Short Communication
EFFICACY OF ANTI-BABESIA DRUGS IN RELATION TO HEMATOLOGICAL PARAMETERS IN DOGS
M. Tayyub1, K. Ashraf1, M. Lateef1, A. A. Anjum2, M. A. Ali2, M. Nawaz2, M. M. Nazir3, S. Naz4 and N. Ahmad1
1Department of Parasitology, 2Department of Microbiology, University of Veterinary and Animal Sciences, Lahore, Pakistan; 3Baha Uddin Zakariya University, Multan-Pakistan; 4Veterinary Research Institute, Lahore, Pakistan
Corresponding Author’s Email: kashraf@uvas.edu.pk
ABSTRACT
Canine babesiosis is characterized by progressive anemia, fever, thrombocytopenia and marked splenomegaly. Different drugs are used for treatment and eradication of canine babesiosis. Present study was conducted to determine efficacy of anti-Babesia drugs and their effect on blood parameters in pet dogs. For this purpose, 30 naturally infected dogs confirmed by PCR were randomly divided into three experimental groups (A, B and C) and ten healthy dogs as control group (D). The dogs of groups A, B and C were treated with imizole (1mL/10Kg), vibramycin (100mg/10Kg) and imizole plus vibramycin, respectively. Blood profile of all the dogs was also recorded before and after treatment for comparison. After 21 days of treatment, maximum recovery (90%) was recorded in group C followed by group A (77%) and B (62.5%), respectively. The level of Hb, PCV, TEC, TLC, glucose, globulin, total protein and neutrophil was less in infected dogs in comparison to healthy dogs, while the level of lymphocytes, eosinophil, monocytes, SGPT and BUN were higher in infected dogs. The PCV returned to normal (39.97±2.12) in group C dogs in relation to group D (40.51±0.96). However, these values were lower (p≤0.01) in group A (38.43±2.08) and B (37.12±2.79). Similar pattern was observed for other parameters such as neutrophil, eosinophil, lymphocytes and hemoglobin value. Babesia specific amplification (339 bp) was successful for infected dogs. It was concluded that combination of imizole and vibramycin was better option for treatment of pet dogs suffering from babesiosis and restores blood parameters to normal after 21 days.
Keywords: Babesiosis; Pet dogs; Imizole; Vibramycin; Blood profile.
https://doi.org/10.36899/JAPS.2020.5.0153
Published online June 25, 2020
INTRODUCTION
Babesiosis is a protozoan disease caused by an intra-erythrocytic protozoan (Babesia: B.) which is distributed throughout the world. The B. canis and B. gibsoni are the major etiological agents of canine babesiosis. Canine babesiosis can be characterized by progressive anemia, fever, thrombocytopenia and marked splenomegaly, although sometime babesiosis can lead to death of animal (Földvári et al., 2005; Salem and Farag, 2014). Transmission of babesiosis in dogs is due to direct transfer from dog bite, trans-placental transmission or vector borne transmission. However, transmission of disease is generally due to infected ticks (Uilenberg, 2006). Clinical signs of babesiosis in canine vary with species of Babesia, age of host, immunity status of host and multiple infections of the affected dogs. Reported mortality rate in Babesia infected dogs is 28-70% (Scott-Moncrieff et al., 2001). Infections caused by B. canis, B. gibsoni and B. conradae vary in severity according to host (Penzhorn, 2011; Solano-Gallego and Baneth, 2011).
Variation in clinico-pathological investigations is important for differentiation of babesiosis caused by different species of Babesia as reported by Jacobson (2006). It has high morbidity and mortality in dogs like malaria in human beings (Kjemtrup et al., 2006). In canine babesiosis hemolytic anemia, systemic inflammation, organ dysfunction account for most clinical signs. Disease onset is acute with infected dogs suffer from fever and lethargy. Infected dogs suffer from liver, pulmonary, kidney, cerebral dysfunction and hemostatic abnormalities (coagulation and electrolytes imbalance) (Eichenberger et al., 2016). Many drugs have been used for treatment and eradication of canine babesiosis including babesiacidal agents, anti-protozoal drugs and some other antibiotics. Single drug is also available for complete treatment and elimination of the babesiosis from infected canine (Kjemtrup et al., 2006). Several anti-babesial drugs and drug combinations effective against canine babesiosis have been reported (Uilenberg et al., 1981; Rowin et al., 1982; Kjemtrup and Conrad, 2006; Vial and Gorenflot, 2006; Di Cicco et al., 2012). But mechanisms of action of these drugs against babesia are unknown or not studied in detail (Checa et al., 2017). It is important to treat the babesiosis with effective anti-protozoal drugs or the combinations.
Frequent clinical signs associated with canine babesiosis are apathy, weakness, anorexia, pale mucous membranes and a poor general condition. All Babesia species can cause fever, enlarged lymph nodes and spleen, anemia, thrombocytopenia, jaundice and pigmenturia. Other abnormalities include hypoalbuminemia and hyperbilirubinemia (Eichenberger et al., 2016; Checa et al., 2017). Imidocarb dipropionate and diminazine aceturate are mostly prescribed with doxycycline or enrofloxacin. Metronidazole is another choice. It is very hard to remove Babesia spp. from infected host completely (Irwin, 2009). Vaccine for babesiosis is also available but its efficacy is variable. However, it protects against the severity of the clinical signs (Brown et al., 2006). Keeping in view the whole scenario, present study was conducted to determine efficacy of anti-Babesia drugs and their effects on blood profile of infected dogs.
MATERIALS AND METHODS
Experimental Animals: A total of 40 pet dogs (German shepherd) were enrolled for this study from different pet clinics in Lahore (Clinic 1, 2, 3 and 4, n=10 dogs from each clinic). Thirty dogs were those presented with babesiosis which was diagnosed and confirmed by microscopy and polymerase chain reaction (PCR). Ten dogs were also enrolled as a control group (Declared negative by PCR) from the same area.
Detection and confirmation of babesiosis: Babesiosis was preliminary diagnosed by presence of ticks on skin (Fig. 1) clinical manifestation (fever, pale gums and tongue, enlarged lymph nodes) and microscopic detection of Babesia spp. from blood through Giemsa staining. DNAs were extracted from blood of suspected dogs following a commercial DNA extraction Kit (Gene All, Korea) and Babesia spp. specific amplification of 18S rDNA was done using a Babesia genus specific primer pair 5–22F(5’-GTTGATCCTGCCAGTAGT’3) and 1661R (5’-AACCTTGTTACGACTTCTC-3’) in a 25 µL reaction mixture containing 10 pmoles of each primer, 25 ng DNA and 12.5 µL of PCR master mix (Wis Bio, Korea) as described previously (Tayyub et al., 2019). The samples showing amplicon of 339bps on 2% agarose gel were confirmed.
Treatment of babesiosis: Babesiosis positive dogs (n=30) were treated with three different treatment strategies (A, B and C) including imidocarb (6 mg/Kg as a single dose), vibramycin (10 mg/Kg once for five days) and a combination of imidocarb (6 mg/Kg as a single dose) plus vibramycin (10 mg/Kg for 5 days). These drugs were administered intra-muscular using sterile technique. Dogs were given respective treatments and returned with their companion. Control group was not given any treatment (Group D).
Efficacy of treatment strategies: Efficacy of three treatment strategies was measured in terms of gradual improvement in clinical signs and improvement in blood parameters. Absence of Babesia spp. in blood of dogs was also determined by Giemsa staining and PCR a weak after treatment. Clinical signs including fever, pale gums and tongue, enlarged lymph nodes were noted and recorded daily during treatment till seven days post treatment. Blood was collected from each dog before treatment (0 day) and after treatment (day 21) and blood profile was determined.
Blood profile: Blood profile was determined by automated hematology analyzer calibrated for animal use. Blood parameters determined included White Blood Cell (WBC), Lymphocyte (LYM), Minimum Inhibitory Dilution (MID), Granulocytes (GRA), Relative percent of Lymphocytes (LY%), Myocardial Infraction (MI%), Relative percent of Granulocytes (GR%), Red Blood Cells (RBC), Hemoglobin (HGB), Hematocrit (HCT), Mean corpuscular volume (MCV), Mean Corpuscular Hemoglobin (MCH), Mean concentration of hemoglobin in Erythrocytes (MCHC), Relative distribution width of Red Blood Cells (RDWC), Platelets (PLT) and Mean platelet volume (MPV).
Statistical Analysis: The data obtained were presented as Mean± SD and analyzed by one-way analysis of variance and means in different groups were compared by Duncan’s Multiple Range (DMR) post hoc test at p ≤ 0.05 by SPSS version 20.0.
RESULTS
Study was planned to investigate relationship of blood profile with babesiosis before and after treatment. Modification in blood profile may be an influencing factor for canine babesiosis. Also, discrepancy in blood profile may be warning of carrier or recovery status of babesiosis in dogs. In this study, a total of (n=30) naturally infected dogs declared positive for babesiosis by PCR were randomly divided into three experimental groups (A, B and C) and ten healthy dogs as control group (D). Positive samples showed amplicon of 339 bps when resolved using 2% agarose gel. While, negative samples did not show any amplicon (Tayyub et al., 2019). The blood profile of all the dogs infected and normal were examined before and after treatment. The blood profile of the dogs before treatment is presented in table 1. Different parameters of blood were examined in both healthy and infected dogs, including; Hb, PCV, TEC, TLC, neutrophil, eosinophil, monocytes, total protein, globulin, albumin, A: G ratio, SGPT, BUN, Glucose and creatinine. The level of Hb, PCV, TEC, TLC, glucose, globulin, total protein and neutrophil was less in infected dogs as associate to healthy dogs, while the level of lymphocytes, eosinophil, monocytes, SGPT and BUN was high in infected dogs as equate to vigorous dogs presented in table 01. The dogs were divided into three groups (A, B and C). The dogs of groups A were treated with (Imidocarb), group B with (vibramycin) and C were treated with (Imidocarb plus vibramycin), and control received no treatment existing in table 02). On day 21 post treatment, there were significant changes in hematological parameters. After 21 days of treatment, maximum recovery (90%) was recorded in group C followed by group A (77%) and group B (62.5%), respectively. All the experimental groups were on the same level of infection as there was non-significant difference (p>0.05). The PCV returned to normal (39.97±2.12) in group C dogs in relation to group D (40.51±0.96). However, these values were lower (p≤0.01) in group A (38.43±2.08) and B (37.12±2.79). Similar pattern was observed for other parameter such as neutrophil, eosinophil, lymphocytes, hemoglobin value and A: G ratio presented in table 02.
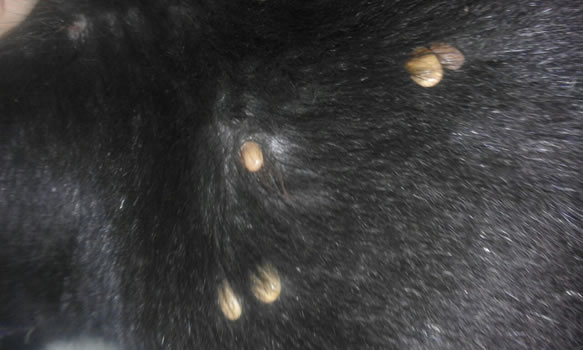
Figure 1: Ticks on skin of a dog
Table 1. Comparison of hematological characters of Babesiosis positive and negative dogs (Mean ± S.E.)
|
Parameter
|
Babesiosis positive
|
Babesiosis negative
|
P Value
|
|
Hb (g/dl)
|
10.12 ± 0.25
|
13.13 ± 0.12
|
0.000
|
|
PCV (%)
|
29.89 ± 7.08
|
40.43 ± 1.98
|
0.001
|
|
TEC x106/cumm
|
3.87 ± 1.43
|
6.42 ± 0.54
|
0.000
|
|
TLC/cumm
|
9002.45 ± 76.54
|
9450.32 ± 40.52
|
0.021
|
|
Neutrophils/cumm
|
3789 ± 142.21
|
6743.45 ± 190.32
|
0.000
|
|
Lymphocytes/cumm
|
4632 ± 190.27
|
2801.76 ± 129.28
|
0.000
|
|
Eosinophils/cumm
|
265 ± 40.62
|
160.87 ± 45.89
|
0.153
|
|
Monocytes/cumm
|
218 ± 90.89
|
70.92 ± 18.09
|
0.005
|
|
Total protein (g/dL)
|
6.04 ± 0.34
|
7.09 ± 0.56
|
0.001
|
|
Albumin (g/dL)
|
2.48 ± 0.12
|
3.34 ± 0.10
|
0.000
|
|
Globulin (g/dL)
|
3.12 ± 0.57
|
3.65 ± 0.19
|
0.104
|
|
A:G ratio
|
0.80 ± 0.20
|
0.87 ± 0.15
|
0.196
|
|
Glucose (mg/dL)
|
89.73 ± 6.65
|
122.46 ± 5.87
|
0.003
|
|
SGPT (ALT) (U/L)
|
54.09 ± 7.90
|
18.89 ± 1.63
|
0.008
|
|
BUN (mg/dL)
|
26.06 ± 2.78
|
17.05 ± 0.65
|
0.003
|
|
Creatinine (mg/dL)
|
1.40 ± 0.23
|
12.97 ± 0.28
|
0.015
|
Non-significant (p > 0.05); * Significant (p ≤ 0.05); ** highly significant (p ≤ 0.01)
Hb: hemoglobin, PCV: Pack cell Volume, TEC: Total erythrocyte count, TLC: Total leukocyte count, SGPT (ALT): serum glutamic-pyruvic transaminase (Alanine aminotransferase), BUN: Blood Urea Nitrogen, %: Percent.
Table 2. Hematological parameters in dogs suffering from babesiosis and healthy on day 21 Post treatment.
|
Parameter
|
Hematological parameters in dogs
|
|
Imidocarb
|
Vibramycin
|
Imidocarb + vibramycin
|
Control
|
|
Hb (g/dl)
|
12.34 ± 0.40
|
11.89 ± 0.56
|
12.98 ± 0.55
|
13.23 ± 0.12
|
|
PCV (%)
|
38.43 ± 2.08
|
37.12 ± 2.79
|
39.97 ± 2.12
|
40.51 ± 0.96
|
|
TEC x106/cumm
|
6.22 ± 0.09
|
6.02 ± 0.09
|
6.35 ± 0.67
|
6.32 ± 0.45
|
|
TLC/cumm
|
9309.98 ± 71.05
|
9250.76± 89.38
|
9434.89 ± 79.03
|
9450.32 ± 38.33
|
|
Neutrophils/cumm
|
6694.89 ± 201.67
|
6590.87 ± 180.08
|
6809.23 ± 154.17
|
6743.45 ± 208.43
|
|
Lymphocytes/cumm
|
2634.09 ± 201.89
|
2601.67 ± 189.76
|
2827.90 ± 154.34
|
2801.76 ± 145.13
|
|
Eosinophils/cumm
|
179.87 ± 32.09
|
194.78 ± 32.70
|
162.98 ± 49.34
|
160.87 ± 53.11
|
|
Monocytes/cumm
|
87.32 ± 27.78
|
97.27 ± 25.25
|
73.87 ± 17.56
|
70.92 ± 16.02
|
|
Total protein (g/dL)
|
6.45 ± 0.69
|
6.30 ± 0.29
|
6.98 ± 0.66
|
7.09 ± 0.47
|
|
Albumin (g/dL)
|
3.14 ± 0.10
|
3.08 ± 0.30
|
3.25 ± 0.20
|
3.31 ± 0.90
|
|
Globulin (g/dL)
|
3.56 ± 0.29
|
3.54 ± 0.43
|
3.76 ± 0.19
|
3.61 ± 0.19
|
|
A:G ratio
|
0.83 ± 0.48
|
0.81 ± 0.53
|
0.86 ± 0.76
|
0.82 ± 0.13
|
|
Glucose (mg/dL)
|
120.46 ± 6.43
|
118.57 ± 6.73
|
125.53 ± 6.43
|
120.55 ± 5.33
|
|
SGPT (ALT) (U/L)
|
19.65 ± 2.09
|
20.89 ± 2.89
|
19.45 ± 2.01
|
18.29 ± 1.63
|
|
BUN (mg/dL)
|
17.39 ± 0.87
|
17.43 ± 0.71
|
17.34 ± 0.89
|
16.85 ± 0.51
|
|
Creatinine (mg/dL)
|
11.80 ± 0.39
|
10.87 ± 0.43
|
13.01 ± 0.43
|
12.44 ± 0.26
|
Hb: hemoglobin, PCV: Pack cell Volume, TEC: Total erythrocyte count, TLC: Total leukocyte count, SGPT (ALT): serum glutamic-pyruvic transaminase (Alanine aminotransferase), BUN: Blood Urea Nitrogen %: Percent
DISCUSSION
Babesiosis is also called as piroplasmosisis, Texas fever, red water or tick fever. It is transmitted via ticks, caused by intra-erythrocytic bloodsuckers of the genus Babesia. It causes substantial wide-reaching pecuniary, medical and veterinary threat (Meissner et al., 2017). Babesia species are transferred by a tick vector which is accomplished of blighting a wide assortment of hordes and even a single infected tick is adequate to spread the disease (Uilenberg et al., 2006). There is not so much data available on canine babesiosis published in recent years. Now a days, babesiosis is diagnosed based on presence of ticks on skin of dogs, signs and symptoms. Sometimes, microscopic examination is also recommended. Imizole alone or with amoxycillin is used for treatment of babesiosis in different animals. Relationship of blood profile with babesiosis in dogs was studied first time in Pakistan in present study. Based on present study, complete blood chemistry analysis along with microscopy may confirm babesiosis in dogs. The first day after treatment, the RBC count was significantly lower. While, the WBC count was significantly high in all groups except control group treated with imizole, vibramycin and combination of imizole and vibramycin. Overall, results revealed that in positive dogs, parasite occurrence affected the white blood cells as well as red blood cell as compared to animals without parasitic infection. The treated dogs of group C showed increased value of TLC, Hb, PCV and other blood parameter in accordance with (Bajer et al., 2013). Contrarily, the CBC results of group B samples showed decrease value of Hb, PCV and TEC. Similar observations were chronicled by other canvassers (Iguchi et al., 2014; Baneth, 2018) Hematological parameters of control dogs from same vicinity were pragmatic in 2014 study. Significant (P≤0.05) reduction in Hb contents, PCV%, neutrophils and TLC along with increase in monocytes count were noted in infected dogs compared to control. TEC reduce count was observed in diseased dogs. Similar study was done by (Salem et al., 2014) and they observed 100% thrombocytopenia in affected dogs.
Treatment of group C (Combination of Imizole and Vibramycin) was better treatment regime (90% recovery) as compared to other groups. Imidocarb dipropionate is used as first choice of treatment in large animals but is not first choice in small animals. Vibramycin successfully killed B. gibsoni isolates of dogs when tested in-vitro (Kumara, 2016). Imizole and vibramycin were used by different researchers and studied their safety in dogs. Similar findings were recorded by other researchers in canine.
There are different regimes used for treatment of babesiosis in canines worldwide. Important treatment options include imidocarb dipropionate and doxycycline. Most of the scientists claim imidocarb dipropionate first choice of treatment against babesiosis in large animals but when used to treat small animals, clinical relapses are very frequent (Hilpertshauser et al., 2006). The combination of dindamycin, diminazeneaceturate and imidocarb dipropionate is also effective against B. gibsoni and used to treat atovaquone-resistant strains of this species. Novel drugs and the synergistic effects of drug combinations against Babesia infection should be explored further to find new treatments for canine babesiosis (Sainz et al., 2000). In present study, combination of imizole and vibramycin was better option which returned PCV to normal (39.37.0±0.39), Hb value (12.98 ± 0.55), and TLC count (9434.89 ± 79.03). Similar results were reported by other researchers (Irwin, 2009). In a study, the effect of combination therapy was evaluated on dogs. Combinations of (Imidocarb + vibramycin) were used for babesiosis treatment in dogs in current study. After treatment with these drugs, improvement in hematological parameters was observed and akin report was done mentioned by previous studies (Sakuma et al., 2009).
It was concluded that there was significant difference in hematological parameters of Babesia infected and healthy dogs and combination of imizole and vibramycin was a better choice for treatment with maximum recovery percentage and reverse hematological parameters to normal values.
REFERENCES
- Bajer, A., A. Rodo, M. Bednarska, E. Mierzejewska, and R. Welc-Faleciak (2013). Babesiacanis and tick-borne encephalitis virus (TBEV) co-infection in a sled dog. Ann. Agric. Environ. Med. 20(3): 426-430.
- Baneth, G. (2018). Antiprotozoal treatment of canine babesiosis. Parasitol. 254: 58-63.
- Brown, W.C., J. Norimine, W.L. Goff, C.E. Suarez, and T.F. McElwain (2006). Prospects for recombinant vaccines against Babesiabovis and related parasites. Parasite Immunol. 28(7): 315-327.
- Checa, R., A. Montoya, N. Ortega, J.L. González-Fraga, A. Bartolomé, R. Gálvez, and G. Miró (2017). Efficacy, safety and tolerance of imidocarb dipropionate versus atovaquone or buparvaquone plus azithromycin used to treat sick dogs naturally infected with the Babesia microti-like piroplasm. Parasit. Vectors. 10(1): 145.
- Di Cicco, M.F., M.E. Downey, E. Beeler, H. Marr, P. Cyrog, L. Kidd, P.P. Diniz, L.A. Cohn and A.J. Birkenheuer (2012). Re-emergence of Babesia conradae and effective treatment of infected dogs with atovaquone and azithromycin. Vet. Parasitol. 187: 23–27.
- Eichenberger, R.M., B. Riond, B. Willi, R. Hofmann‐Lehmann, and P. Deplazes (2016). Prognostic markers in acute Babesia canis J. Vet. Int. Med. 30(1): 174-182.
- Földvári, G., E. Hell, and R.J.V.P. Farkas (2005). Babesia canis in dogs from Hungary: Detection by PCR and sequencing. Vet. Parasitol. 127(3-4): 221-226.
- Hilpertshauser, H., P. Deplazes, M. Schnyder, L. Gern, and A. Mathis (2006). Babesia identified by PCR in ticks collected from domestic and wild ruminants in southern Switzerland. Appl. Environ. Microbiol. 72(10): 6503-6507.
- Iguchi, A., Shiranaga, A. Matsuu, and Y. Hikasa (2014). Efficacy of Malarone® in dogs naturally infected with Babesiagibsoni. J. Vet. Med. Sci. 76(9): 1291-1295.
- Irwin, P.J. (2009). Canine babesiosis: from molecular taxonomy to control. Parasit.Vectors. 2(S1): S4. doi:10.1186/1756-3305-2-S1-S4.
- Jacobson, L.S. (2006). The South African form of severe and complicated canine babesiosis: clinical advances. Vet. Parasitol. 138(1-2): 126-139.
- Kjemtrup, A.M. and P.A. Conrad (2006). A review of the small canine piroplasms from California: Babesia conradae in the literature. Vet. Parasitol. 138: 112-117.
- Kjemtrup, A.M., Wainwright, M. Miller, B.L. Penzhorn, and R.A. Carreno (2006). Babesiaconradae, sp. nov., A small canine Babesia identified in California. Vet. Parasitol. 138(1-2): 103-111.
- Kumara, W.R. (2016). Antibabesial treatment protocols against canine babesiosis. L. Vet. J. 63(1): 15-21.
- Meissner, E.G.,W. McGillicuddy, J. Squires, D. Skipper, S. Self, D. Wray, and S. Nadig (2017). Across state lines: fulminant Babesiamicroti infection in a liver transplant recipient. Transpl. Infect. Dis. 19(5): 12741.
- Penzhorn, B.L. (2011). Why is Southern African canine babesiosis so virulent? An evolutionary perspective. Parasites Vectors. 4(1): 51.
- Rowin, K.S., H.B. Tanowitz and M. Wittner (1982). Therapy of experimental babesiosis. Ann. Intern. Med. 97(4): 556-558.
- Sainz, A.,A. Tesouro, I. Amusategui, F. Rodriguez, F. Mazzucchelli, and M. Rodriguez (2000). Prospective comparative study of 3 treatment protocols using doxycycline or imidocarb dipropionate in dogs with naturally occurring ehrlichiosis. J. Vet. Int. Med. 14(2): 134-139.
- Sakuma, M., A. Setoguchi, and Y. Endo (2009). Possible emergence of drugresistant variants of Babesiagibsoni in clinical cases treated with atovaquone and azithromycin. J. Vet. Int. Med. 23(3): 493-498.
- Salem, N. and H. Farag (2014). Clinical, hematologic, and molecular findings in naturally occurring Babesia canis vogeli in Egyptian dogs. Vet. Med. Int. 2014: 270345. doi.10.1155/2014/270345.
- Scott-Moncrieff, J.C., N.G. Treadwell, S.M. McCullough, and M.B. Brooks (2001). Hemostatic abnormalities in dogs with primary immune-mediated hemolytic anemia. J. Am. Anim. Hosp. Assoc. 37(3): 220-227.
- Solano-Gallego, L. and G. Baneth (2011). Babesiosis in dogs and cats-Expanding parasitological and clinical spectra. Vet. Parasitol. 181(1): 48-60.
- Tayyub, M., Ashraf, M. Lateef, A.A. Anjum, M.A. Ali, N. Ahmad, and M.M. Nazir (2019). Genetic diversity of canine Babesia species prevalent in pet dogs of Punjab, Pakistan. Anim. 9(7): 439.
- Uilenberg, G. (2006). Babesia-A historical overview. Vet. Parasitol. 138(1-2): 3-10.
- Uilenberg, G., P.A. Verdiesen, and D. Zwart (1981). Imidocarb: A chemoprophylactic experiment with Babesia canis. Vet. Q. 3(3): 118-123.
- Vial, H.J. and A. Gorenflot (2006). Chemotherapy against babesiosis. Vet. Parasitol. 138: 147-160.
|





