0.05).
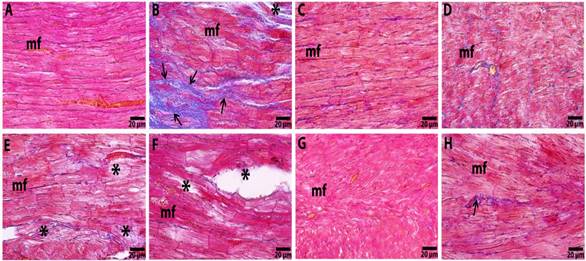
Figure 3. Micrograph
of myocardial section of the Control (A), 5 FU (B), Rutin 100 (C), Q100 (D), Rutin 50+5-FU (E), Q50+5-FU (F), Rutin 100+5-FU (G) and Q100+5-FU (H) groups. mf: cardiac
myofiber; star: myofibrillar degeneration; arrows: fibrous tissue deposition in
the heart. Crossman’s modified Mallory triple staining.
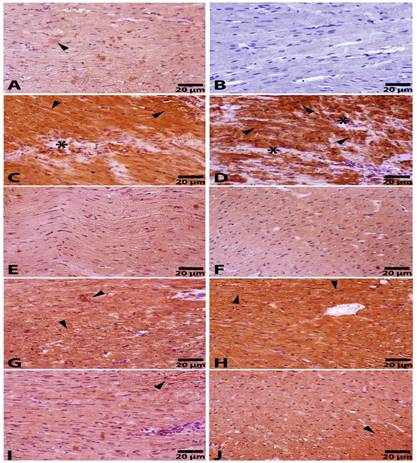
Figure 4. Immunohistochemically
staining for the β-MHC for the Control (A), Negative Control (B), 5-FU (C-D), Rutin100 (E), Q100 (F), Rutin50+5-FU (G), Q50+5-FU (H), Rutin100+5-FU (I) and Q100+5-FU (J) groups. Arrowheads show β-MHC
immunopositivity in cardiac myofibrils; star: degenerative areas; Negative control; not
primer antibody used sections of control and other groups. Streptavidin–biotin peroxidase
staining.
Table 2. Immunohistochemical evaluation scoring of β-MHC antibody (n=10).
|
Experimental Groups
|
β-MHC
|
|
Control
|
+
|
|
5-FU
|
++++
|
|
Q50+5-FU
|
+++
|
|
Q100+5-FU
|
+
|
|
Q100
|
-
|
|
Rutin50+5-FU
|
++
|
|
Rutin100+5-FU
|
+
|
|
Rutin100
|
-
|
The positive cell intensity was scored as follows: none = -; weak = +; moderate = ++; strong = +++; very strong = ++++.
5-FU has been commonly used as a chemotherapeutic in the treatment of gastrointestinal malignancies for years (Jia-Cheng et al., 2016). Unfortunately, as well as its beneficial antitumor effects, 5-FU also has some toxicities, and cardiotoxicity is one of these toxicities (Kosmas et al., 2008). Some flavonoids are widely used in research to prevent or treat the possible side effects of anticancer agents (Gelen et al., 2017; Razavi-Azarkhiavi et al., 2016; Şengül et al., 2017). The Q and Rutin compounds used for this purpose are found in the structure of many fruits and plants (Gibellini et al., 2011; Manach et al., 1997; Rahal et al., 2014b). In this study, the 5-FU-induced cardiotoxicity model in rats determined the possible protective effects of Q and Rutin.
Sera LDH, CK, CK-MB, AST, ALT and cTnI levels are commonly used parameters in the evaluation of cardiotoxicity (Andreadou et al., 2007; Cardinale et al., 2010). These enzymes increase in the case of cardiotoxicity, cardiac injury, heart failure, myocarditis and myocardial infarction. LDH is a specific enzyme released into the blood and cytoplasm during cardiotoxic dysfunction. AST and ALT enzymes are critical transaminases that are released as a result of cardiac metabolism. cTnI, CK and CK-MB enzymes are specific biomarkers in cases of heart failure (Alhumaidhaa et al., 2015; Benstoem et al., 2015; Bertinchant et al., 2003; Viswanatha Swamy et al., 2013). In some studies, it has been reported that 5-FU infusion elevated cardiac enzyme levels (De Forni et al., 1992; Robben et al., 1993). In the isoproterenol-induced cardiac necrosis in rats, it has been reported that cardiac enzyme levels increases and Q administration have reduced these increases (Panda and Kar, 2015). Furthermore, in doxorubicin-induced cardiotoxicity, Rutin administration has significantly inhibited the DOX-induced increase of some cardiac levels (Hozayen and Seif, 2011). In our study, 5-FU administration significantly increased the LDH, CK, CK-MB, AST, ALT and cTnI levels. In the Q and Rutin-administered groups, the increase in the level of these enzymes was inhibited. This is thought to be due to the strong antioxidant properties of Q and Rutin, preventing 5-FU-induced damage in cardiac muscle cells. We are of the opinion that Q and Rutin achieved the lowering of the cardiac enzymes levels by preventing cardiotoxicity.
The production of the oxidant agents is balanced by the antioxidant system. The imbalance between the antioxidant and oxidant system is called oxidative stress. MDA and antioxidant system parameters as GSH and SOD are commonly used parameters for determining oxidative damage (Kara et al., 2016, Preiser, 2012). In this study, the cardiac tissue samples showed decreased SOD and GSH activity and increased MDA level, suggesting oxidative stress was induced by 5-FU. These findings are related to 5-FU-induced toxicity as described in previous study (Rashid et al., 2014). Q and Rutin modifications of these parameters originating from 5-FU are due to the possible antioxidant and anti-inflammatory effects of Q and Rutin. These parameters are an indication that Q and Rutin are protective effects on cardiotoxicity induced by 5-FU, especially in high dose Q and Rutin administrated groups. The effects of Q and Rutin may be due to antioxidant and anti-inflammatory effects of these compounds.
The pathologies (as thickening of the left ventricular wall, myocardial effusion, fibrillation in cardiac myocytes and interstitium) are the histopathologic changes that are seen in cardiotoxicity (Dhesi et al., 2013). The 5-FU single dose demonstrated that massive hemorrhagic myocardial infarction associated with coronary spasms. Besides, the 5-FU treatment caused some changes, such as interstitial hemorrhages, multifocal myofibril necrosis and pericarditis in the heart (Tsibiribi et al., 2006). Our histopathological examinations revealed that the cardiac tissue samples obtained from 5-FU treated rats were degenerated cardiac myofibril and fibrosis free. It was determined that these changes were reduced, especially in the heart tissues obtained from rats in the group of Rutin100+5-FU.
Myocardial hypertrophy and disorganization of cardiac myocytes are generally attributed to an imbalance in the expression of several hypertrophic genes, such as myosin heavy chains (MHC) (Barry et al., 2008). β-MHC is an isoform of MHC that is commonly expressed in ventricles (Liu et al., 2015). β-MHC levels are increased in patients with cardiac injury, left ventricular dysfunction and heart failure (Henderson et al., 2009; Van der Meer et al., 2005). In cyclophosphamide and doxorobicin-induced cardiotoxicity, it was reported that β-MHC mRNA expression has increased cardiac tissue (Liu et al., 2015; Richard et al., 2011). In the Carfilzomib-induced cardiotoxicity, it was determined that the Rutin treatment significantly reduced the β-MHC expression (Imam et al., 2017). Also, in the rats with aortic constriction, the Q administration reduced the cardiac β-MHC expression level (Jalili et al., 2006). In the present study, the β-MHC positivity was higher in the 5-FU-induced cardiotoxicity group sections compared to the control group. Also, immunopositivity of β-MHC was significantly decreased in the Q100+5-FU and Rutin100+5-FU groups. In this way, it was determined that our findings are consistent with the literature.
Conclusion: This study determined that 5-FU induced cardiac injury and Q and Rutin have protective effects againts 5-FU-induced cardiotoxicity. The protective effects of Q and Rutin in the 5-FU-indeced cardiotoxic rat model have been studied for the first time by us. This study will overcome this gap in the literature.
Conflict of Interest: The authors declared that there no conflict of interest.
REFERENCES
- Alhumaidhaa, K.A., D.O. Salehb, M.A. Abd El Fattah, W.I. El-Erakyb, and H. Moawada (2015). Cardiorenal protective effect of taurine against cyclophosphamide-induced toxicity in albino rats. Can. J. Physiol. Pharmacol. 94(2): 131-139.
- Ali, N.E.H.M. (2012). Protective effect of captopril against 5-fluorouracil-induced hepato and nephrotoxicity in male albino rats. J. Am. Sci. 8(2): 680-685.
- Andreadou, I., F. Sigala, E.K. Iliodromitis, M. Papaefthimiou, C. Sigalas, N. Aligiannis, P. Savvari, V. Gorgoulis, E. Papalabros, and D.T. Kremastinos (2007). Acute doxorobicin cardiotoxicity is successfully treated with the phytochemical oleuropein through suppression of oxidative and nitrosative stress. J. Mol. Cell. Cardiol. 42(3): 549-558.
- Atanassova M. and V. Bagdassaria (2009). Rutin content in plant products. J. Univ. Chem. Technol. Metallurgy 44(2): 201-203.
- Barry, S.P., S.M. Davidson, and P.A. Townsend (2008). Molecular regulation of cardiac hypertrophy. Int. J. Biochem. Cell Biol. 40(10): 2023-2039.
- Becker, K., J. Erckenbrecht, D. Haussinger, and T. Frieling (1999). Cardiotoxicity of the antiproliferative compound fluorouracil. Drugs 57(4): 475-484.
- Benstoem C., A. Goetzenich, and S. Kraemer (2015). Selenium and its supplementation in cardiovascular disease-what do we know. Nutrients 7(5): 3094-3118.
- Bertinchant, J.P., A. Polge, J.M. Juan, M.C. Oliva-Lauraire, I. Giuliani, C. Marty-Double, J.Y. Burdy, P. Fabbro-Peray, M. Laprade, J.P. Bali, C. Granier, J.E. de la Coussaye, and M. Dauzat (2003). Evaluation of cardiac troponin I and T levels as markers of myocardial damage in doxorobicin-induced cardiomyopathy rats, and their relationship with echocardiographic and histological findings. Clin. Chim. Acta. 329(1-2): 39-51.
- Canale, M.L., A. Camerini, S. Stroppa, R.M. Porta, P. Caravelli, M. Mariani, A. Balbarini, and S. Ricci (2006). A case of acute myocardial infarction during 5-fluorouracil infusion. J. Cardiovasc. Med. 7(11): 835-837.
- Carbonaro M., and G. Grant (2005). Absorption of quercetin and rutin in rat small intestine. Ann. Nutr. Metab. 49(3): 178-182.
- Cardinale, D., A. Colombo, R. Torrisi, M.T. Sandri, M. Civelli, M. Salvatici, G. Lamantia, N. Colombo, S. Cortinovis, M.A. Dessanai, F. Nolè, F. Veglia, and C.M. Cipolla (2010). Trastuzumab-induced cardiotoxicity: Clinical and prognostic implications of troponin I evaluation. J. Clin. Oncol. 28(25): 3910391-3910396.
- Chong, J.H., and A.K. Ghosh. (2019). Coronary artery vasospasm induced by 5-fluorouracil: Proposed mechanisms, existing management options and future directions. Cardiol. 14(2): 89–94.
- Çalık, A.N., E. Çeliker, Y. Velibey, M. Çağdaş, and Ö. Güzelburç (2012). Initial dose effect of 5-fluorouracil: rapidly improving severe, acute toxic myopericarditis. Am. J. Emerg. Med. 30(1): 257e1-257e3.
- Dechant, C., M. Baur, R. Böck, M. Czejka, A. Podczeck-Schweighofer, C. Dittrich, and G. Christ (2012). Acute reversible heart failure caused by coronary vasoconstriction due to continuous 5-fluorouracil combination chemotherapy. Case. Rep. Oncol. 5(2): 296-301.
- De Forni, M., M. Malet-Martino, P. Jaillais, R.E. Shubinski, J.M. Bachaud, L. Lemaire, P. Canal, C. Chevreau, D. Carrié, and P. Soulié (1992). Cardiotoxicity of high-dose continuous infusion florouracil: A prospective clinical study. J. Clin. Oncol. 10(11): 1795-1801.
- Dhesi, S., M.P., Chu, G. Blevins, I. Paterson, L. Larratt, G.Y. Oudit, and D.H. Kim (2013). Cyclophosphamide-induced cardiomyopathy: A case report, review and recommendations for management. J. Investig. Med. High Impact Case Rep. 1(1): 1-7.
- Ensley, J., J. Kish, E. Tapazoglou, B. Patel, R. Kloner, J. Wynne, and M Al-Sarraf (1986). 5-fluorouracil infusions associated with an ischemic cardiotoxicity syndrome. Proc. Am. Soc. Clitf. Oncol. 5(1): 142.
- Gedikli, S., and E. Şengül (2019). Ratlarda siklofosfamid ile indüklenen hepatotoksisite üzerine kuersetinin etkileri. Dicle Tip Derg. 46(1): 41-50.
- Gelen, V., E. Şengül, S. Yıldırım, and G. Atila (2018a). The protective effects of naringin against 5-fluorouracil-induced hepatotoxicity and nephrotoxicity in rats. Iran. J. Basic Med. Sci. 21(4): 404-410.
- Gelen, V., and E. Şengül (2018b). Hematoprotective effect of naringin on 5-fu toxicity in rats. Chem. Res. J. 3(1):127-130.
- Gelen, V., E. Şengül, S. Gedikli, G. Atila, H. Uslu, and M. Makav (2017). The protective effect of rutin and quercetin on 5-fu-induced hepatotoxicity in rats. APJTB 7(7): 930-934.
- Gelen, V., and E. Şengül (2020) Antioxidant, anti-inflammatory and antiapoptotic effects of Naringin on cardiac damage induced by cisplatin. Indian J. Tradit. Know. 19(2): 459-465.
- Gibellini, L., M. Pinti, M. Nasi, J.P. Montagna, S. De Biasi, E. Roat, L. Bertoncelli, E.L. Cooper, and A. Cossarizza (2011). Quercetin and cancer chemoprevention. Evid. Based Complement. Alternat. Med. 2011: 591356.
- Guo, R., P. Wei, and W. Liu (2007). Combined antioxidant effect of Rutin and vitamin C in Triton X 100 micelles. J. Pharm. Biochem. Anal. 43(4): 1580.
- Henderson, K.K., S. Danzi, J.T. Paul, G. Leya, I. Klein, and A.M. Samarel (2009). Physiological replacement of T3 improves left ventricular function in an animal model of myocardial infarction-induced congestive heart failure. Circ. Heart Fail. 2(3): 243-252.
- Hozayen, W.G., and H.S.A. Seif (2011). Protective effects of rutin and hesperidin against doxorubicin-induced lipodystrophy and cardiotoxicity in albino rats. J. Am. Sci. 7(12): 765-775.
- Imam, F., N.O. Al-Harbi, M.M. Al-Harbia, H.M. Korashy, M.A. Ansari, M.M. Sayed-Ahmed, M.N. Nagi, M. Iqbal, M. Khalid Anwer, I. Kazmi, M. Afzal, and S. Bahashwan (2017). Rutin attenuates carfilzomib-induced cardiotoxicity through inhibition of NF-κB, hypertrophic gene expression and oxidative stress. Cardiovasc. Toxicol. 17(1): 58-66.
- Jalili, T., J. Carlstrom, S. Kim, D. Freeman, H. Jin, T.C. Wu, S.E. Litwin, and J.D. Symons (2006). Quercetin-supplemented diets lower blood pressure and attenuate cardiac hypertrophy in rats with aortic constriction. J. Cardiovasc. Pharmacol. 47(4): 531-541.
- Jensen, S.A., and J.B. Sørensen (2012). 5-fluorouracil-based therapy induces endovascular injury having potential significance to development of clinically overt cardiotoxicity. Cancer Chemother. Pharmacol. 69(1): 57-64.
- Jeong, J.H., J.Y. An, Y.T. Kwon, J.G. Rhee, and Y.J. Lee (2009). Effects of low dose quercetin: Cancer cell-specific inhibition of cell cycle progression. J. Cell Biochem. 106(1): 73-82.
- Jia-Cheng, T., F. Yi-Li, L. Xiao and C. Xiu-Jun (2016). Autophagy in 5-Fluorouracil Therapy in Gastrointestinal Cancer: Trends and Challenges. Med. J. 129(4): 456-463.
- Kara, A., S. Gedikli, E. Sengul, V. Gelen, and S. Ozkanlar (2016). Oxidative stress and autophagy. In free radicals and diseases. InTech. 72-86.
- Kelly, G.S. (2011). Quercetin. Monograph. Altern. Med. Rev. 16(2): 172-194.
- Kosmas, C., M. Kallistratos, P. Kopterides, J. Syrios, H Skopelitis, N Mylonakis, A Karabelis, and N. Tsavaris (2008). Cardiotoxicity of fluoropyrimidines in different schedules of administration: A prospective study. J. Cancer Res. Clin. Oncol. 134(1): 75-82.
- Lévy, E., P. Piedbois, M. Buyse, J.P. Pignon, P. Rougier, L. Ryan, R. Hansen, B. Zee, B. Weinerman, J. Pater, C. Leichman, J. Macdonald, J. Benedetti, J. Lokich, J. Fryer, G. Brufman, R. Isacson, A. Laplanche, E. Quinaux, and P. Thirion (1998). Toxicity of fluorouracil in patients with advanced colorectal cancer: Effect of administration schedule and prognostic factors. J. Clin. Oncol. 16(11): 3537-3541.
- Liu, Y., D. Tan, L. Shi, X. Liu, Y. Zhang, C. Tong, C. Tong, D. Song, and M. Hou (2015). Blueberry anthocyanins-enriched extracts attenuate cyclophosphamide-induced cardiac injury. PloS one 10(7): 1-18.
- Malet-Martino, M., P. Jolimaitre, and R. Martino (2002). The prodrugs of 5-fluorouracil. Curr. Med. Chem. Anticancer Agents 2(2): 267-310.
- Manach, C., C. Morand, C. Demigné, O. Texier, F. Régérat, and C. Rémésy (1997). Bioavailability of rutin and quercetin in rats. FEBS Lett. 409(1): 12-16.
- Miura, K., M. Kinouchi, K. Ishida, W. Fujibuchi, T. Naitoh, H. Ogawa, T. Ando, N. Yazaki, K. Watanabe, S. Haneda, C. Shibata, and I. Sasaki (2010). 5-FU metabolism in cancer and orally-administrable 5-FU drugs. Cancers 2(3): 1717-1730.
- Muhammad F., M. Ahsan and W. Abdul (2018). Quercetin- A mini review. Mod. Concep. Dev. Agrono. 1(2): MCDA.000507.
- Nagasaki, E., A. Takahara, S. Koido, Y. Sagawa, K. Aiba, H. Tajiri, H. Yagita, and S. Homma (2010). Combined treatment with dendritic cells and 5-fluorouracil elicits augmented NK cell-mediated antitumor activity through the tumor necrosis factor-alpha pathway. J. Immunother. 33(5): 467-474.
- Panda, S., and A. Kar (2015). Combined effects of vincristine and quercetin in reducing isoproterenol-induced cardiac necrosis in rats. Cardiovasc. Toxicol. 15(4): 291-299.
- Placer, Z.A., L.L. Cushmanni, and B.C. Johnson (1966). Estimation of products of lipid peroxidation (as malondialdehyde) in biochemical systems. Anal. Biochem. 16(2): 359-364.
- Polk, A., K. Vistisen, M. Vaage-Nilsen, and D.L. Nielsen (2014). A systematic review of the pathophysiology of 5-fluorouracil-induced cardiotoxicity. BMC Pharmacol. Toxicol. 15(1): 47.
- Preiser, J.C. (2012). Oxidative stress. JPEN J. Parenter. Enteral. Nutr. 36(2): 147-154.
46. Rahal, A., A. Kumar, V. Singh, B. Yadav, R. Tiwari, S. Chakraborty, and K. Dhama (2014). Oxidative stress, prooxidants, and antioxidants: the interplay. BioMed Res. Int. 761264.
- Rashid, S., N. Ali, S. Nafees, S.K. Hasan, and S. Sultana (2014). Mitigation of 5-fluorouracil induced renal toxicity by chrysin via targeting oxidative stress and apoptosis in Wistar rats. Food Chem. Toxicol. 66(4): 185-193.
- Razavi-Azarkhiavi, K., M. Iranshahy, A. Sahebkar, K. Shirani, and G. Karimi (2016). The protective role of phenolic compounds against doxorubicin-induced cardiotoxicity: A comprehensive review. Nutr. Cancer. 68(6): 892-917.
- Richard, C., S. Ghibu, S. Delemasure-Chalumeau, J.C. Guilland, C. Des Rosiers, M. Zeller, Y. Cottin, L. Rochette, and C. Vergely (2011). Oxidative stress and myocardial gene alterations associated with doxorobicin-induced cardiotoxicity in rats persist for 2 months after treatment cessation. J. Pharmacol. Exp. Ther. 339(3): 807-814.
- Robben, N.C., A.W. Pippas, and J.O. Moore (1993). The syndrome of 5-fluorouracil cardiotoxicity: an elusive cardiopathy. Cancer 71(2): 493-509.
- Russo, M., C. Spagnuolo, I. Tedesco, S. Bilotto, and G.L. Russo (2012). The flavonoid quercetin in disease prevention and therapy: Facts and fancies. Pharmacol. 83(1): 6-15.
- Sedlak, J., and R.H.C. Lindsay (1968). Estimation of total protein bound and nonprotein sulfhydryl groups in tissue with Ellmann’s reagent. Anal. Biochem. 25(1): 192-205.
- Sorrentino, M.F., J. Kim, A.E. Foderaro, and A.G. Truesdell (2012). 5-fluorouracil induced cardiotoxicity: Review of the literature. Cardiol. J. 19(5): 453-458.
- Sun, Y., W.O. Larry, and L. Ying (1988). A simple method for clinical assay of superoxide dismutase. Clin. Chem. 34(3): 497-500.
- Şengül, E., V. Gelen, S. Gedikli, S. Özkanlar, C. Gür, F. Çelebi, and A. Çınar (2017). The protective effect of quercetin on cyclophosphamide-induced lung toxicity in rats. Biomed. Pharmacother. 92(5): 303-307.
- Tsibiribi, P., J. Descotes, C. Lombard-Bohas, C. Barel, B. Bui-Xuan, M. Belkhiria, A. Tabib, and Q. Timour (2006). Cardiotoxicity of 5-fluorouracil in 1350 patients with no prior history of heart disease. Bull. Cancer 93(3): 27-30.
- Van der Meer, P., E. Lipsic, E., R.H. Henning, K. Boddeus, J. van der Velden, A.A. Voors, D. J. van Veldhuisen, W.H. van Gilst, and R.G. Schoemaker (2005). Erythropoietin induces neovascularization and improves cardiac function in rats with heart failure after myocardial infarction. J. Am. Coll. Cardiol. 46(1): 125-133.
- Viswanatha Swamy, A.H., U.M. Patel, B.C. Koti, P.C. Gadad, N.L. Patel, and A.H. Thippeswamy (2013). Cardioprotective effect of Saraca indica against cyclophosphamide induced cardiotoxicity in rats: A biochemical, electrocardiographic and histopathological study. Indian J. Pharmacol. 45(1): 44-48.
- Yang, J., J. Guo, and J. Yuan (2008). In vitro antioxidant properties of Rutin. LWT - Food Sci. Technol. 41(6): 1060-1066.
- Yuan, C., H. Parekh, C. Allegra, J. George and J.S. Starr (2019). 5-FU induced cardiotoxicity: case series and review of the literature. Cardio-Oncology 5(13): 1572.
|





