Content for New Div Tag Goes Here
RATIONAL MANAGEMENT OF PHENOCOCCUS SOLENOPSIS THROUGH CONSERVATION AND AUGMENTATION OF AENASIUS BAMBAWALEI AND CRYPTOLAEMUS MONTROUZIERI IN PAKISTAN
M. N. Aslam*1, Ehsan-ul-Haq1 and M. Aslam2
1Department of Plant and Environmental Protection, PARC Institute of Advanced Studies in Agriculture, National Agricultural Research Centre, Islamabad, The University of Agriculture, Peshawar, Pakistan; 2Department of Entomology, Faculty of Agricultural Sciences and Technology, BZU, Multan
*Corresponding Author’s email : naeemmalghani80@gmail.com
ABSTRACT
Cotton mealybug (Phenococcus solenopsisTinsley 1898) is one of the major sucking insect pest of cotton in Pakistan. This study was carried out during 2012 & 2013 in Multan District, Punjab, Pakistan to evaluate the response and conservation of biocontrol agents i.e. Aenasius bambawalei Hayat(= A. arizonensis Fallahzadeh, 2014) and Cryptolaemous montrouzieri Mulsant, 1850 against cotton mealybug (CMB). Biological responses of these biocontrol agents were assessed at different temperatures and humidity levels in the laboratory; which helped to design the rearing technologies for field releases. Data was collected at 100, 200 and 400 meters from NEFR surroundings after 07, 15 and 22 days of releases (DAR) of biocontrol agents. Results showed a significant decrease in CMB population; at 100 meter as 46%, 69% and 94% respectively; at 200 meter as 32%, 53% and 83% respectively; at 400 meter as 25%, 45% and 77% respectively. Cost benefit ratio of NEFR assessed was 1:1.8 and 1:3.2 when the crop losses reduced 20 to 50 percent in comparison with control field.
Keywords: Biological control, NEFR, natural enemies, population dynamics
https://doi.org/10.36899/JAPS.2020.3.0089 Published online March 25, 2020.
INTRODUCTION
Cotton (Gossypium hirsutum) is one of the major cash crop in Pakistan and contributes about 10% in GDP (Economic Survey of Pakistan, 2015-16). It undergoes pest pressure during its seasonal cycle and faces reduced production imposing economic implications to the Pakistan. Cotton mealybug (Phenococcus solenopsis) is among the major insect pest of cotton which sucks cell sap and affects plant growth in multiple ways. Initially, leaves turn yellow, crinkle and curly leading towards drop off. Infested flowers often drop and sometimes produce fewer deformed bolls or no fruit. (Yonas et al., 2019). Severely damaged plants suffer premature dehydration and give the appearance of defoliator spray on cotton plant. Afterwards, plant becomes stunted and death occurs. Under indirect infestation, it produces honeydew resulting in growth of sooty mold on leaves that hinders photosynthesis (Mark and Gullan, 2005; Saeed et al., 2007).
In Pakistan tropical and subtropical climate with summer rains and short winter favours mealybug breeding to keep it active throughout the year. All stages are evident with many overlapping generations found damaging plant parts such as leaves, shoots, twigs, branches, flowers and roots. Infestation occurs in patches that make huge loss to cotton yield (Mahmood et al., 2011).
Farmers in developing countries including Pakistan solely relies on application of chemical sprays to get immediate results for its management (Joshi et al., 2010). In addition, non judicial use of toxic pesticides impose environment and health implications due to their residual behavior and entry into food chain. There is need to evaluate and adopt some alternate management approach to manage cotton mealybug below economic threshold level. In this regard, exploration and utilization of biocontrol agents associated with cotton mealybug has received considerable attention all over the world. However, this concept is not familiar in developing countries due to lack of technical capacity and awareness among farming communities. Furthermore, biological control requires extensive identification and exploration of biocontrol agents and then designing their rearing and release technologies for rapid adoption at farm level.
Chrysoperla carnea and Coccinellid beetles are reported promising biocontrol agents to regulate population of cotton mealybug below economic threshold level (Agarwal et al., 1988; Gautam, 1996; Ali et al., 2014). Coccinellid beetles such as Cryptolaemus montrouzieri (known as mealybug destroyer), Brumoides suturalis, C. septumpunctata, Hippodamia variegata, Menochilus sexmaculatus and Hyperaspis maindron, are considered great destroyers of all stages of mealybugs (Ali et al., 2014). Among parasitoids, Aenasius bambawalei (= A. arizonensis) has high searching ability, short life cycle and high parasitism rate (upto 93%). One of the pre-requisites to achieve successful biocontrol program is the availability and survival of the biocontrol agents before the pest population emerges.
The current study was designed to establish a baseline for the future biological control programs in Pakistan by monitoring P. solenopsis and their associated natural enemies in southern Punjab (Multan). For strengthening the study goals, laboratory rearing techniques of native A. bambawalei (= A. arizonensis), with view point of their on-farm conservation was also established.
MATERIALS AND METHODS
The study was conducted at farmer fields at five different locations (Basti Malook, Basti Laar, Billiwala, Makhdoom Rasheed and Sariwala) in Multan district. The crop at each location was sown and all the appropriate agronomic practices were duly performed. The cotton mealybug and respective natural enemies population (predators and parasitoids) was recorded to know the trend on cotton crop at Multan district.
Methodology:
Population dynamics of CMB: Five different locations were randomly selected in such a manner that each field was five to ten kilometers apart from each other. The cotton variety FH-142 was sown in the 3rd week of April during both study years (2012 & 2013). The population data for mealybug and its predators and parasitoids was taken at fifteen days intervals. The data were recorded for two consecutive years (2012 and 2013) from cotton. For recording the population data, five inches terminal twigs of 15 cm length were selected randomly from cotton. These samples were brought to the laboratory to record the mealybug population and its natural enemies which were kept for two weeks in the plastic jars (10 kg) covered with the muslin cloth. Population data of predators and parasitoids from collected samples were recorded on daily basis. Mealybug predators and parasitoids, after recording the data, were removed from samples. The parasitoids population was calculated in percentage on the basis of number of mummified mealybugs in each sample.
Rearing of A. bambawalei:Population of cotton mealybug was collected from field and reared on potato sprouts under lab conditions. The uniform aged 3rd instar (260) CMB were separated and placed with newly emerged A. bambawalei (= A. arizonensis) pair in 5 Kg plastic jar having muslin cloth on its lid with six replications. The experiment was conducted in environment growth chamber having five temperatures regimes (20, 24, 28, 32, 36 ºC) and four relative humidity levels (50, 60, 70, 80 %). Each replication comprised of four jars of each treatment. The honey solution was provided on daily basis to improve the performance. The data was recorded after seven, fourteen and twenty one DAR by counting mummified and emerged parasitoids. Techniques for A. bambawalei releases : Three techniques (cards with mummies, adults and mummies with host) were used to release the A. bambawalei (= A. arizonensis) under field conditions. Cards with mummies were organized and taken from the Biological Control Laboratory, CABI, Multan. Each card comprised of 4 inches white paper having 200 mummies pasted on it. The reared adults from laboratory of uniform aged were released in the field. The mummies with host were shifted to the field. Thirty-five hundred (3500) mummies /adults/acre were released under field conditions. The data were recorded on the basis of mummified mealybugs after seven, fourteen and twenty one DAR.
Cryptolaemus montrouzieri Rearing: Adults of C. montrouzieri were collected from CABI. Thirty (30) adults were placed in plastic jars and then released in the transparent celluloid made cage (48 x 17 x 14 cm) having sleeves as well as mesh windows. The net improved aeration to avoid the growth of sooty mold. Mealybug infested pumpkin were used for oviposition until they were perished. The adults prey all stages of cotton mealybug. The grubs began to appear after one week of egg laying, while the pupae after three and half week, and the adults after approximately forty days (Sidhapara etal.,2013). Each cage was put under different ecological conditions (20, 24, 28, 32, 36 ºC temp. and 50, 60, 70, 80 % RH) having five replications.
The green cloth pieces along with folding papers were placed in the cages. The grubs of C. montrouzieri were examined in the rearing cages. Then, grubs were shifted on potato sprouts having cotton mealybug. The counting was done and potato sprouts with C. montrouzieri were then shifted to thejars, maintained for the pupation. These grubs were also separated from cages and after counting put into the petri dishes and mealybugs were given as food in each petri dish. The observations were made for pupation. It was also observed that the cannibalism was present in C. montrouzieri. To overcome this cannibalism a plastic sheet apparatus (1×1 feet) has been designed having 144 blocks of one square inch. A wire gauze was fitted as ventilation source, underside of this apparatus and a moving lid was prepared at the top to avoid the grub’s movement to another block. Data were collected to find most suitable conditions for rearing.
Techniques for releases: Two different techniques were used in releases of C. montrouzieri, because both larvae and adults are predaceous. In this respect, one thousand (1000) adults / larvae were released (Sarwar, 2016) in an acre in the cotton mealybug infested field and the data were collected after one, three and seven DAR.
Natural enemy field reservoir (NEFR) for the management of CMB: For on-farm mass multiplication of C. montrouzieri and A. bambawalei (= A. arizonensis), natural enemy field reservoir (NEFR) was established and further tested with field pretreatment population. For this purpose, a shed (15×10×12 feet) was constructed in the center of 5-acre cotton experimental area. Under the shed, eight field mass iron rearing trays (2×4×2 feet) were placed. In order to avoid mealybug escape from trays, grease bands were wrapped along these mass rearing trays and their legs were submerged in water filled bowls. A water channel of 9 inches width was constructed around the shed and filled with kerosene mixed water to prevent the entry of mealybug in the field. In each tray, mealybug infested leaves, twigs, branches, fruiting parts of host plant were placed to rear mealybug natural enemies which were replaced on regular basis. The assistance / small releases of both natural enemies were given at NEFR to support the population level. The preferred hosts were grown near NEFR to conserve natural enemies.
Three mealybug infested fields (100, 200 and 400 meters) away from the shed were selected and the data of natural enemies C. montrouzieri and A. bambawalei (= A. arizonensis) and mealybug were recorded before construction of shed (pretreatment data). After the construction, provision and application of all material, the data were recorded for natural enemies C. montrouzieri and A. bambawalei (= A. arizonensis) and mummified mealybug after seven, fifteen and twenty two days from nine (9) randomly selected plants. A set of nine plants was considered as a replication. The cost benefit ratio (CBR) of NEFR technology was also calculated (Bajwa et al., 2018).
Statistical Analysis: The data were analyzed statistically using Statistix 8.1 (Analytical software, 2005). Factorial ANOVA was done and means were compared following LSD test.
RESULTS
Fig. 1 presents the overall population trend of cotton mealybug (CMB) during 2012 and 2013. The results clearly revealed that CMB population per twig was recorded 41.35 and 70.70, at the end of May which was statistically different at fortnight interval. Population increased gradually and steadily while reaching at first peak on 25th July (331.75 and 347.25 per twig, during 2012 and 2013 respectively). The highest CMB peak reached on 10th August (381.25 and 422.35, during 2012 and 2013 respectively). Then per twig population of CMB started decreasing onward from 25th August (281.6 and 367.45) and minimum population was recorded on 25th September (28.6 and 30.40 during 2012 and 2013).
During the survey, six natural enemies including five predator species and one parasitoid were recorded predating / parasitizing the CMB population, during the both study years (table 1). The data also described that all the natural enemies remained present throughout the study period.
Fig. 2 reveals that 70% relative humidity with 28 °C temperature was found the most suitable and exhibited maximum A. bambawalei (= A. arizonensis) population. The highest parasitism on CMB, 64.83, 132.867 and 168.34 followed by 47.75 and 41.42, 68.68 and 51.276 and, 96.932 and 61.25 at 70% RH and 32 °C temperature, and 70% RH and 24 °C temperature, after seven, fourteen and twenty-one DAR, respectively. While minimum population and parasitism was recorded in condition with 50% RH and 20 °C temperature. The rearing of life stages of C. montrouzieri under four levels of RH and five levels of temperature revealed that 70% RH and 28 °C temperature was the optimum and exhibited duration, i.e., 2.72, 2.49, 1.831, 60.3 and 75.2 days to complete 1st, 2nd, 3rd instars and adult its life stage whereas 2.35 days taken by 4th instar grub at 70% RH & 24 °C temperature (Figure 3).
Table 1: Mean Trend of predators and parasitoid population associated with CMB during 2012 and 2013.
|
Brumus suturalis |
Scymnus coccivora |
Menochilus sexmaculatus |
Hyperaspis maindroni |
Chrysoperla carnea |
A.bambawalei parasitism |
25 May |
2.250 c |
3.250 d |
5.750 a |
6.750 a |
0.750 c |
38.563 a |
10 Jun |
5.750 a |
5.250 ab |
6.250 a |
1.250 bc |
1.250 bc |
20.695 b |
25 Jun |
5.500 a |
5.000 abc |
6.250 a |
1.000 bc |
1.000 c |
20.195 b |
10 Jul |
6.500 a |
6.500 a |
4.500 b |
2.250 b |
2.750 a |
11.805 c |
25 Jul |
6.250 a |
6.250 a |
4.250 b |
2.000 b |
2.500 ab |
16.108 bc |
10 Aug |
3.750 b |
4.250 bcd |
3.750 b |
1.250 bc |
1.250 bc |
16.527 bc |
25 Aug |
3.500 bc |
3.750 bcd |
3.500 b |
1.000 bc |
1.000 c |
11.438 c |
10 Sept |
3.250 bc |
3.500 cd |
1.250 c |
0.250 c |
1.750 abc |
43.708 a |
25 Sept |
3.000 bc |
3.250 d |
1.000 c |
0 |
1.500 abc |
42.807 a |
LSD value |
1.368 |
1.623 |
1.001 |
1.318 |
1.298 |
7.344 |
*Means showing same letter are similar with each other.
The most suited conditions for fecundity are 70% RH and 28 °C temperature recorded to produce maximum eggs, i.e., 238.43 eggs in a whole life. Whereas minimum number of eggs and maximum duration at 50% RH and 36 °C temperature.
The figure 4 showed that 407.04, 409.266 and 406.4 mean number of mealybugs were recorded before the releases of A. bambawalei (= A. arizonensis) in three selected fields. The rate of percent parasitism was recorded 40.31, 60.6 and 73.998, 32.16, 44.47 and 56.62 and 27.74, 39.33 and 49.91 after seven, fourteen and twenty one DAR, in case of mummies with host, cards and adults released fields, respectively.
Effective releases for larvae and adults of C. montrouzieri are presented in Table 4. The results showed that 402.92 and 403.89 mealybugs were examined before adult and larval release. The results indicated population decrease, i.e., 315.716 and 338.78 mealybugs after one day followed by 162.983 and 237.96, and 97.856 and 166.37 mealybugs after three and seven days, respectively. The results also indicated that C. montrouzieri adult population was more than larvae after one, three and seven days, respectively (Figure 5).
The results (Table 2) depicted that all the treatments differed significantly from each other. The data also showed the CMB population per twig was statistically similar with other, i.e., 536.67, 537.19 and 537.56 at 100, 200 and 400 meters, distance respectively on pre-treatment. This population per twig started to decrease slowly but steadily by leaving CMB population 294.72, 362.17 and 404.3, followed by 167.58, 251.11 and 297.64, and 32.11, 94.83 and 121.83 individuals after seven, fifteen and twenty-two days at 100, 200 and 400 meter distance, respectively.
In case of A. bambawalei (= A. arizonensis) the parasitism was statistically similar with other, i.e., 2.22, 2.083 and 1.83 at 100, 200 and 400 meters, distance respectively, on pre-treatment observation. This percent parasitism started to build-up slowly but steadily 19.528, 13.444 and 8.333 followed by 42.639, 33.25 and 24.139, and 56.694, 48.75 and 33.611 after seven, fifteen and twenty-two days at 100, 200 and 400 meter distance, respectively.
The results for C. montrouzieri predation was statistically similar with other, i.e., 0 at 100 and 200 meters, distance respectively, on pre-treatment observation. This predation started to build-up slowly 1.639 and 0.861 followed by 3.528 and 2.694, and 4.305 and 3.889 after seven, fifteen and twenty-two days at 100 and 200 meter distance, respectively.
Cost Benefit Ratio of NEFR Technology: The cost benefit ratio (CBR) reflected that with the expenditure of one rupee (01) on NEFR technology, the farmer got benefitted at the rate of 1.81 and 3.257 when the crop was damaged 20 to 50 percent by CMB (table 3).
Table 2: Trend of CMB, A. bambawalei and C. montrouzieri population at and nearby NEFR.
|
Distance (meter) |
Population of CMB |
Population of A. bambawalei |
Population of C. montrouzieri |
Pre-treatment data |
100 |
536.37a |
2.22 g |
0 |
200 |
537.19a |
2.083 g |
0 |
400 |
537.56a |
1.83 g |
- |
After 7 days |
100 |
294.72d |
19.528 e |
1.639cd |
200 |
362.17c |
13.444 f |
0.861d |
400 |
404.03b |
8.333 f |
- |
After 15 days |
100 |
167.58f |
42.639 c |
3.528ab |
200 |
251.11e |
33.25 d |
2.694bc |
400 |
297.64d |
24.139 e |
- |
After 22 days |
100 |
32.11 i |
56.694 a |
4.305a |
200 |
94.83 h |
48.75 b |
3.889ab |
400 |
121.83 g |
33.611 d |
- |
*Means showing same letter are similar with each other
Table 3: Cost benefit ratio of NEFR technology (01USD = 155 Rs.)
Cost of Mud Shed (15×10×12 feet) |
= Rs. 200,000/- (Useable for 10 years) |
Cost of Iron Trays |
= Rs. 12000/- @ 1500/ tray (Permanent) |
Labour Charges |
= Rs. 1000/- (Permanent) |
Cost of Grease |
= Rs. 5000/- |
Cost of Kerosene Oil |
= Rs. 5000/- |
Others |
= Rs. 4000/- |
Total Expenditure |
= Rs. 35300/- |
Average Production per Acre |
= 25 maunds |
Value per Acre |
= 25 × 2300 = Rs. 57500/- |
Crop damage ranged between 20 to 50% |
|
So |
|
Crop damage averted due to NEFR |
= 20% of net produce, i.e., 5 maunds |
Net Saving |
= 5 × 4 acre = 20 maunds |
Value |
= 20 × 2300 = Rs. 64000/- |
Cost Benefit Ratio (CBR) |
= 1: 1.813 |
Crop damage averted due to NEFR |
= 50% of net produce, i.e., 12.5 maunds/acre |
Net Saving |
= 12.5 × 4 acre = 50 maunds |
Value |
= 50 × 2300 = Rs. 1,15,000/- |
Cost Benefit Ratio (CBR) |
= 1: 3.257 |
DISCUSSION
The field surveys showed similar trend at all the locations (Basti Malook, Basti Laar, Billiwala, Makhdoom Rasheed and Sariwala), of Multan district. However, the peak population per twig was recorded 381.25 and 422.35, on 10th of August, during 2012 and 2013, respectively. It is further added that CMB remained prevalent on the cotton crop from May to end of September and the population pressure and intensity increased in 2013 study. This is because of regular evidence on the CMB under the field. Moreover, land was never fallow and each time cotton was sown in the same field so intensity of the pest multiplied. Six different natural enemies were found predating cotton mealybug. These natural enemies consisted of five predators and one parasitoid. These results are in agreement with Sreedevi et al. (2013) and Noureen et al. (2016) who recorded prevalence and destruction of cotton mealybug during Kharif season and then shifted to other plant species. The most favourable micro and macro climatic conditions such as temperature, relative humidity and wind speed encouraging the cotton mealybug population prevailed during the month of August.
Field prevalence and parasitism of A. bambawalei (= A. arizonensis) was observed by Ram et al. (2009) against cotton mealybug at Haryana, India. In Pakistan, Arif et al. (2012a and 2012b) surveyed cotton crop and recorded parasitoid, A. bambawalei (= A. arizonensis) and predators, Brumoides suturalis, Scymnus coccivora, Menochilus sexmaculatus and spiders. Cotton mealybug mummies (per 6 inch-twig) were observed on shoe flower followed by cotton, tomato, okra, sunflower, silvery and brinjal. Highest parasitism was observed 81.3% on shoe flower followed by cotton, tomato, sunflower, okra, silvery and brinjal. All these results are in line with ours.
Earlier researchers (Zain-ul-Abdin et al., 2013, Iqbal et al., 2016 and Zhang et al., 2016) worked in-vitro, comprehensively on the growth and biological characteristics of Aenasius bambawalei (= A. arizonensis) on rate of parasitism with respect to synchronization. Three different techniques (adults, mummies with cards and mummies with host) were used for A. bambawalei (= A. arizonensis) releases but mummies with host proved the most effective and efficient method than cards and adults.
The results showed that the CMB pre-treatment population per twig was 536.67, 537.19 and 537.56 at 100, 200 and 400 meters, distance. This population started to decrease slowly but steadily in response to build-up of natural enemy by reducing CMB population 294.72, 362.17 and 404.3, followed by 167.58, 251.11 and 297.64, and 32.11, 94.83 and 121.83 individuals after seven, fifteen and twenty-two days at 100, 200 and 400 meters distance, respectively. In the same pattern, the A. bambawalei (= A. arizonensis) population was also built-up in terms of number and distance (dispersal). The results showed that the A. bambawalei (= A. arizonensis) parasitism before reservoir construction was 2.22, 2.083 and 1.83 at 100, 200 and 400 meters, distance respectively. This percent parasitism started to increase steadily 19.528, 13.444 and 8.333 followed by 42.639, 33.25 and 24.139, and 56.694, 48.75 and 33.611 after seven, fifteen and twenty-two days at 100, 200 and 400 meter distance, respectively. Whereas the increasing trend observed in case of C. montrouzieri 1.639 and 0.861 followed by 3.528 and 2.694, and 4.305 and 3.889 after seven, fifteen and twenty-two days at 100 and 200 meter distance, respectively.
Classical biological control is control of insect with its native Bio-control agents whereas inundative means large releases of biological control agents to supplement the population in a definite period of time. Earlier investigations (Rosas-Garcia et al., 2009; Atif et al., 2011) evaluated different life stages of C. montrouzieri against citrus mealybug and concluded efficiency of all stages. But earlier instar had less demand to consume the administered feed, so these 1st and 2nd instars consumed lesser amounts of mealybugs whereas later developmental stages such as 3rd instar and adults showed more thrust of food. These stages devour the mealybugs in less time and consume higher number of mealybugs (Ahi et al., 2015) and hatching percentage is also increased remarkably. This study will not only provide the model for future P. solenopsis IPM programs but also will lead scientists towards its classical biological control in Pakistan.
Conclusions: Natural enemies especially A. bambawalei (= A. arizonensis) and C. montrouzieri can effectively manage the building population of CMB when they are conserved properly through establishment of natural enemy field reservoir (NEFR) and can reduce the crop losses.

Fig. 1: Trend of CMB population during both study years (2012 and 2013).
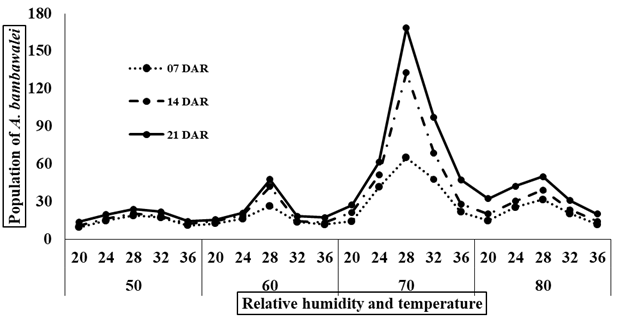
Fig. 2: Rearing of A. bambawalei under laboratory conditions.

Fig. 3: Rearing technique of C. montrouzieri.
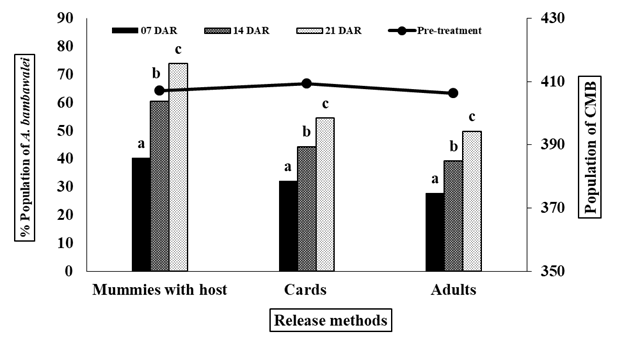
Fig. 4: Release techniques of A. bambawalei.
REFERENCES
- Agarwal, B.K., Das and M. Senchowdhuri. (1988). Biology and food relation of Micraspis discolor (F.) an aphidophagous coccinellidae in India. J. Aphidol. 2(1-2): 7-17.
- Ali, M., R. Perveen, M.J. Yousuf, S. Khawja and M. Amin. (2014). Predatory potential of five coccinellid predators against cotton mealy bug Phenacoccus solenopsis (Tinsley) in laboratory and field conditions from Sindh, Pakistan. Pak Entomol. 36(1): 7-12.
- Analytical software, 2005. Statistix for windows. Analytical software, Tallahassee, Florida.
- Arif, M.J., M. Rafiq, S. Wazir, N. Mehmood and A. Ghaffar. (2012a). Studies on cotton mealybug, Phenacoccus solenopsis (Pseudococcidae: Homoptera), and its natural enemies in Punjab, Pakistan. Int. J. Agri. Biol. 14: 557-562.
- Arif, M.J., M.D. Gogi, M. Arshad, A. Ashraf, A. Suhail, Zain-ul-Abdin, A. Nawaz. (2012b). Host-plants mediated population dynamic of cotton mealybug, Phenacoccus solenopsis (Hemiptera: Pseudococcidae) and its parasitoid, Aenasius bambawalei Hayat (Hymenoptera: Encyrtidae). Pak. Entomol. 34(2): 179-184.
- Atif, J. Y. M., K. S. A. Naser, M. E. Elsherif and A.A. Al-Humiari. (2011). Functional response of the adult predator of Cryptolaemus montrouzieri (Mulsant), reared on the third nymphal instar of Planococcus citri (Risso). Egyptian J. Biol. Pest Control. 21(2): 267-270.
- Ahi, G. A. A., A. Afshari, V. Baniameri, H. Dadpour, M. Yazdanian and A. Golizadeh. (2015). Laboratory survey on biological and demographic parameters of Cryptolaemus montrouzieri (Mulsant) (Coleoptera: Coccinellidae) fed ontwo mealybug species. J. Crop Prot. 4 (3): 267-276.
- Bajwa, B., M. S. Mazhar, M. K. Bashir and S. F. Honey. 2018. Environmental, Economic and Social Impact of Biological Control Interventions in Papaya Farming in Sindh, Pakistan. Pak. J. Life Soc. Sci. (2018), 16(1): 27-34.
- Economic Survey of Pakistan, 2015-16. Economic Survey of Pakistan, 2015-2016. In: Government of Pakistan. Finance Division Economic Adviser's Wing, Islamabad.
- Fallahzadeh M, Japoshvili G, Abdimaleki R, Saghaei N. 2014. New records of Tetracneminae (Hymenoptera, Chalcidoidea, Encyrtidae) from Iran. Turk J Zool. 38: 515–518.
- Gautam, R.D. (1996). Reports on the use of coccinellid predators for the management of hibiscus (pink) mealy bug in the Caribbean. Centeno (Trinidad and Tobago): Ministry of Agriculture, Land and Marine Resources Central Experiment Station. vp. (07774/H10.G3).
- Iqbal, M.S., Zain-ul-Abdin, M. Arshad, S. K. Abbas, M. Tahir, A. Jamil and A. Manzoor. (2016). The role of parasitoid age on the fecundity and sex ratio of the parasitoid, Aenasius bambawalei (Hayat) (Hymenoptera: Encyrtidae). Pakistan J. Zool. 48(1): 67-72.
- Joshi, M.D., P.G. Butani, V.N. Patel and P. Jeyakumar. (2010). Cotton mealy bug, Phenacoccus solenopsis. Agric. Rev. 31: 113–119.
- Mahmood, R., M.N. Aslam, G.S. Solangi and A. Samad. (2011). Historical perspective and achievements in biological management of cotton mealybug Phenacoccus solenopsis Tinsley in Pakistan. Proceedings of 5th Meeting of ICAC’s Asian Cotton Research and Development Network, 23-25 Feb., 2011, held at Lahore, Pakistan.
- Mark, P. C. and P. J. Gullan. (2005). A new pest of tomato and other records of mealy bugs (Hemiptera: Pseudococcidae) from Espírito Santo, Brazil. Zootaxa. 964: 1–8.
- Mulsant E. Supplement a la monographie des coleopteres trimeres securipalpes. Opuscula Entomologica 1853; 3:1–178.
- Noureen, N., M. Hussain, S. Fatima and M. Ghazanfar. 2016. Cotton Mealybug Management: A Review. Journal of Entomology and Zoology Studies. 4(4): 657-663.
- Ram, P., R. K. Saini and Vijaya. (2009). Preliminary studies on field parasitization and biology of Solenopsis mealybug parasitoid, Aenasius bambawalei Hayat (Encyrtidae: Hymenoptera). J. cotton Res. Dev. 23(2): 313-315.
- Rosas-Garcia, N. M., E. P. Duran Martinez, E.D.J. de Luna-Santillana, and J.M. Villegas-Mendoza. (2009). Potential of depredation of Cryptolaemus montrouzieri Mulsant Hacia Planococcus citri Risso, Southw. Entomol. 34(2): 179-188.
- Saeed, S., M. Ahmad and Y.J. Kwon. (2007). Insecticidal control of the mealybug Phenacoccus gossypiphilous (Hemiptera: Pseudococcidae), a new pest of cotton in Pakistan. Entomol. Res. 37:76-80.
- Sarwar, M., (2016). Biological control to maintain natural densities of insects and mites by field releases of lady birds (Coleoptera: Coccinellidae). Inter. J. Ent. Nematol. 2(1): 21-26.
- Sidhapara, M.R., S. G. Dumaniya, M. B. Patel and N. V. Patel, (2013). Biology of lady bird beetle, Cryptolaemus montrouzieri (Mulsant) on cotton mealy bug, Phenacoccus solenopsis (Tinsley). The Bioscan. 8(2): 523-527.
- Sreedevi G., Y.G.Prasad, M. Prabhakar, G.R Rao, S. Vennila and B. Venkateswarlu. 2013. Bioclimatic Thresholds, Thermal Constants and Survival of Mealybug, Phenacoccus solenopsis (Hemiptera: Pseudococcidae) in Response to Constant Temperatures on Hibiscus. PLoS ONE 8(9): e75636. doi:10.1371/ journal.pone.0075636.
- Tinsley JD. 1898. Notes on Coccidae with descriptions of new species. Canadian Entomologist 30, 317-320.
- Yonas, M.W, T. Qadir, K. Mubeen and A. Shakoor, 2019. Yield declining trend of cotton in Pakistan: Different agronomic and biological approaches for control of cotton mealy bug (Phenacoccus solenopsis). J. Glob. Innov. Agric. Soc. Sci. 2019, 7(2): 65-.72.
- Zain-ul-Abdin, F. Hussain, M. A. Khan, S. K. Abbas, A. Manzoor and H. Shaina. (2013). Reproductive Fitness of Mealybug Parasitoid, Aenasius bambawalei Hayat (Hymenoptera: Encyrtidae). World Appl. Sci. J. 26 (9): 1198-1203.
- Zhang, J., J. Huang, Y. Lu and T. Xia. (2016). Effects of temperature and host stage on the parasitization rate and offspring sex ratio of Aenasius bambawalei Hayat in Phenacoccus solenopsis Tinsley. Peer J.4: 1-13.
|





