|
Volume 31, No. (1), 2021 (February)
(Impact Factor 0.481; JCR 2019) |
|
GENETIC VARIABILITY AND SEASONAL INFLUENCE OF FALL
ARMYWORM (SPODOPTERA FRUGIPERDA (J. E. SMITH)) DAMAGE TO EARLY
VEGETATIVE GROWTH OF AFRICAN MAIZE ACCESSIONS IN THE HUMID TROPICS OF
SOUTH-EASTERN NIGERIA
E. Obok*, M. Ittah, I. Udo, E. Ojubi, J. Ulafor
and S. Umoetok
Crop
Improvement and Protection Unit, Department of Crop Science, Faculty of
Agriculture, Forestry and Wildlife Resources Management, University of Calabar,
Calabar, Cross River State, Nigeria
*Corresponding
author’s email: e.e.obok@unical.edu.ng
ABSTRACT
Fall
armyworm (Spodoptera
frugiperda (J. E. Smith)) (FAW) is a petrifying invasive pest of
maize (Zea mays L.) in Africa. Ten tropical maize accessions from
the International Plant Genetic Resources Institute (IPGRI) collections and a check (Ikom White variety) were studied, under
rainfed conditions, in a multi-environment trial (MET) to assess their
responses to natural infestation of FAW. The study was conducted in the 2018 first
and second maize cropping seasons in Cross River State, Nigeria using a
genotype x season interaction (GSI) model. The combined analyses of variance
showed non-significant difference (p > 0.05) for genotypes and significant (p
≤ 0.05) genotype x season interactions for seedling emergence, plant
height, number of leaves, leaf area and FAW infestation. The most FAW-damaged
maize accession in this study was TZm-304 while TZm-1427 was the least
FAW-damaged maize accession. Overall, FAW infestation was significantly (p ≤
0.01) higher in the second maize cropping season (Akpabuyo) than in first maize
cropping season (Calabar). This subsequently led to corresponding decreases in early
vegetative growth in the second maize cropping season. The IPGRI African maize
accessions evaluated in this study were all susceptible to FAW damage at their
early vegetative growth stages. However, the infestation dynamics of FAW on
maize in Africa is presently being understudied. Thus, there is a dire need for
the adoption of a genotype x location x year (GLY) model in subsequent MET studies
on FAW across all maize-growing States in Nigeria and selection for maize
genotypes in breeding for resistance to FAW should consider early vegetative
growth stage and initial establishments in the field as useful aids to
selection.
Key
words: Fall
armyworm, Ikom White, IPGRI, maize, multi-environment trial, Nigeria.
https://doi.org/10.36899/JAPS.2021.1.0202
Published
online August 26, 2020
INTRODUCTION
Fall
armyworm Spodoptera frugiperda (J. E. Smith) (FAW), a noctuid moth, is a
peril to the production of maize (Zea mays L.) in temperate and tropical
regions of the world. There is a dire need of long-term solutions and
remediations to this deleterious invasive pest, which has presently affected
more than 80 different crops including grasses and legumes (Food and
Agriculture Organization of the United Nations, 2018; Nagoshi et al.,
2018; Prasanna et al., 2018; Toepfer et al., 2018). Though fall
armyworm has its origin in the tropical regions of the Western hemisphere, it
has now become a hostile pest of significant economic importance in African and
Asian countries (Brévault et al., 2018; Chen et al., 2019;
Kalleshwaraswamy et al., 2019; Nagoshi et al., 2017; Nagoshi et
al., 2018; Otim et al., 2018). It was first reported in Nigeria in
2016 (Goergen et al., 2016). Plants of the grass family are predominant
hosts of fall armyworm; maize been one of the most affected cereal.
Maize is a staple in
Central and West African countries; providing food and source of income to
small- and large-scale farmer in these areas. However, there have been concerns
of low soil fertility, drought and biotic stress such as pest and diseases. The
arrival of the fall armyworm, originally from the tropical regions of the
Americas, tends to increase these present concerns. There is presently no
single effective management strategy to FAW. Farmers in Africa are losing their
crops to its attack in quite an alarming rate. In Africa, maize production loss
estimates caused as a result of FAW infestations ranges from 50% to 100% (Baudron
et al., 2019; Rwomushana et al., 2018).
Though there have been
reports on the use of Bt technology (Burtet et al., 2017;
Oliveira et al., 2018), biological (Hay-Roe et al., 2016; Meagher
et al., 2016) and chemical (Burtet et al., 2017; Food and
Agriculture Organization of the United Nations, 2018) control measures for FAW,
these reports have failed to provide the necessary solution needed to match the
geometric crop losses accompanying fall armyworm infestations. Maize crops are
attacked by fall armyworm at larval stages, the most destructive phase in
armyworm lifecycle. However, the adult (males and females) are not destructive
to host crops. The attack is mostly on the young leaves. Though the larvae
could also feed on maize silk and tassel tissues, the ultimate feeding site preference
is the succulent and tender leaf tissues, especially at whorl stages.
Apparently, the mature leaves are classified as unsuitable site for the growth
and development of fall armyworm (Pannuti et al., 2016). The distinct
characteristic mouthpart of fall armyworm larvae encourages rapid defoliation which
often increases with the age of the larvae. The first to third larval instar
causes injuries to maize plant leaves ranging from perforations to
defoliations. Maize leaf stalks and ribs are less likely affected by fall
armyworm. Meanwhile, the critical period for early vegetative growth of maize
(i.e. establishment in the field) is usually between the first four to six
weeks. This growth period coincides with the early period of FAW incidence and infestation
and could lead up to a 20% yield reduction at a larvae-per-plant density of 0.2
to 0.8 (Marenco et al., 1992). Since the first few weeks of maize
establishment in the field overlaps with the early damage to the crop by FAW, especially
the vegetative growth stages, there could be a huge threat capable of
destroying the entire growth and yield potentials of the plants.
Aside from the biotic
pressure of FAW on the growth of maize, seasonal variations could also have
influence on incidence and infestation levels of FAW on the maize crop. In the South
Eastern humid tropics of Nigeria, there are basically two major cropping
seasons for maize production. While the first (early) cropping season usually
commences in mid-March, the second (late) cropping season starts in
mid-September in Nigeria. To the best of our knowledge and available published
literature, this study is the first to look at possible influences that the two
main growing seasons of maize could have on the level of FAW infestation on maize
grown in the humid tropics of Nigeria. Notably, present reports have only
highlighted either the presence or absence of FAW on maize and other related
host crops without giving information on seasonal variations. The present study
was designed to examine the response of different African maize accessions to FAW
damage under natural infestation in the humid tropical agroecology of South Eastern
Nigeria during early and late maize cropping seasons. The variance components
of the early vegetative growth traits were also estimated. The collection of
information is paramount, especially for an invasive pest of such economic
magnitude. From the depth of results obtained from the present study, we are
confident that new vistas for research will arouse interest on in-depth studies
on FAW damage in maize across different maize-growing areas and environments in
Nigeria.
MATERIALS AND METHODS
Ten
tropical maize accessions of diverse African origins were sourced through
International Institute of Tropical Agriculture (IITA), Ibadan, Nigeria, from
Biodiversity International (formerly International Plant Genetic Resources
Institute), Italy (Table 1). Complete descriptors of each of the maize
accessions are available at http://my.iita.org/accession2/ under
the heading ‘Mazie collection’. Ikom White, a white floury maize variety
bred for South Eastern Nigeria (Eijnatten, 1965), was used as a ‘check’. The experiments
were conducted in two seasons in 2018 namely, the Cross River State
Agricultural Development Programme experimental and demonstration farms in
Calabar (N04o 57’ E08o 20’ at an elevation of 35m above
sea level) (i.e. first cropping season) and Akpabuyo (N04o 56’ E08o
23’ at an elevation of 22m above sea level) (i.e. second cropping season).
Table 1. Documented origins of African maize accessions and ‘check’
variety used in the present study.
|
Genotype
|
Reported
African Origin
|
Source
|
|
Accession
|
|
|
|
TZm-109
|
Republic
of Benin
|
Biodiversity
International
|
|
TZm-299
|
Republic
of Chad
|
Biodiversity
International
|
|
TZm-304
|
Republic
of Chad
|
Biodiversity
International
|
|
TZm-378
|
Republic
of Congo
|
Biodiversity
International
|
|
TZm-687
|
Ghana
|
Biodiversity
International
|
|
TZm-1097
|
Unconfirmed
|
Biodiversity
International
|
|
TZm-1153
|
Unconfirmed
|
Biodiversity
International
|
|
TZm-1163
|
Burkina
Faso
|
Biodiversity
International
|
|
TZm-1241
|
Unconfirmed
|
Biodiversity
International
|
|
TZm-1427
|
Republic
of Congo
|
Biodiversity
International
|
|
Check
|
|
|
|
Ikom
White variety
|
Nigeria
|
Cross
River State Agricultural Development Programme
|
The experiments were both carried
out on a 12-month fallow farmland which were not previously planted to either
maize crop or crops documented as hosts to FAW. Six representative soil samples
were randomly collected with an ungalvanized soil auger from each of the experimental
site, at a depth of 15 cm. The samples were bulked according to each block
(replicate) for soil physical and chemical properties analyses at the
Department of Soil Science Analytical Laboratory, University of Calabar,
Calabar. Student t-tests were conducted for soil properties and weather data
obtained from Calabar and Akpabuyo. In the laboratory, wet towel germination
test was conducted to ascertain the viability of the maize seeds. A 100-percent
seed viability was recorded for all the maize genotypes, allowing the planting
of one seed per hole in the field. The first experiment began in the field on
15 March 2018 (i.e. the first cropping season) while the second experiment
commenced on 15 September 2018 (i.e. the second cropping season). Both
experiments were conducted under rainfed conditions. In each of the seasons,
the experiment was laid out in a randomized complete block design (RCBD) with
three replications (i.e. blocks) and each block had eleven (11) treatment plots
representing 11 maize genotypes (i.e. ten tropical maize accessions and one check
variety). The plant spacing was 75cm x 25cm at a seeding depth of 2-3cm.
Seedling emergence was
observed at seven days after sowing (DAS) and expressed as a percentage of the
total number of plants emerged (i.e. visible true leaves above soil level) to the
total number of seeds sown in each treatment plot. For analysis of variance
(ANOVA), data on seedling emergence count was transformed as [log(counts+1)] to
reduce the heterogeneity of variance for seedling emergence. Each treatment
plot size measured 3m x 1.5m, giving a total number of 30 plants per treatment
plot i.e. 53,333 plants/ha. Inorganic fertilizer, NPK 15:15:15, was applied at
the rate of 400kg/ha by ring method at sowing (i.e. 60kgN/ha). Post-emergence
weed control (hand hoeing) was carried out at 14 DAS. In both seasons, fall
armyworm incidence occurred as natural infestations. There were no FAW management
practices carried out i.e. no chemical, biological and/or cultural control
measure was applied in the fields. Scouting and identification of FAW
infestation started at 3-7 days after seedling emergence. All the plants,
especially the leaves, in each of the treatment plots were examined twice daily
– in the morning (07:30) and evening (16:00). At 14 DAS and 28 DAS, the number
of plants naturally infested by fall armyworm were recorded and expressed as a
percentage of the total number of plants per treatment plot. Data on selected
growth traits at the early vegetative stages of the maize plants were collected
at 14 DAS and 28 DAS. These included plant height (PH) (cm), stem width (SW)
(mm), average number of leaves (NL) per plant, leaf area (LA) (cm2)
and leaf area index (LAI) (Elings, 2000).
Combined
ANOVA using GenStat 16.1 (VSN International Ltd., Hemel Hempstead,
England, United Kingdom), were conducted for genotypes over season to estimate
the mean performance and response of the maize genotypes to early vegetative
growth and fall armyworm infestation, respectively. Significant mean
differences were compared using Duncan’s multiple range test (DMRT) (Gomez and
Gomez, 1984). Variance components and expected mean squares for genotypes and
genotype x season interactions were estimated (Kehr and Gardner, 1960; Yassin,
2009). Genotypic coefficient of variability (GCV) and phenotypic coefficient of
variability (PCV) were also calculated (Falconer, 1960). The model used for the
analysis was the genotype x season interaction (GSI) model adopted from Can and
Yoshida (1999). Here, each season was considered as a test environment. Thus,
ANOVA of the RCBD model which assumed independent and heterogeneous error
variance of seasons was carried out to assess mean differences.
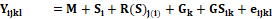
Where: Yijkl
= the measurement on plot l in season i, block j,
containing genotype k; M = the overall mean of all plots in all seasons;
Si = the effect of season i; R(S)j(i) = the
effect of replicate j within season i; Gk = the effect of
genotype k; GSik = the interaction of genotype i
with season k and eijkl = the plot residual. The analysis of variance
for the combined data for the two cropping seasons was as follows, assuming all
factors random:
Table 2. Combined analysis of variance for early vegetaive growth
and FAW infestation.
|
Source of Variation
|
df
|
MS
|
Expected MS
|
|
Season (S)
|
s-1
|
MSS
|
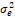 + r + r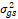 + g + g + r + r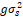
|
|
Replicates within season
|
s(r-1)
|
MSRep(S)
|
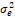 + g + g
|
|
Genotype (G)
|
g-1
|
MSG
|
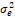 + r + r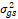 + sr + sr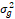
|
|
G x S interaction
|
(s-1) (g-1)
|
MSGS
|
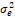 + r + r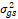
|
|
Pooled error (E)
|
s(r-1) (g-1)
|
MSE
|
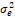
|
s
= season, g = genotype, r = replicate, 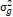 = genotypic variance, = genotypic variance, 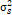 = season variance, = season variance,  = genotype x season interaction
variance, = genotype x season interaction
variance, 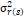 = replicate within
season variance, = replicate within
season variance, 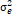 = pooled error variance,
df = degree of freedom, MS = Mean square. = pooled error variance,
df = degree of freedom, MS = Mean square.
RESULTS AND DISCUSSION
Soil
properties and weather conditions in the first and second maize cropping
seasons: Analytical
test results of physical and chemical soil properties at Calabar and Akpabuyo
showed that these soils were loamy sand in texture with low total nitrogen
(0.11 – 0.14%) and slightly acidic to near neutral in pH (5.2 – 6.9). Akpabuyo
soil was richer in available soil phosphorus and potassium ion whereas Calabar
soil had higher organic carbon content which was, however, not significantly
different (p
> 0.05) from Akpabuyo soil. Although lacking in Al3+,
exchangeable cations and effective cation exchange capacity were generally
higher in Akpabuyo soils than in Calabar soils, ranging from 1.49 to 7.20 cmol
kg-1. It was not unusual to record a higher base saturation in
Akpabuyo than in Calabar given the high exchangeable acidity and low
exchangeable bases obtained in this area (Akpan-Idiok, 2012). These
observations were in consonant with soil analysis reports for south-eastern
soils of Nigeria (Akpan-Idiok, 2012; Onwudike, 2015). The weather conditions (WorldWeatherOnline,
2018) were characteristically marked by high rainfall in the second cropping
seasons (mid-September). Overall, these often have implications for high soil
erosion and leaching of soil nutrients, poor fertility status as primary
features peculiar to most soils of the humid tropical rainforest of
south-eastern Nigeria (Akpan-Idiok, 2012). Meanwhile, at the commencement of
the early season planting, rainfall (and rainy days) were lower as the rains
were yet to be fully established by mid-March. The UV index was high, ranging
from 6 to 7 with an accompanying sun hour (sun days) ranging from 155 to 254
hr, respectively. Overall, Akpabuyo and Calabar were not significantly
different (p > 0.05) in terms of UV index, temperature and wind during the
study. The average temperature, relative humidity and air pressure were
comparatively low in the first cropping season during the study. These results
were in consonant with recent yearly weather reports for Akpabuyo and Calabar (Nigerian
Meteorological Agency (NiMET), 2018).
Early
vegetative growth and influence of FAW on African maize
accessions: Seasonal analysis of variance for the early vegetative
growth traits and response to FAW damage (Figures 1,
2 and 3) of all the maize genotypes at Calabar and Akpabuyo showed
significant differences (p ≤). The highest percentage of seedlings
emergence at 7DAS was recorded by accession TZm-1163 (98.15±1.85%) in the
second planting season, at Akpabuyo. Conversely, TZm-1163 had the least number
of seedlings that emerged (33.33±3.2%) at 7DAS sowing in the first planting
season, at Calabar. In both seasons, the check variety, Ikom White, had
an average seedling emergence ranging from 60% to 80% at 7DAS. In the first
cropping season, plant height ranged from 13.15cm (TZm-1163) to 30.8cm (Ikom
White) at 2WAS. Though average plant height in the second cropping season
ranged from 8.5cm (TZm-1427) to 12.3 cm (TZm-1153) at 2WAS, the maize genotypes
were not significantly different (p > 0.05) from the check in height
(8.9±0.2 cm). However, the check variety at 2WAS was not significantly
different (p > 0.05) from TZm-109, TZm-299 and TZm-304 in the first cropping
season. At 4WAS, in both seasons, TZm-304 was significantly (p ≤ 0.05) taller
than all the maize genotypes. Overall, the maize genotypes were taller at both
2WAS and 4WAS in the first planting season (Calabar) than in the second
planting season (Akpabuyo). The average stem width at 2WAS ranged from 10.0±0.6
mm (TZm-1241 at Calabar) to 18.3±1.2 mm (TZm-304 at Calabar); these
observations were not significantly different (p > 0.05) from Ikom White at
Akpabuyo and Calabar, respectively. TZm-1241 had the thickest stems (19.3±0.3
mm) in Akpabuyo and the thinnest stems (12.1±1.0 mm) in Calabar at 4WAS.
Meanwhile, though the accessions differed in stem width, the checks had similar
stem widths (16 mm) and were not significantly different (p > 0.05) from all
the accessions at Calabar and Akpabuyo. The highest number of leaves (6.2±0.4) were
obtained in Akpabuyo (TZm-1163) at 2WAS. TZm-1163 also recorded the lowest
number of leaves (4.2±0.2) in Calabar at 2WAP. Ikom White had
significantly (p ≤ 0.05) lower number of leaves (5.0±0.2) when compared
with TZm-1163 (6.2±0.4) in Akpabuyo at 2WAS. In Calabar, Ikom
White was significantly different (p ≤ 0.05) from TZm-1163, TZm-1097
and TZm-1241 in the number of leaves recorded at 2WAS, but at 4WAS in Calabar,
the check had the highest number of leaves (8.1±0.4) and was statistically
similar (p > 0.05) to other accessions. Remarkably, the number of leaves had
reduced for accessions TZm-109, TZm-1097, TZm-1163, TZm-1241, TZm-1427,
TZm-299, TZm-378 and Ikom White at 4WAS. Accession TZm-304 had the
largest leaf area at 2WAS (505.4±68.5 cm2) and 4WAS (1348.1±201 cm2)
in Akpabuyo. Comparatively, across seasons, larger leaf area was observed for
all the accessions in the second cropping season, except for Ikom White which
also had a lower leaf area (237.5±56.3 cm2) in Akpabuyo than in
Calabar (266.8±46.1 cm2) at 2WAS and vice versa at 4WAS.
TZm-1097 had the lowest
leaf area at 2WAS during the first cropping season whereas in the second
cropping season TZm-1241 recorded the least leaf area. At 4WAS, all the
accessions were not significantly different (p > 0.05) from the check in
terms of leaf area observations at Calabar. A similar trend was observed for
the check at Akpabuyo. The leaf area index (LAI) of the maize genotypes at 2WAS
ranged from 0.25 to 0.65 in Calabar and 0.55 to 1.54 at Akpabuyo. Overall, the
LAI was higher in the second cropping season at 4WAS. Ikom White showed
the highest LAI (1.48±0.2) in Calabar and was not significantly different (p
> 0.05) from TZm-1163 (0.52±0.1) and other maize accessions at 4WAS.
Although TZm-1427 had the lowest LAI (1.76±0.3) at 4WAS in Akpabuyo, this was
not significantly different (p > 0.05) from the check (1.84±0.8). Other
accessions in Akpabuyo had LAI values that were higher than Ikom White and
TZm-1427. When FAW infestation comparisons were made between seasons it was
observed that the mean severity of damage on the maize accessions were
generally higher in the second planting season (Akpabuyo) than in the first
planting season (Calabar) (Table 4), with exception to Ikom White which
recorded low FAW infestation at 2WAS and 4WAS at Akpabuyo. From the combined
ANOVA, TZm-299 recorded the highest seedling emergence (88.89%) while the
lowest percentage of seedlings that emerged at 7DAS was recorded for TZm-1097
(61.11%) which was not significantly different (p > 0.05) from the check
(69.44%) and other accessions except TZm-299, TZm-304 and TZm-687. The tallest
plants at 2WAS were Ikom White (19.9 cm) and were significantly
different (p ≤ 0.05) from accessions TZm-378 (13.1 cm), TZm-1241 (13.3
cm) and TZm-1163 (12.5 cm). At 4WAS TZm-304 was taller (39.5 cm) than Ikom
White (29.1cm) but not significantly different (p > 0.05) from other
accessions. The average stem girth ranged from 11.2 mm to 17.5 mm at 2WAS. Ikom
White recorded 12.5 mm and was not significantly different (p > 0.05)
from other maize accessions except TZm-304 (p ≤ 0.05). When the plants
grew to 4WAS, the trend in stem width differences was similar for the thickest
and thinnest stems, TZm-304 and TZm-1163 accessions, respectively. Combined
ANOVA did not show a significant difference (p > 0.05) for number of leaves
per plant and leaf area index (LAI) at 4WAS (Table 5).
Table 3. Soil properties and weather conditions at Calabar and
Akpabuyo in the 2018 maize cropping seasons.
|
Soil
Properties†
|
Early
Season1
(Calabar)
|
Late
Season 2
(Akpabuyo)
|
t-test
(df = 10)
|
|
Texture
(g kg-1)
|
|
|
|
|
Sand
|
800±3.3
|
839±3.3
|
***
|
|
Silt
|
123±3.0
|
47±3.1
|
***
|
|
Clay
|
77±0.5
|
114±3.7
|
***
|
|
Textural
Class (USDA)
|
Loamy
Sand
|
Loamy
Sand
|
|
|
pH
(in 1:2.5 Soil-H2O ratio)
|
5.2±0.09
|
6.9±0.41
|
***
|
|
Organic
Carbon (%)
|
1.59±0.018
|
1.17±0.237
|
ns
|
|
Total
Nitrogen (%)
|
0.11±0.003
|
0.14±0.006
|
ns
|
|
Available
Phosphorus (mg kg-1)
|
21.16±1.020
|
36.27±0.96
|
***
|
|
Exchangeable
Cations (cmol kg-1)
|
|
|
|
|
Ca2+
|
3.53±0.067
|
7.20±3.160
|
***
|
|
Mg2+
|
1.53±0.065
|
2.07±0.521
|
ns
|
|
K+
|
0.12±0.007
|
1.62±0.128
|
***
|
|
Na+
|
0.06±0.003
|
1.49±0.309
|
***
|
|
Exchangeable
Acidity (cmol kg-1)
|
|
|
|
|
Al3+
|
0.80±0.012
|
0.00±0.00
|
***
|
|
H+
|
1.13±0.067
|
0.60±0.10
|
***
|
|
Effective
Cation Exchange Capacity (cmol kg-1)
|
7.19±0.110
|
12.98±4.120
|
***
|
|
Base
Saturation (%)
|
72.9±0.58
|
94.8±0.73
|
***
|
|
Weather
Conditions††
|
Early
Season1
(Calabar)
|
Late
Season 2
(Akpabuyo)
|
t-test
(df = 10)
|
|
Rainfall
(mm)
|
113.04
(63.06 – 163.01)
|
334.46
(271.92 – 397.00)
|
**
|
|
Rainy
Days
|
25.5
(24 – 27)
|
29.5
(29 – 30)
|
*
|
|
Relative
Humidity (%)
|
77.5
(77 – 78)
|
85.5
(83 – 88)
|
**
|
|
Ultraviolet
Radiation Index
|
6.5
(6 – 7)
|
5.5
(5 – 6)
|
ns
|
|
Ambient
Temperature (oC)
|
26.5
(26 – 27)
|
27
(25 – 29)
|
ns
|
|
Air
Pressure (mb)
|
1010.9
(1010.7 –
1011.1)
|
1011.85
(1012.2 – 1011.5)
|
*
|
|
Sun
Hour (hr)
|
247.3
(234.0 – 253.5)
|
155.8
(155.5 – 156.0)
|
***
|
|
Sun
Days
|
4.5
(4 – 5)
|
0.5
(0 – 1)
|
***
|
|
Wind
(kmph)
|
6.15
(6.0 – 6.3)
|
5.9
(4.6 – 7.2)
|
ns
|
1
15
March 2018 – 15 April 2018; 2 15 September 2018 – 15 October 2018. †Protocols:
Bates (1973); Black et al. (1965); Klute (1986); Murphy and Riley
(1962); Sparks et al. (1996). ††WorldWeatherOnline (2018) –
mean and range (in parenthesis). *, **,
*** and ns imply p ≤ 0.05, ≤ 0.01, ≤ 0.001 and
non-significance, respectively. USDA – United States Department of Agriculture.
Table 4. Early vegetative growth and fall armyworm infestation on African
maize accessions in early and late cropping seasons.
|
Genotype
|
Seedling Emergence (%)
|
Plant Height (cm)
|
Stem Width (mm)
|
Number of Leaves
|
Leaf Area (cm2)
|
Leaf Area Index
|
Fall Armyworm
Infestation (%)
|
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
|
Early Season (Calabar)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TZm-109
|
77.78±3.21abcdefg
|
24.4±6.7abc
|
32.6±9.9bcdef
|
11.7±2.8bc
|
12.8±2.9de
|
5.6±0.5abcd
|
7.3±1.3abc
|
153.2±92.5cde
|
194.4±80.1d
|
0.62±0.5bc
|
0.85±0.5de
|
4.17±2.1ef
|
16.67±2.1d
|
|
TZm-1097
|
51.85±14.46hij
|
17.9±3.0cdefg
|
44.1±6.4abc
|
11.7±1.4bc
|
13.5±1.4cde
|
4.3±0.3fg
|
6.7±0.7abcd
|
50.9±22.6e
|
150.4±31.4d
|
0.60±0.2bc
|
0.56±0.2e
|
10.42±5.5cdef
|
25.00±10.8cd
|
|
TZm-1153
|
68.52±4.90cdefgh
|
20.8±5.0bcde
|
46.3±14.1ab
|
13.0±2.5abc
|
14.6±1.8abcde
|
4.5±0.4efg
|
7.9±0.7a
|
114.5±39.9cde
|
170.1±41.7d
|
0.44±0.3bc
|
0.72±0.2de
|
6.25±6.3def
|
20.83±5.5cd
|
|
TZm-1163
|
33.33±3.2j
|
13.5±1.5defgh
|
27.8±5.8bcdef
|
11.0±0.9bc
|
12.9±1.0de
|
4.2±0.2g
|
6.8±0.3abcd
|
86.0±47.5de
|
144.8±34.7d
|
0.34±0.1c
|
0.52±0.1e
|
2.08±2.1f
|
20.83±4.2cd
|
|
TZm-1241
|
74.07±4.90bcdefgh
|
17.4±3.4cdefgh
|
34.5±3.8abcde
|
10.0±0.6c
|
12.1±1.0e
|
4.3±0.3fg
|
6.7±0.6abcd
|
150.2±47.4cde
|
190.3±36.4d
|
0.25±0.2c
|
0.70±0.2de
|
6.25±0.0def
|
16.67±2.1d
|
|
TZm-1427
|
83.33±3.31abcdef
|
19.8±3.3bcdef
|
37.4±3.5abcd
|
15.3±2.2abc
|
16.2±2.0abcde
|
4.9±0.3cdefg
|
6.8±0.7abcd
|
147.3±63.7cde
|
195.9±51.6d
|
0.45±0.2bc
|
0.74±0.3de
|
12.50±12.5bcdef
|
33.33±13.7bcd
|
|
TZm-299
|
90.74±4.90abc
|
26.7±2.3ab
|
43.6±8.9abc
|
15.4±1.4abc
|
16.2±1.5abcde
|
5.0±0.3bcdefg
|
7.9±0.3a
|
199.5±35.0cde
|
246.8±11.5cd
|
0.45±0.2bc
|
1.04±0.1de
|
14.58±9.1bcdef
|
31.25±3.6bcd
|
|
TZm-304
|
94.44±1.10ab
|
26.8±2.0ab
|
51.1±5.7a
|
18.3±1.2a
|
18.7±1.2ab
|
5.4±0.2abcde
|
8.0±0.4a
|
171.7±70.3cde
|
259.6±43.8cd
|
0.45±0.2bc
|
1.12±0.2cde
|
47.92±5.5a
|
70.83±5.5a
|
|
TZm-378
|
79.63±9.26abcdefg
|
16.7±4.2cdefgh
|
27.8±5.1bcdef
|
13.3±2.7abc
|
14.4±2.3bcde
|
4.8±0.2cdefg
|
7.6±1.2ab
|
90.0±56.2de
|
161.4±47.9d
|
0.39±0.2c
|
0.71±0.3de
|
41.67±14.6abc
|
58.33±17.1ab
|
|
TZm-687
|
88.89±3.21abcd
|
21.2±1.8bcd
|
35.9±6.1abcde
|
15.5±2.9abc
|
16.7±2.6abcde
|
4.7±0.1defg
|
7.6±.06ab
|
83.7±16.0de
|
138.2±22.7d
|
0.41±0.2bc
|
0.56±0.1e
|
20.83±9.1abcdef
|
35.42±9.1bcd
|
|
Ikom White
|
79.63±6.68abcdefg
|
30.8±2.73a
|
43.7±2.5abc
|
14.5±2.0abc
|
16.2±1.1abcde
|
5.3±0.1abcde
|
8.1±0.4a
|
266.8±46.1cd
|
342.2±26.5cd
|
0.65±0.3bc
|
1.48±0.2bcde
|
25.00±12.5abcdef
|
43.75±12.5abcd
|
|
Late Season (Akpabuyo)
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
TZm-109
|
65.74±3.34efgh
|
9.7±1.1gh
|
27.0±1.7cdef
|
16.2±2.3abc
|
18.4±0.7ab
|
5.5±0.4abcde
|
5.3±0.9d
|
498.9±117.0a
|
1056.0±179.0ab
|
1.54±0.5a
|
3.15±1.1ab
|
41.67±11abc
|
52.08±11.0abc
|
|
TZm-1097
|
70.37±4.04cdefgh
|
10.0±1.6gh
|
17.8±2.5ef
|
11.9±0.7 bc
|
17.6±0.2abc
|
6.0±0.4ab
|
5.7±0.4bcd
|
315.5±89.8abc
|
835.0±198.0b
|
1.02±0.3abc
|
2.49±0.6bcde
|
35.42±7.5abcde
|
45.83±8.33abcd
|
|
TZm-1153
|
77.78±5.56abcdefg
|
12.3±0.3efgh
|
18.7±5.0def
|
16.0±1.8abc
|
18.2±0.9abc
|
5.7±0.6abcd
|
5.8±0.6bcd
|
303.7±57.9bc
|
1019.1±266.0ab
|
0.92±0.2abc
|
3.23±1.1ab
|
29.17±2.1abcdef
|
47.92±2.1abcd
|
|
TZm-1163
|
98.15±1.85a
|
11.4±1.1fgh
|
21.3±0.8def
|
13.9±1.9abc
|
17.1±0.5abcd
|
6.2±0.4a
|
5.2±0.2d
|
238.4±45.0cde
|
1066.5±222.0ab
|
0.81±0.2abc
|
3.02±0.8abc
|
43.75±9.6ab
|
52.08±11.0abc
|
|
TZm-1241
|
67.59±1.85defgh
|
9.3±0.2gh
|
17.8±2.9ef
|
12.4±0.7abc
|
19.3±0.3a
|
5.3±0.5abcde
|
5.2±0.1d
|
202.1±41.5cde
|
944.5±21.0ab
|
0.57±0.1bc
|
2.61±0.4bcd
|
37.50±9.6abcd
|
47.92±7.5abcd
|
|
TZm-1427
|
43.52±2.45ij
|
8.5±0.7h
|
13.7±2.1f
|
13.2±0.9abc
|
19.2±0.7ab
|
5.2±0.5abcdefg
|
4.8±0.1d
|
203.2±18.2cde
|
709.8±166.0bc
|
0.55±0.1bc
|
1.76±0.3bcde
|
20.83±12.7abcdef
|
33.33±12.7bcd
|
|
TZm-299
|
87.04±3.34abcde
|
11.3±1.6fgh
|
19.4±3.6def
|
14.1±0.9abc
|
18.5±0.4ab
|
5.8±0.6abc
|
4.8±0.2d
|
274.8±61.5cd
|
897.6±223.0ab
|
0.86±0.2abc
|
2.38±0.7bcde
|
41.67±13.7abc
|
58.33±14.6ab
|
|
TZm-304
|
62.04±6.48fghi
|
11.1±0.7gh
|
28.0±2.6bcdef
|
16.6±1.5ab
|
18.5±1.0ab
|
5.5±0.2abcde
|
6.3±0.8abcd
|
505.4±68.5a
|
1348.1±201a
|
1.46±0.1a
|
4.65±1.1a
|
47.92±15.0a
|
60.42±15.0ab
|
|
TZm-378
|
59.26±1.85ghi
|
9.5±1.1gh
|
23.5±4.4def
|
14.2±1.3abc
|
18.9±0.7ab
|
5.3±0.2abcdef
|
5.0±0.3d
|
323.7±90.2abc
|
1135.0±165ab
|
0.95±0.3abc
|
3.08±0.6abc
|
31.25±12.5abcdef
|
43.75±12.5abcd
|
|
TZm-687
|
68.52±19.40cdefgh
|
10.2±1.6gh
|
22.1±5.8def
|
15.1±2.2abc
|
18.8±0.5ab
|
4.8±0.1cdefg
|
5.4±0.2cd
|
485.0±28.2ab
|
1057.2±235ab
|
1.25±0.1ab
|
3.17±0.9ab
|
37.50±9.6abcd
|
47.92±11.6abcd
|
|
Ikom White
|
59.26±5.16ghi
|
8.9±0.2h
|
14.4±2.3f
|
10.5±1.7bc
|
16.1±0.7abcde
|
5.0±0.2bcdefg
|
4.9±0.3d
|
237.5±56.3cde
|
714.9±302bc
|
0.64±0.2bc
|
1.84±0.8bcde
|
14.58±7.5bcdef
|
20.83±7.5cd
|
WAS
= Weeks after sowing. Mean ± standard error with the same letter(s) in
superscript within the same column in each season are not significantly
different at 95% confidence limit using Duncan’s Multiple Range Test.
The average number of leaves recorded for Ikom
White (5.2) at 2WAS was statistically similar (p > 0.05) to those of
TZm-109 (5.5) and TZm-1241 (4.8), with the highest and lowest number of leaves
per plant, respectively. Average leaf area over the two cropping seasons ranged
from 183.2 cm2 to 338.6 cm2 at 2WAS and 452.8 cm2 to
803.8 cm2 at 4WAS. Though leaf area of accessions TZm-209 and
TZm-304 were higher and significantly different (p ≤ 0.05) from TZm-1097,
TZm-1163, TZm-1241 and TZm-1427 at 2WAS, all maize accessions were not
significantly different (p > 0.05) from the check. The LAI at 2WAS was
literally doubled at 4WAS for all the maize accessions and the check. The range
of LAI obtained was 0.41 to 1.07 2WAS and 1.25 to 2.88 at 4WAS. Ikom White
recorded LAI of 0.64 and 1.66 at 2WAS and 4WAS, respectively. Fall armyworm
(FAW) infestations were progressive and severe over the number of days after
sowing within each season (Table 4) and across the seasons (Table 5). Accession
TZm-304 recorded the highest FAW infestation (47.67%) in Calabar at 2WAS. Overall
FAW infestations were higher at 4WAS in both Calabar and Akpabuyo (Table 4). Also,
though 25% of Ikom White were affected by FAW at 2WAS in Calabar, the
least affected of all the maize genotypes were TZm-1163 (2.08%), TZm-109
(4.17%), TZm-1241 (6.25%) and TZm-1153 (6.25%). Across the two seasons, FAW
infestations recorded for the maize accessions were not significantly different
(p > 0.05) from the check (19.79%) at 2WAS, except (p ≤ 0.05) TZm-304
(47.92%) (Table 5). Although a study was conducted in Ghana that involved
TZm-1153 and TZm-1097 accessions used in the present study, one of the main aim
of that study was to obtain information for the breeding potentials of the
maize accession by assessing their genetic variability, diversity and
relatedness using phenotypic evaluation (Twumasi et al., 2017). However,
of all the 21 agro-morphological traits data obtained from Twumasi et al. (2017),
no reference was made to fall armyworm damage despite the economic significance
of this pest to maize production in Central and West Africa.
Genotype
x season interaction (GSI) model describing early vegetative growth and fall armyworm
damage on African maize accessions in early (Calabar) and late (Akpabuyo) maize
cropping seasons in 2018: The precision and
predictive power of a single season trial is very low and severely biased. To
predict the genotypes’ performance within the target population environments
(TPE), the adoption of a multi-environment trial (MET) is practiced. In this
study, the two maize growing seasons represented two test environments and the
ten (10) African maize accessions and a local maize variety (check) represented
the eleven genotypes. Early vegetative growth traits and FAW recorded higher
mean values at 4WAS (Table 5). From results of the combined ANOVA, there were
no significant differences (p > 0.05) among the genotypes for the traits
studied which could imply that there were either no or nondetectable presence
of genetic variability (Table 6). The non-significant (p > 0.05) mean
squares for genotype confirmed that the trend of growth performance and response
to FAW by these genotypes were in a similar manner in the two cropping seasons.
Estimates of variance components (Table 7) showed that the proportion of
genotypic variance contained in the phenotypic variance for most of the growth
traits were 0%, except for number of leaves per plant (24%) at 4WAS, stem width
(47%) and LAI (25%) at 2WAS and FAW (2%) at 2WAS. This also highlighted the
fact that there was no conspicuous resistance to FAW damage among the maize
accessions and the check, Ikom White, grown in Calabar and
Akpabuyo. There was, however, significant genotype x season interactions (GSI)
for seedling emergence (p ≤ 0.001), plant height (p ≤ 0.05), number
of leaves per plant (p ≤ 0.01), leaf area at 2WAS (p ≤ 0.01) and
FAW damage at 4WAS (p ≤ 0.05) (Table 6). This indicated that the maize
genotypes could apparently exhibit different phenotypic performance either in
the early or second cropping seasons for the five traits. These observations
were inconsonance with Fentaw et al. (2015) and (Tonk et al.,
2011). In considering the proportion of GSI
variance in the phenotypic variance, the following traits made more than 50% contribution
namely, plant height at 2WAS (54%), FAW damage at 4WAS (58%), leaf area at 2WAS
(64%), number of leaves per plant at 2WAS (71%) and seedling emergence (89%)
(Table 7). The proportion of phenotypic variance attributed to seasonal effects
was highest in leaf area (100%) and leaf area index (92%) at 4WAS, but slightly
above 50% in FAW infestation at 2WAS. Seedling emergence was less likely to be influenced
by seasonal effect (11%). Apparently, the two experimental sites (Calabar and
Akpabuyo) had few relatively similar soil properties and weather conditions.
Generally low to moderate ratios of the genotypic variance to phenotypic
variance (<0.6) with accompanying low to moderately high genetic coefficient
of variability (GCV) (3.55 – 14.28%) indicated that the observed early
vegetative growth trait(s) difference(s) in the present study were not highly
heritable. There have been reports indicating that it is unreliable to consider
economic yield as the only determinant for genotype performance and selection (Fentaw
et al., 2015; Ittah and Obok, 2019; Kang et al., 1991; Odor et
al., 2017; Tonk et al., 2011). Also, none of these reports
considered the actual performance of a potential superior crop genotype at its early
vegetative growth stage and initial establishments in the field as addressed in
the present study.

Figure 1 (top): Fall
armyworm (FAW) infestation on African maize accession at 14 and 28 days after
sowing (DAS) during early and second cropping seasons in 2018. Figure 2 (bottom left): FAW on a seven-day old maize seedling showing patches. Figure
3 (bottom right): FAW larval feeding with leaves defoliation; leaving moist
sawdust-like frass in the funnel and on flag leaves.
Table 5. Mean early
vegetative growth performance and fall armyworm infestation on African maize
accessions.
|
Genotype
|
Seedling
Emergence
(%)
|
Plant
Height (cm)
|
Stem
Width (mm)
|
Number
of Leaves
|
Leaf
Area (cm2)
|
Leaf
Area Index
|
Fall
Armyworm
Infestation
(%)
|
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
|
TZm-109
|
71.76bc
|
17.1abc
|
29.8ab
|
13.9ab
|
15.6ab
|
5.5a
|
6.3a
|
326.1a
|
625.2a
|
1.07a
|
2.00ab
|
22.92b
|
34.38b
|
|
TZm-1097
|
61.11c
|
13.9abc
|
30.9ab
|
11.8b
|
15.6ab
|
5.1ab
|
6.2a
|
183.2b
|
492.7a
|
0.81ab
|
1.53ab
|
22.92b
|
35.42b
|
|
TZm-1153
|
73.15bc
|
16.5abc
|
32.5ab
|
14.5ab
|
16.4ab
|
5.1ab
|
6.8a
|
209.1ab
|
594.6a
|
0.68ab
|
1.98ab
|
17.71b
|
34.38b
|
|
TZm-1163
|
65.74bc
|
12.5c
|
24.5b
|
12.6b
|
15.0b
|
5.2ab
|
5.9a
|
162.2b
|
605.7a
|
0.57ab
|
1.77ab
|
22.92b
|
36.46b
|
|
TZm-1241
|
70.83bc
|
13.3bc
|
26.2b
|
11.2b
|
15.7ab
|
4.8b
|
5.9a
|
176.2b
|
567.4a
|
0.41b
|
1.66ab
|
21.88b
|
32.29b
|
|
TZm-1427
|
63.43bc
|
14.2abc
|
25.5b
|
14.3ab
|
17.7ab
|
5.0ab
|
5.8a
|
175.3b
|
452.8a
|
0.50ab
|
1.25b
|
16.67b
|
33.33b
|
|
TZm-299
|
88.89a
|
18.9ab
|
31.5ab
|
14.7ab
|
17.4ab
|
5.4ab
|
6.4a
|
237.2ab
|
572.2a
|
0.66ab
|
1.71ab
|
28.12ab
|
44.79b
|
|
TZm-304
|
78.24ab
|
18.9ab
|
39.5a
|
17.5a
|
18.6a
|
5.5ab
|
7.2a
|
338.6a
|
803.8a
|
0.96ab
|
2.88a
|
47.92a
|
65.62a
|
|
TZm-378
|
69.44bc
|
13.1bc
|
25.7b
|
13.8ab
|
16.7ab
|
5.0ab
|
6.3a
|
206.9ab
|
648.2a
|
0.67ab
|
1.90ab
|
36.46ab
|
51.04ab
|
|
TZm-687
|
78.70ab
|
15.7abc
|
28.9ab
|
15.3ab
|
17.7ab
|
4.8b
|
6.5a
|
284.3ab
|
597.7a
|
0.83ab
|
1.87ab
|
29.17ab
|
41.67b
|
|
Ikom
White
|
69.44bc
|
19.9a
|
29.1ab
|
12.5b
|
16.1ab
|
5.2ab
|
6.5a
|
252.2ab
|
528.6a
|
0.64ab
|
1.66ab
|
19.79b
|
32.29b
|
WAS = Weeks after sowing. Mean ± standard error with the
same letter(s) in superscript within the same column are not significantly
different at 95% confidence limit using Duncan’s Multiple Range Test.
Table 6. Mean squares of combined ANOVA of early vegetative growth
and fall armyworm infestation on African maize accessions.
|
Source
of Variation
|
df
|
Seedling
Emergence
(%)
|
Plant
Height (cm)
|
Stem
Width (mm)
|
Number
of Leaves
|
Leaf
Area (cm2)
|
Leaf
Area Index
|
Fall
Armyworm
Infestation
(%)
|
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
|
Season
(S)††
|
1
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
|
Rep
(Season) ††
|
4
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
-
|
|
Genotype
(G)
|
10
|
369.62ns
|
41.53ns
|
109.16ns
|
18.963ns
|
7.564ns
|
0.3693ns
|
0.9606ns
|
22876ns
|
50143ns
|
0.2293ns
|
1.0031ns
|
506.5ns
|
638.5ns
|
|
G x S
|
10
|
1252.53***
|
44.09*
|
131.56ns
|
10.057ns
|
7.585ns
|
0.8655**
|
0.6583ns
|
29511**
|
67007ns
|
0.1542ns
|
1.1302ns
|
494.9ns
|
658.9*
|
|
Pooled
error
|
40
|
138.06
|
20.19
|
88.06
|
10.027
|
5.77
|
0.2725
|
0.9742
|
10514
|
71082
|
0.1873
|
1.051
|
271.7
|
275.4
|
†† It was noted that the associated variance
components for the mean squares had very large standard errors. – The degree of
freedom was inadequate for the test of significance in Season (S) and Replicates
within Season (Rep (Season)) to be valid. *, **, *** and ns imply
p ≤ 0.05, ≤ 0.01, ≤ 0.001 and non-significance, respectively.
Table 7. Estimates of variance components and
their ratios for early vegetative growth and fall armyworm infestations on
African maize accessions.
|
Seedling
Emergence
(%)
|
Plant
Height (cm)
|
Number
of Leaves
|
Stem
Width (mm)
|
Leaf
Area (cm2)
|
Leaf
Area Index
|
Fall
Armyworm
Infestation
(%)
|
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
2WAS
|
4WAS
|
|
Mean
|
71.9
|
15.8
|
29.5
|
5.1
|
6.3
|
13.8
|
16.6
|
231.9
|
589.9
|
0.7
|
1.8
|
26.0
|
40.2
|
|

|
-147.15†
|
-0.43†
|
-3.73†
|
-0.08†
|
0.05
|
1.48
|
-0.01†
|
-1105.79†
|
-2810.61†
|
0.01
|
-0.02†
|
1.93
|
-3.39†
|
|
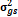
|
371.49
|
7.97
|
14.50
|
0.20
|
-0.11†
|
0.01
|
0.61
|
6332.27
|
-1358.28†
|
-0.01†
|
0.03
|
74.42
|
127.80
|
|

|
138.06
|
20.19
|
88.06
|
0.27
|
0.97
|
10.03
|
5.77
|
10514.20
|
71081.60
|
0.19
|
1.05
|
271.66
|
275.45
|
|
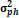
|
208.75
|
7.35
|
21.93
|
0.14
|
0.21
|
3.16
|
1.26
|
4918.50
|
11846.93
|
0.04
|
0.19
|
84.42
|
109.81
|
|
 / /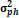
|
-
|
-
|
-
|
-
|
0.24
|
0.47
|
-
|
-
|
-
|
0.25
|
-
|
0.02
|
-
|
|
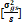 / /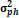
|
0.89
|
0.54
|
0.33
|
0.71
|
-
|
0.002
|
0.24
|
0.64
|
-
|
-
|
0.08
|
0.44
|
0.58
|
|
 / /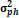
|
0.11
|
0.46
|
0.67
|
0.32
|
0.77
|
0.53
|
0.76
|
0.36
|
1.00
|
0.79
|
0.92
|
0.54
|
0.42
|
|
GCV
(%)
|
-
|
-
|
-
|
-
|
3.55
|
8.82
|
-
|
-
|
-
|
14.28
|
-
|
5.34
|
-
|
|
PCV
(%)
|
20.09
|
17.16
|
15.87
|
7.34
|
7.27
|
12.88
|
6.76
|
30.24
|
18.45
|
28.57
|
24.22
|
35.33
|
26.07
|
†Value is negative, estimated
as zero and represented as ‘-‘ in subsequent calculations. s = season, g =
genotype, r = replicate
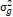 = genotypic variance, = genotypic variance, 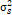 = season variance, = season variance,  = genotype x season
variance, = genotype x season
variance, 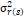 = replicate within
season variance, = replicate within
season variance, 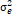 = pooled error variance, = pooled error variance,
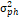 = = 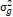 + + 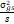 + +  = =  e,
GCV = genotypic coefficient of variability and PCV = phenotypic coefficient of
variability. e,
GCV = genotypic coefficient of variability and PCV = phenotypic coefficient of
variability.
Conclusions: The plant breeder
is often faced with the task of exploiting genetic diversity for the
improvement of a crop’s economic yield and yield-related traits. This could be
by the modification of abiotic factors especially growing condition and
requirements e.g. soil fertility and irrigation. However, it is not uncommon to
unintentionally ignore biotic factors such as an invasive pest. It is always
safe to consider those traits in a crop that could give information on their
early growth and performance when challenged with biotic stresses. These traits
and their timing could serve as aids to selection for maize accessions that
could serve as possible breeding materials for improvement of maize against
fall armyworm during early vegetative growth stages, field establishments and
subsequently, avoid yield losses. Our results showed that fall armyworm
infestation was higher in the second planting season than in the first planting
season. However, the GSI model used in the current study
could be improved to shed lights on the number of years or locations at which
trials should be conducted. To achieve this improvement, a genotype x locations
x year (GLY) model could be adopted in subsequent multi-environment trial (MET)
studies to permit well-informed decisions on number of years, locations (sites)
and replicates essential to attain a robust level of precision in the
assessment of the response of African maize genotypes to fall armyworm (FAW) damage
during the early vegetative growth stages.
REFERENCES
- Akpan-Idiok, A. U. (2012). Physicochemical properties, degradation rate and vulnerability potential of soils formed on coastal plain sands in Southeast, Nigeria. Int. J. of Agric. Res. 7: 358-366. doi: 10.3923/ijar.2012.358.366
- Bates, R.G. (1973). Determination of pH: theory and practice. New York: Wiley.
- Baudron, F., M.A. Zaman-Allah, I. Chaipa, N. Chari and P. Chinwada (2019). Understanding the factors influencing fall armyworm (Spodoptera frugiperda J.E. Smith) damage in African smallholder maize fields and quantifying its impact on yield. A case study in Eastern Zimbabwe. Crop Prot. 120: 141-150. doi: 10.1016/j.cropro.2019.01.028
- Black, C.A., D.D. Evans, J.L. White, L.E. Ensminger and F.E. Clark (1965). Methods of soil analysis. In: Methods of soil analysis, Agronomy, 1572. Madison, Wis.: American Society of Agronomy.
- Brévault, T., A. Ndiaye, D. Badiane, A.B. Bal, M. Sembène, P. Silvie and J. Haran (2018). First records of the fall armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae), in Senegal. Entomol Gen. 37: 129-142. doi: 10.1127/entomologia/2018/0553
- Burtet, L. M., O. Bernardi, A. A. Melo, M.P. Pes, T.T. Strahl and J.V.C. Guedes (2017). Managing fall armyworm, Spodoptera frugiperda (Lepidoptera: Noctuidae), with Bt maize and insecticides in southern Brazil. Pest Manag. Sci. 73: 2569-2577. doi: 10.1002/ps.4660
- Can, N.D. and T. Yoshida (1999). Genotypic and phenotypic variances and covariances in early maturing grain sorghum in a double cropping. Plant Prod. Sci. 2: 67-70. doi: 10.1626/pps.2.67
- Chen, M., R.N. Nagoshi, I. Dhanani, R. Asokan, H.M. Mahadevaswamy, C.M. Kalleshwaraswamy, S and R. L. Meagher (2019). Genetic characterization of fall armyworm infesting South Africa and India indicate recent introduction from a common source population. PLoS One 14: e0217755. doi: 10.1371/journal.pone.0217755
- Eijnatten, C.L.M. van (1965). Towards the improvement of maize in Nigeria, PhD Thesis, Agricultural University, Wageningen, The Netherlands.
- Elings, Anne (2000). Estimation of leaf area in tropical maize. Agron. J. 92: 436-444. doi: 10.2134/agronj2000.923436x
- Falconer, D.S. (1960). Introduction to quantitative genetics: Oliver & Boyd, Edinburgh & London.
- Fentaw, A., E. Melkamu and M. Yeshitila (2015). Genotype-environment interaction and stability analysis of hybrid maize varieties in North West Ethiopia. Int. J. of Plant Breed. and Genet. 9: 247-254.
- Food and Agriculture Organization of the United Nations (2018). Integrated management of the fall armyworm on maize: a guide for farmer field schools in Africa. Rome, Italy: FAO.
- Goergen, G., P. L. Kumar, S. B. Sankung, A. Togola and M. Tamo (2016). First report of outbreaks of the fall armyworm Spodoptera frugiperda (J.E. Smith) (Lepidoptera, Noctuidae), a new alien invasive pest in West and Central Africa. PLoS One 11: e0165632. doi: 10.1371/journal.pone.0165632
- Gomez, K. A. and A. A. Gomez (1984). Statistical procedures for agricultural research. Singapore: Wiley-Interscience Publication, John Wiley and Sons, Inc.
- Hay-Roe, M.M., R.L. Meagher, R.N. Nagoshi and Y. Newman (2016). Distributional patterns of fall armyworm parasitoids in a corn field and a pasture field in Florida. Bio. Control 96: 48-56. doi: https://doi.org/10.1016/j.biocontrol.2016.02.003
- Ittah, M.A. and E.E. Obok (2019). Breeding potential and multivariate analyses of morphological and yield traits in industrial sugarcane (Saccharum officinarum L.) accessions in a humid tropical agroecology. Int. J. of Plant & Soil Sci. 27: 1-10.
- Kalleshwaraswamy, C. M., R. Asokan, H. M. Mahadevaswamy and Sharanabasappa (2019). First record of invasive fall armyworm, Spodoptera frugiperda (J.E. Smith) (Lepidoptera: Noctuidae) on rice (Oryza sativa). India. J. of Entomol. and Zool. Stud. 7: 332-337.
- Kang, M.S., D.P. Gorman and H.N. Pham (1991). Application of a stability statistic to international maize yield trials. Theor. Appl. Genet. 81: 162-165. doi: 10.1007/BF00215718
- Kehr, W. R. and C. O. Gardner (1960). Genetic variability in Ranger alfalfa. Agron. J. 52: 41-44.
- Klute, Arnold (1986). Methods of soil analysis Part 1: Physical and mineral methods. In: Methods of soil analysis Part 1: Physical and mineral methods, ed. Arnold Klute, Agronomy, 770. Madison, Wis.: American Society of Agronomy : Crop Science Society of America : Soil Science Society of America.
- Marenco, R. J., R.E. Foster and C.A. Sanchez (1992). Sweet Corn Response to Fall Armyworm (Lepidoptera: Noctuidae) Damage During Vegetative Growth. J. of Econ. Entomol. 85: 1285-1292. doi: 10.1093/jee/85.4.1285
- Meagher, R. L., G.S. Nuessly, R.N. Nagoshi and M.M. Hay-Roe (2016). Parasitoids attacking fall armyworm (Lepidoptera: Noctuidae) in sweet corn habitats. Bio. Control 95: 66-72. doi: https://doi.org/10.1016/j.biocontrol.2016.01.006
- Murphy, J. and J. P. Riley (1962). A modified single solution method for the determination of phosphate in natural waters. Anal. Chim. Acta. 27: 31-36. doi: https://doi.org/10.1016/S0003-2670(00)88444-5
- Nagoshi, R. N., D. Koffi, K. Agboka, K. A. Tounou, R. Banerjee, J. L. Jurat-Fuentes and R. L. Meagher (2017). Comparative molecular analyses of invasive fall armyworm in Togo reveal strong similarities to populations from the eastern United States and the Greater Antilles. PLoS One 12: e0181982. doi: 10.1371/journal.pone.0181982
- Nagoshi, R. N., G. Goergen, K.A. Tounou, K. Agboka, D. Koffi and R.L. Meagher (2018). Analysis of strain distribution, migratory potential, and invasion history of fall armyworm populations in northern Sub-Saharan Africa. Sci. Rep. 8: 3710. doi: 10.1038/s41598-018-21954-1
- Nigerian Meteorological Agency (NiMET) (2018). Agrometeorological Bulletin. In: Agrometeorological Bulletin. Bill Clinton Drive, Nnamdi Azikiwe International Airport, Abuja: National Weather Forecasting and Climate Research Centre.
- Odor, E. O., G. A. Iwo and E. E. Obok (2017). Graphical assessment of yield stability and adaptation of cucumber (Cucumis sativus L) genotypes in Cross River State, Nigeria. J. of Agric. and Crop Res. 5: 108-116.
- Oliveira, M. A., F. H. I. Filho and R.T. Thuler (2018). Eficiency of Bacillus thuringiensis in controlling the corn fall armyworm in laboratory conditions. Revista Inova Ciência & Tecnologia, Uberaba 4: 14-19.
- Onwudike, S. U. (2015). Effect of land use types on vulnerability potential and degradation rate of soils of similar lithology in a tropical soil of Owerri, Southeastern Nigeria. Int. J. of Soil Sci. 10: 177-185. doi: 10.3923/ijss.2015.177.185
- Otim, M. H., W. T. Tay, T. K. Walsh, D. Kanyesigye, S. Adumo, J. Abongosi, S. Ochen, J. Sserumaga, S. Alibu, G. Abalo, G. Asea and A. Agona (2018). Detection of sister-species in invasive populations of the fall armyworm Spodoptera frugiperda (Lepidoptera: Noctuidae) from Uganda. PLoS One 13: e0194571. doi: 10.1371/journal.pone.0194571
- Pannuti, L. E., E. L. Baldin, T. E. Hunt and S. V. Paula-Moraes (2016). On-plant larval movement and feeding behavior of fall armyworm (Lepidoptera: Noctuidae) on Reproductive Corn Stages. Environ. Entomol 45: 192-200. doi: 10.1093/ee/nvv159
- Prasanna, B.M., J. E. Huesing, R.Eddy and V. M. Peschke (2018). Fall armyworm in Africa: A Guide for integrated pest management, (1st ed.) eds. B.M. Prasanna, J. E. Huesing, R. Eddy and V.M. Peschke. Mexico: CDMX: CIMMYT.
- Rwomushana, I., M. Bateman, T. Beale, P. Beseh, K. Cameron, M. Chiluba, V. Clottey, T. Davis, R. Day, R. Early, J. Godwin, P. Gonzalez-Moreno, M. Kansiime, M. Kenis, F. Makale, I. Mugambi, S. Murphy, Nunda. W., N. Phiri, C. Pratt and J Tambo (2018). Fall armyworm: impacts and implications for Africa. Evidence Note: Knowledge for Life. United Kingdom: Oxfordshire: CABI.
- Sparks, D. L., A. L. Page, P. A. Helmke and R. H. Loeppert (1996). Methods of Soil Analysis Part 3—Chemical Methods. In Methods of Soil Analysis Part 3—Chemical Methods, eds. D. L. Sparks, A. L. Page, P. A. Helmke and R. H. Loeppert, SSSA Book Series, 1390. Madison, WI: Soil Science Society of America, American Society of Agronomy.
- Toepfer, S., U. Kuhlmann, M. Kansiime, D. O. Onyango, T. Davis, K. Cameron and R. Day (2018). Communication, information sharing, and advisory services to raise awareness for fall armyworm detection and area-wide management by farmers. J. of Plant Diseases and Prot. 126: 103-106. doi: 10.1007/s41348-018-0202-4
- Tonk, F. A., E. Ilker and M. Tosun (2011). Evaluation of genotype x environment interactions in maize hybrids using GGE biplot analysis. Crop Breed. and Appl. Biotech. 11: 01-09.
- Twumasi, P., A. Y Tetteh, K. B. Adade, S. Asare and R. A. Akromah (2017). Morphological diversity and relationships among the IPGRI maize (Zea mays L) landraces held in IITA. Maydica 62:M25.
- WorldWeatherOnline (2018). Monthly Climate Averages: Calabar and Akpabuyo - Cross River State, Nigeria. In Monthly Climate Averages: Calabar and Akpabuyo - Cross River State, Nigeria.
- Yassin, T. E. (2009). Genotypic and phenotypic variances and correlations in field beans (Vicia faba L.). The J. of Agric. Sci. 81: 445-448. doi: 10.1017/S0021859600086494.
|
|
|
|
|





